JAMUNI LAL COLLEGE HAJIPUR VAISHALI B R A

JAMUNI LAL COLLEGE HAJIPUR, VAISHALI (B. R. A. BIHAR UNIVERSITY, MUZAFFARPUR) DR. BIJOYENDRA NARAYAN (PRINCIPAL) DEPARTMENT OF PHYSICS JAMUNI LAL COLLEGE, HAJIPUR
ROTATION SPECTRA OF A DIATOMIC MOLECULE For B. Sc. Physics (Hons) Part–III Paper-VII(C)

INTRODUCTION �To excite a molecule energy is given from outside. The amount of energy required for rotation of the molecule is the least. If the energy amount is greater than this least amount vibration of the molecule is found, and in case of still higher energy electronic excitation is observed. Let the total energy received by a molecule is E, then

E= Erot + Evib + Eel Erot denotes the rotation energy. Evib denotes the vibrational energy. Eel denotes the electronic energy. If due to external supply of energy to a molecule the total energy of the molecule is ∆E, then ∆E= ∆Erot + ∆Evib + ∆Eel i. e. ∆Erot consumes 0. 001 ev(least) ∆Evib consumes ~0. 1 ev ∆Eel requires ~1 ev

Thus, depending upon the order of supply of external energy to the molecule three kinds of spectra are observed due to transitions between concerned levels: 1. Rotation Spectra 2. Vibration Spectra 3. Electronic Spectra

ROTATION SPECTRA Let us discuss rotation spectra of a diatomic molecule, which is the concern of the present lecture. What is a diatomic molecule? A diatomic molecule consists of two nuclei corresponding to two atoms, A and B; if the diatomic molecule considered is denoted by AB. The electrons contained in these atoms exist in a number of energy levels. When excited transition of electrons take place from an orbit of low energy to that of higher energy


� Now let us begin the topic exactly. What is the requirement for having rotation spectra from a diatomic molecule (linear)? � Rotational spectra are exhibited only by the molecules having permanent (electric) dipole moment. Only such molecules are able to interact with exciting electromagnetic radiation. Pure rotational spectra occur in the infrared region, which is from 700 nm to 1 mm. It is observed in absorption. It consists of a series of absorption maxima which are equidistant on a wavenumber scale as given below,

RIGID ROTATOR MODEL: A rotating diatomic molecule can be assumed to be rigid rotator with a fixed inter nuclear distance. The presence of fixed inter nuclear distance between the two atoms (diatomic) is itself the criterion of a being a rigid rotator (Fig. -III).
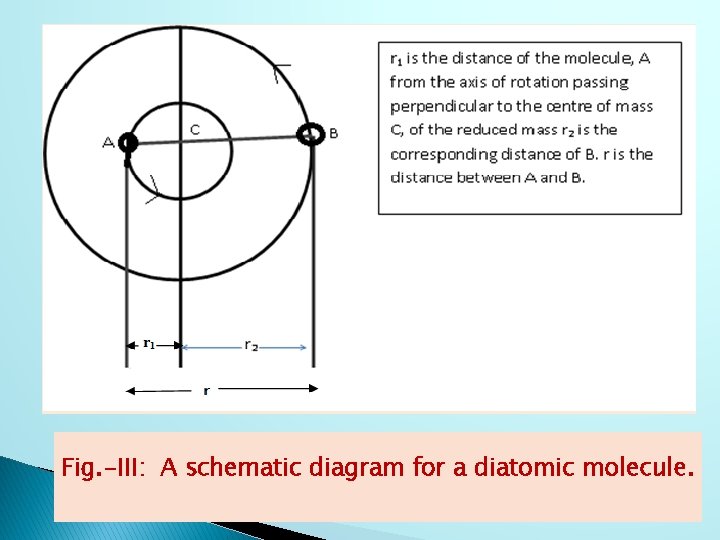
Fig. -III: A schematic diagram for a diatomic molecule.
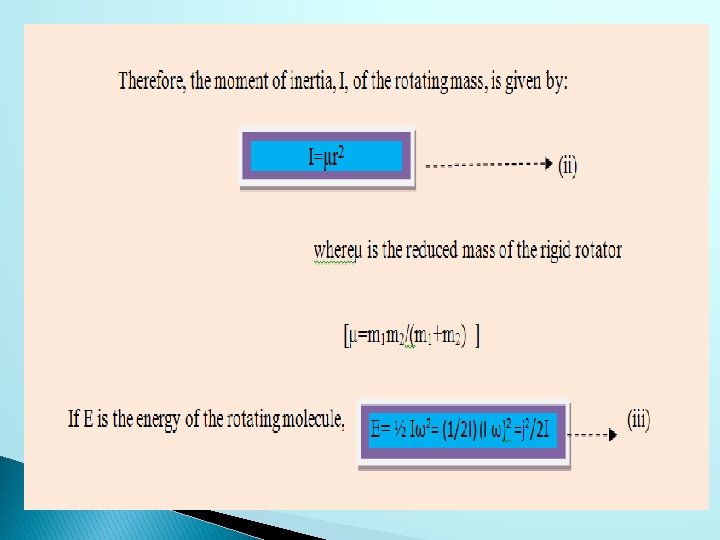


jf is the rotational quantum number of the final state and ji is the rotational quantum number of the initial state.

By selection rule one knows that the molecule can change the rotational quantum number by +1 or -1 Change of quantum number by more than +1 or -1 are forbidden. A representative diagram for rotation spectra is shown below:

Fig. IV : Energy Levels of rotational spectrum

(Spectral lines due to transitionsare shown in the lower part of the diagram)



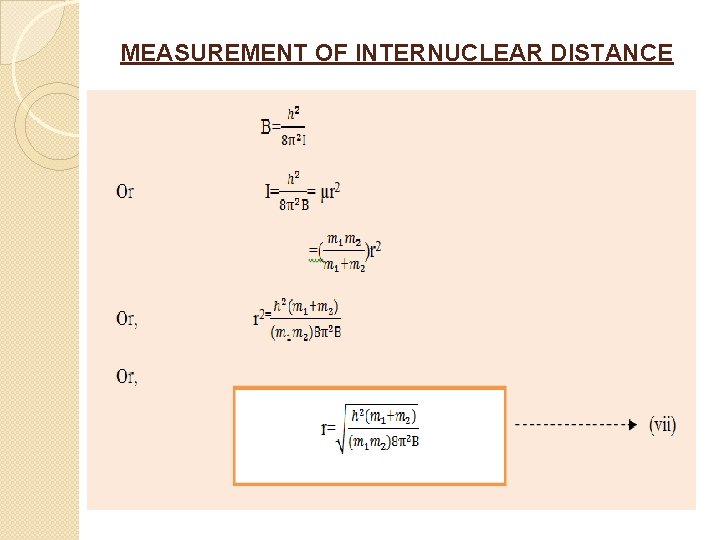
MEASUREMENT OF INTERNUCLEAR DISTANCE

HOW INTENSE WILL BE THE SPECTRAL LINE ? Under this heading we want to know the relative intensities of the spectral lines. Are all the lines of the spectrum equally intense? To answer this question one has to find out the probability of a change of quantum number between energy levels. As we know the probability of change as per ∆j=± 1 rule is almost equally likely to occur.

It means each spectral line should be equally intense. But, this is not the case. Since the number of molecules in each energy level is different, the number of molecules in each level taking part in transition will be different. Also, the intrinsic probabilities of transition are the same. That is why the line intensities are proportional to the initial number of molecules in each level. As per the Boltzmann Distribution law,


We see that with the molecular population in each level decreasing exponentially, the number of degenerate levels available increases rapidly with j. Thus, the total relative population at an energy level, Ej is given as,
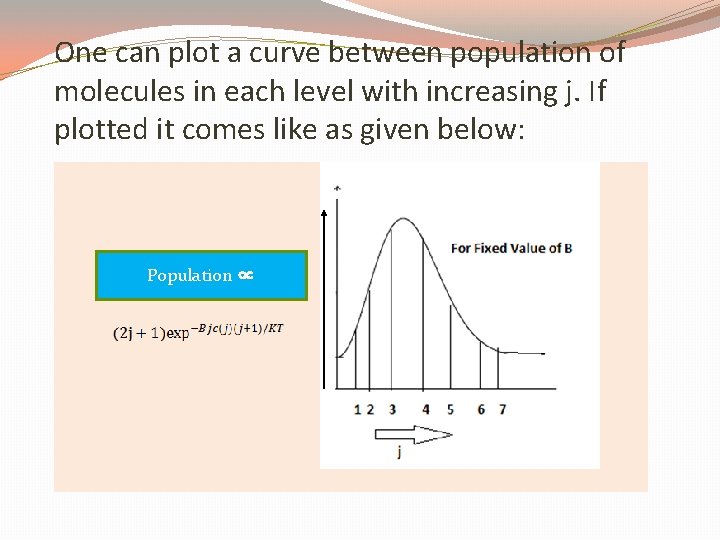
One can plot a curve between population of molecules in each level with increasing j. If plotted it comes like as given below: Population ∝


It means transitions between levels with very low or very high j values are having small intensities while it is maximum at or near the j value given by equation.
- Slides: 27