Jaggregates and Haggregates 1 A 1 butanol magenta
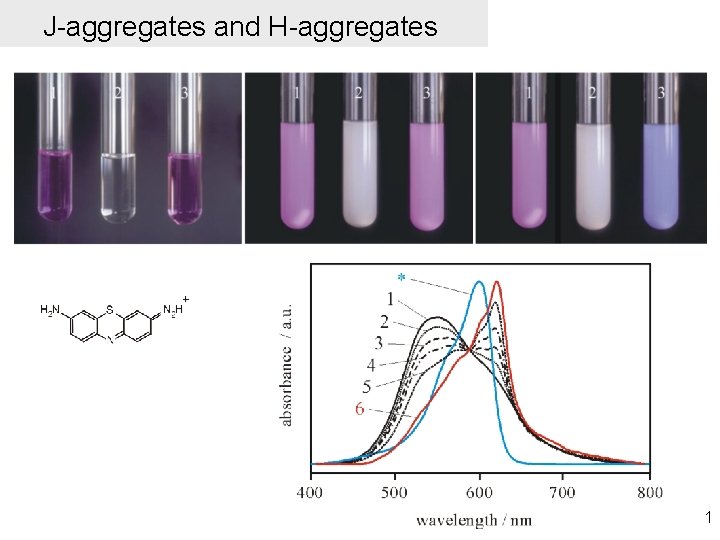
J-aggregates and H-aggregates 1
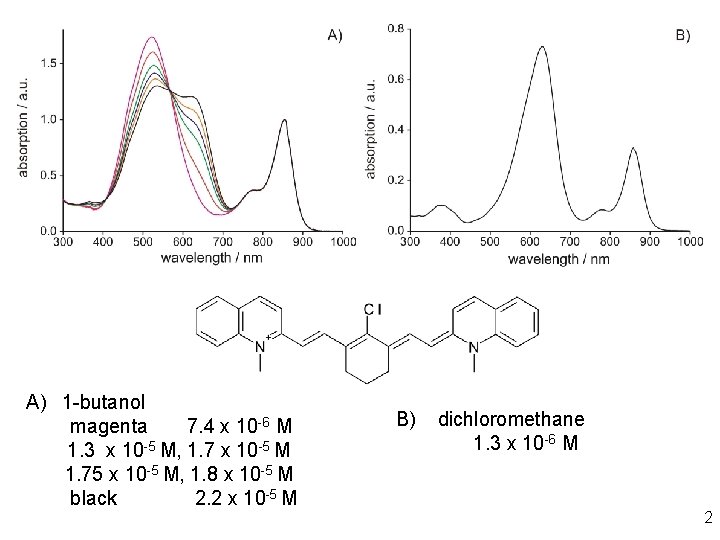
A) 1 -butanol magenta 7. 4 x 10 -6 M 1. 3 x 10 -5 M, 1. 7 x 10 -5 M 1. 75 x 10 -5 M, 1. 8 x 10 -5 M black 2. 2 x 10 -5 M B) dichloromethane 1. 3 x 10 -6 M 2
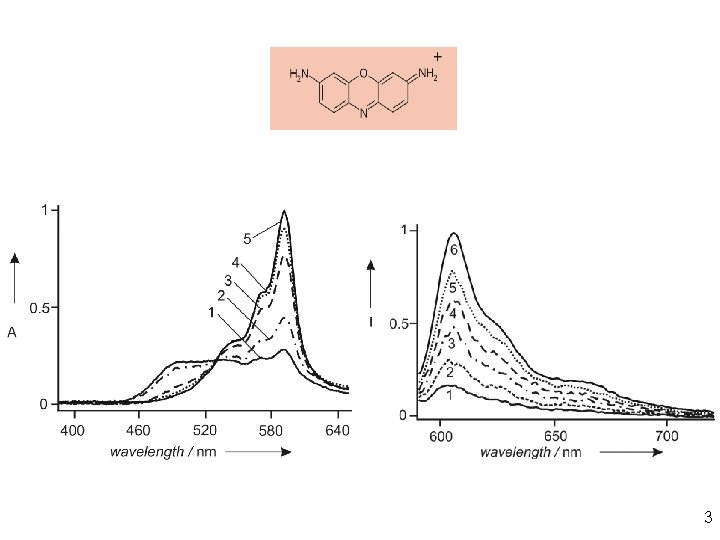
3
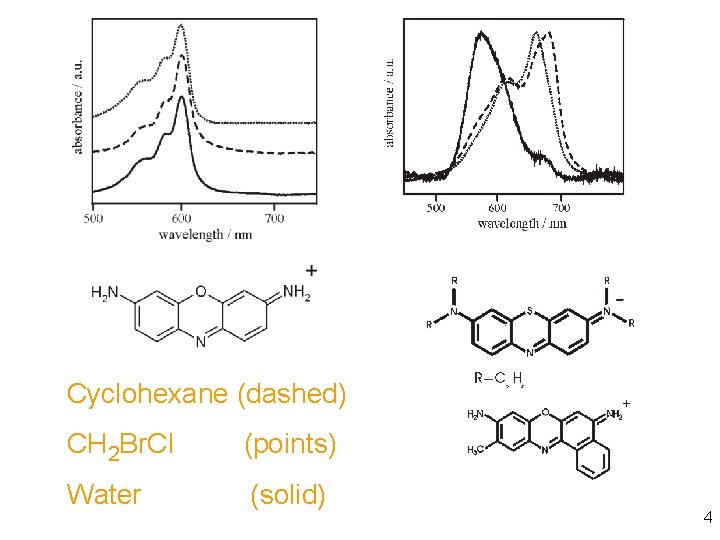
Cyclohexane (dashed) CH 2 Br. Cl (points) Water (solid) 4

Interactions between 2 molecules. One of them is in an electronically excited state Yi : Electronic wave function for the initial excited state (D excited but not A) Yf : Electronic wave function for the final excited state (A excited but not D) Interaction H’ between the initial and the final state: 5
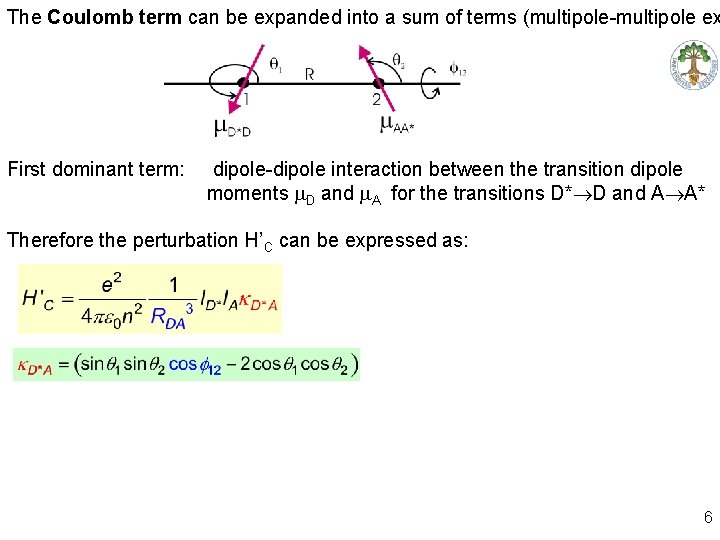
The Coulomb term can be expanded into a sum of terms (multipole-multipole ex First dominant term: dipole-dipole interaction between the transition dipole moments m. D and m. A for the transitions D*®D and A®A* Therefore the perturbation H’C can be expressed as: 6

Weak interaction between donor and acceptor molecules Strong interaction: the electronic spectra of a mixture of donors and acceptors are different from those of their diluted solutions. Weak interaction: the electronic spectrum of a mixture of donors and acceptors is a superposition of the spectra of the diluted solutions. Medium and exciton theory strong interaction: molecular orbital theory Only the Coulomb term plays a role for weak interactions. The exchange term requires orbital overlap between the D* and A, which causes larger interaction. 7

Only the Coulomb term plays a role for weak interactions, since the exchange term requires orbital overlap between the D* and A, which causes larger interaction. l. D and l. A = coordinates of the electrons belonging to D and A electronic transition moments 8
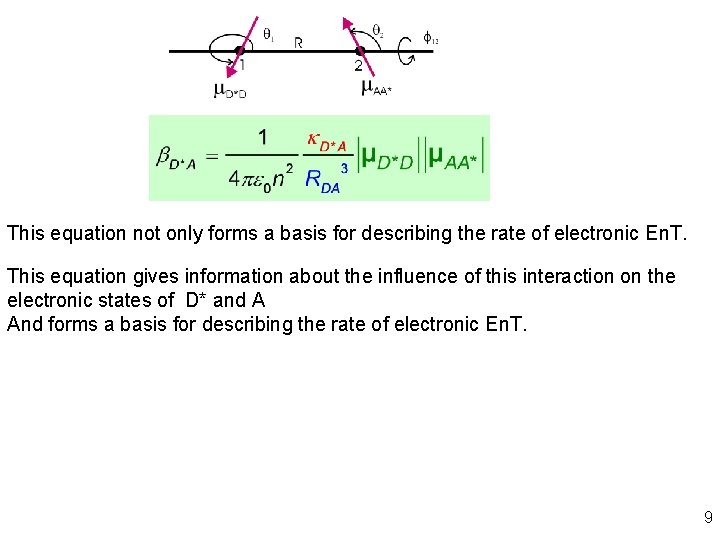
This equation not only forms a basis for describing the rate of electronic En. T. This equation gives information about the influence of this interaction on the electronic states of D* and A And forms a basis for describing the rate of electronic En. T. 9

J-aggregates and H-aggregates (Scheibe 1932) Pair of molecules Ai and Ak at a distance R. R is so large that the interaction in the electronic ground state is negligibly small. Electronically excited states: A*i…Ak resp. Ai…A*k Negligible overlap of the wave functions between (A*i and Ak ) and (Ai and A*k). This does not necessarily mean that the interaction between the electronically excited states A*i…Ak and Ai…A*k is so weak that the splitting of these states is negligible. Ground state: Energy E 0 Excited state: Energy E 1 H’C caused by the electronic transition dipole moments. The excited state is described by a linear combination of 10

The excitation is collective or delocalized. symmetric antisymmetric In both stationary states F+ and Fthe excitation is on both molecules i and k. Phase relation of the wave functions which describe the interaction between A*i…Ak and Ai…A*k caused by m. AA* (k=± 2). The node corresponding to the minus sign in F- is an excitation node (not an electron orbital node). At an excitation node, the phase relation between the transition moments of the respective molecular centres change phase 11
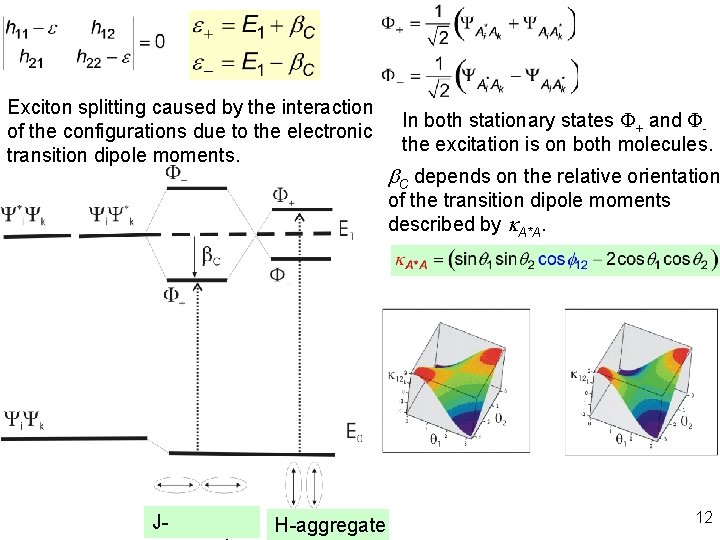
Exciton splitting caused by the interaction of the configurations due to the electronic transition dipole moments. In both stationary states F+ and Fthe excitation is on both molecules. b. C depends on the relative orientation of the transition dipole moments described by k. A*A. J- H-aggregate 12
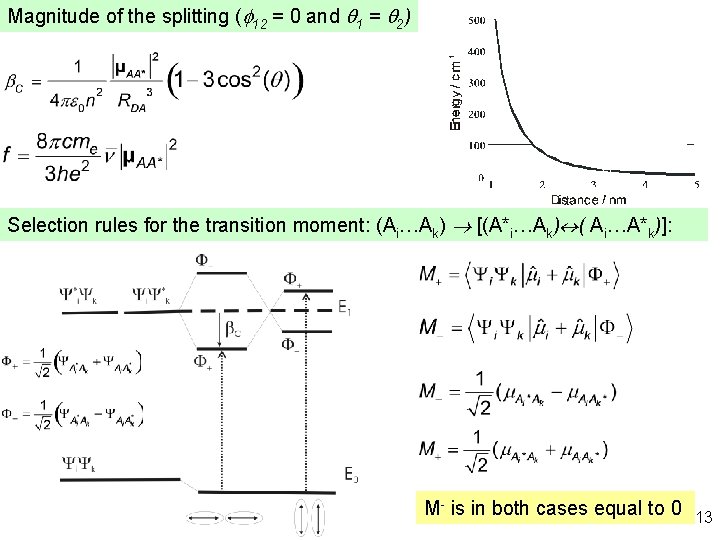
Magnitude of the splitting (f 12 = 0 and q 1 = q 2) Selection rules for the transition moment: (Ai…Ak) ® [(A*i…Ak) «( Ai…A*k)]: M- is in both cases equal to 0 13
- Slides: 13