IYNT 2014 TEAM GEORGIANS PROBLEM N 8 DROPLET

IYNT 2014 TEAM “GEORGIANS” PROBLEM N◦ 8 DROPLET reporter: Ekaterine Dadiani

About problem: 2 Droplet Place a droplet of salt water on a glass plate and study the process of drying. How does the deposit of dried droplets depend on the salinity of water? Perform the same with a droplet of black tea. Team „Georgians“ – Problem no. 8 – droplet

Presentation Plan: 3 basic theoretical model for drop drying; Theoretical observation; Experiments; difference between black tea drop and salty water droplet- drying process; Comparison of experiments’ and theoretical observations; Conclusion; Team „Georgians“ – Problem no. 8 – droplet

Wetting & surface roughness: two extreme cases 4 Roughness “enhances” wetting Team „Georgians“ – Problem no. 8 – droplet

Basic theory: 5 When a spilled drop dries on a solid surface, it leaves a dense, ring-like stain along the perimeter. • Pictures a and b show an increment of evaporation viewed in cross section. Picture a shows the result of evaporation without flow: the droplet shrinks. Picture b shows the compensating flow needed to keep contactno. line 8 – droplet Team „Georgians“ – the Problem • Contact line is pinned; Outward flow because losses, caused by evaporation have to be compensated.
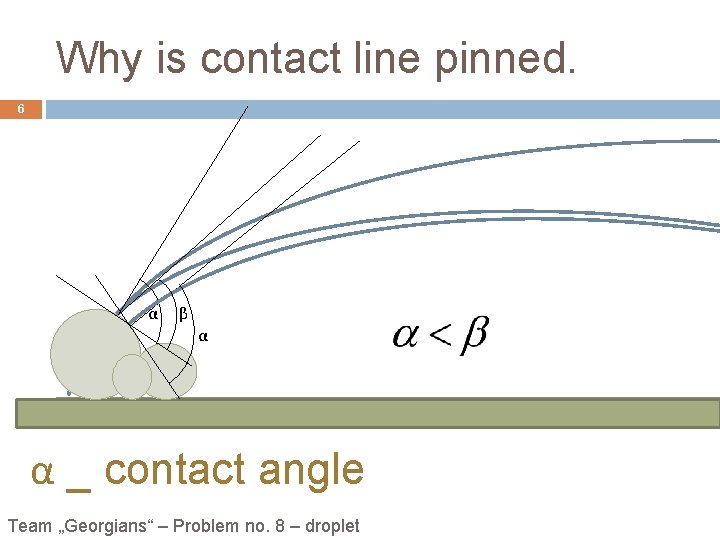
Why is contact line pinned. 6 α β α α _ contact angle Team „Georgians“ – Problem no. 8 – droplet

Theoretical observation; 7 small, circular drop of fixed radius l, slowly drying on a solid surface. h(0, t) h(r , t) r l V 0 - Volume of drop; f - The volume of water that evaporates from a unit area per unit of time; t - Time period; r l Team „Georgians“ – Problem no. 8 – droplet h(0, t) r DV 1 - volume of water evaporated inside the r; DV 1` - volume of water in which light part decrease. DV 2` - volume of water in which dark part decrease
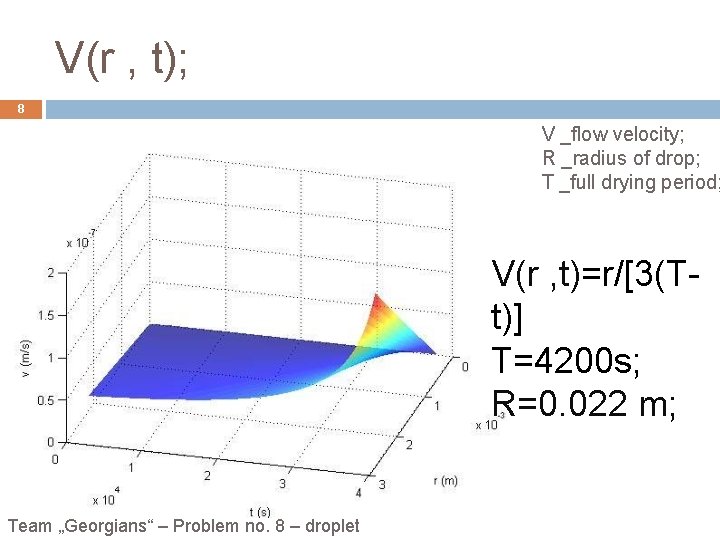
V(r , t); 8 V _flow velocity; R _radius of drop; T _full drying period; V(r , t)=r/[3(Tt)] T=4200 s; R=0. 022 m; Team „Georgians“ – Problem no. 8 – droplet
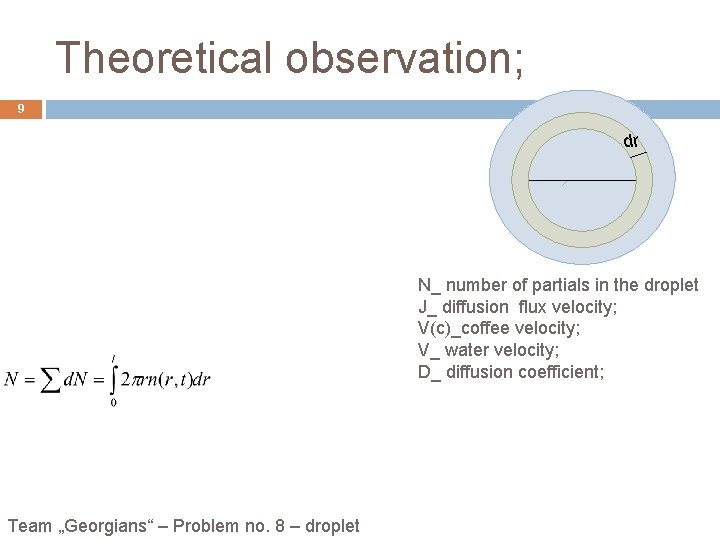
Theoretical observation; 9 dr N_ number of partials in the droplet J_ diffusion flux velocity; V(c)_coffee velocity; V_ water velocity; D_ diffusion coefficient; Team „Georgians“ – Problem no. 8 – droplet
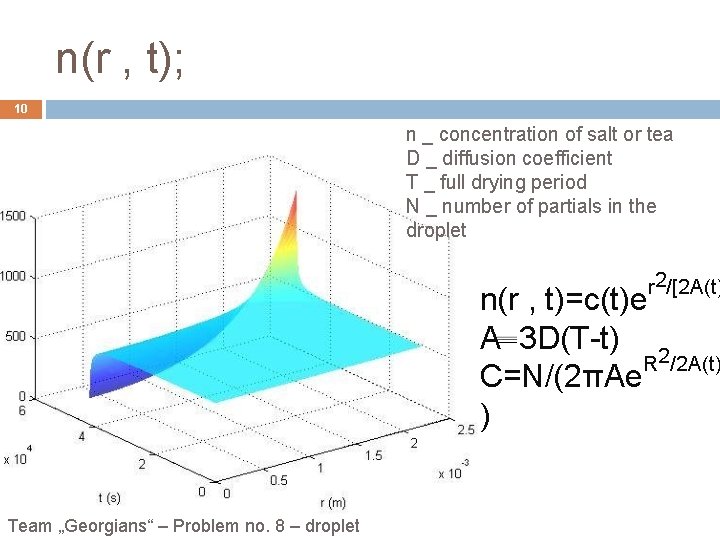
n(r , t); 10 n _ concentration of salt or tea D _ diffusion coefficient T _ full drying period N _ number of partials in the droplet r 2/[2 A(t) n(r , t)=c(t)e A 3 D(T-t) 2 R /2 A(t) C=N/(2πAe ) Team „Georgians“ – Problem no. 8 – droplet
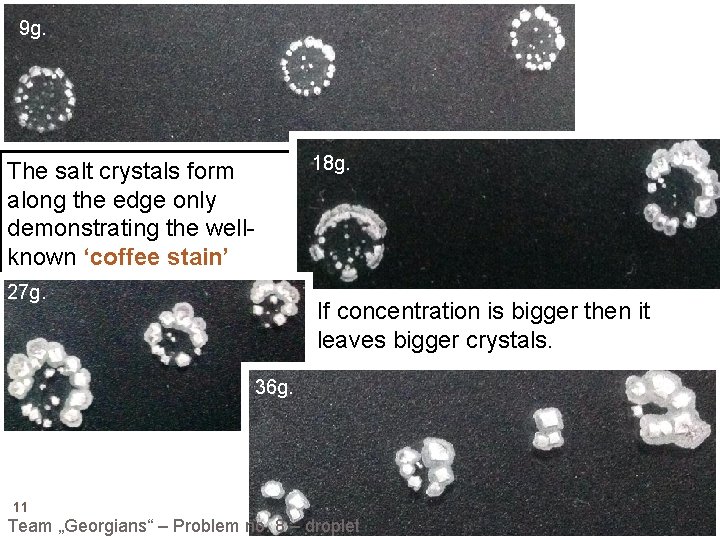
9 g. The salt crystals form along the edge only demonstrating the wellknown ‘coffee stain’ effect. 27 g. 18 g. If concentration is bigger then it leaves bigger crystals. 36 g. 11 Team „Georgians“ – Problem no. 8 – droplet

12
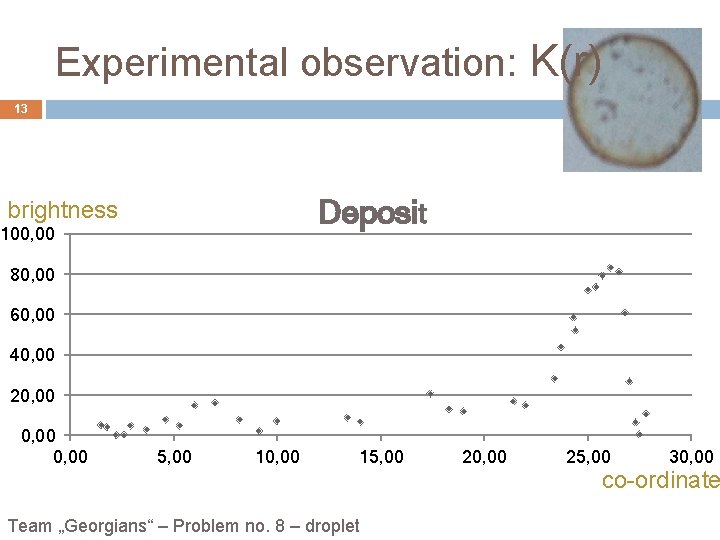
Experimental observation: K(r) 13 Deposit brightness 100, 00 80, 00 60, 00 40, 00 20, 00 5, 00 10, 00 15, 00 Team „Georgians“ – Problem no. 8 – droplet 20, 00 25, 00 30, 00 co-ordinate
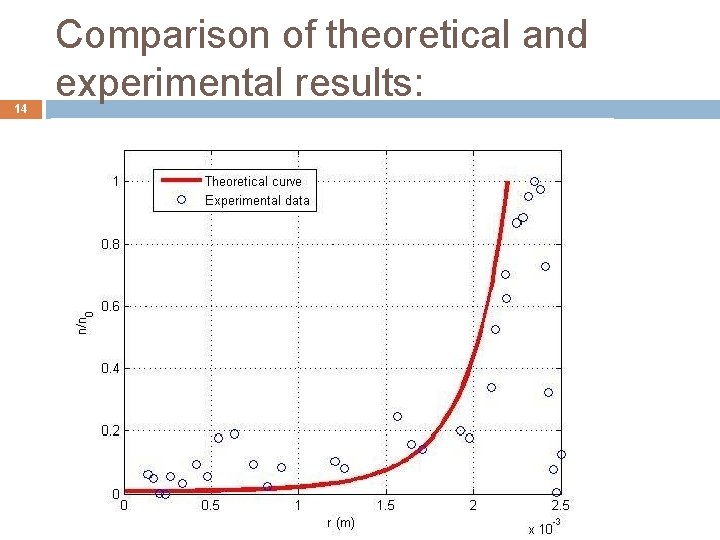
14 Comparison of theoretical and experimental results:
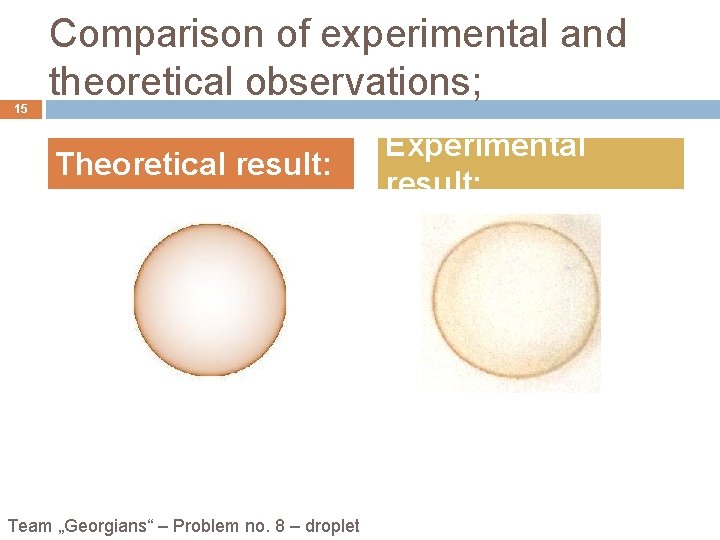
15 Comparison of experimental and theoretical observations; Theoretical result: Team „Georgians“ – Problem no. 8 – droplet Experimental result:

Dependence: 16 Droplet � Surface tension; � Concentration of particles; � Temperature; Surface � Hydrophobic surface → smaller stain; evaporates slowly; � Hydrophilic surface → larger stain; evaporates fast; � Particles cause a structure in the stain; Other � Environmental � Humidity temperature; of air; Team „Georgians“ – Problem no. 8 – droplet

Conclusion: 17 Contact line is pinned. Salty water droplet and black tea droplet leave most of dense along the perimeter. Flow velocity is bigger at the edge then at the center. Bigger concentrated salty water leaves bigger crystals at the edge. r 2/[2 A(t)] n(r , t)=c(t)e V(r , t)=r/[3(T-t)] Team „Georgians“ – Problem no. 8 – droplet

References: 18 http: //stilton. tnw. utwente. nl/people/gelderblom/docs/thesis_ HGelderblom. pdf http: //arxiv. org/ftp/arxiv/papers/1208. 0397. pdf http: //jfi. uchicago. edu/~tten/Coffee. drops/Nagel. 7. 4. pdf http: //www. newton. dep. anl. gov/askasci/chem 03/ch em 03950. htm http: //jfi. uchicago. edu/~tten/Coffee. drops/Deegan. t hesis. pdf http: //www. newton. dep. anl. gov/askasci/chem 03/ch em 03950. htm http: //www. wisegeek. com/what-iscrystallization. htm Team „Georgians“ – Problem no. 8 – droplet

Thanks for attention!
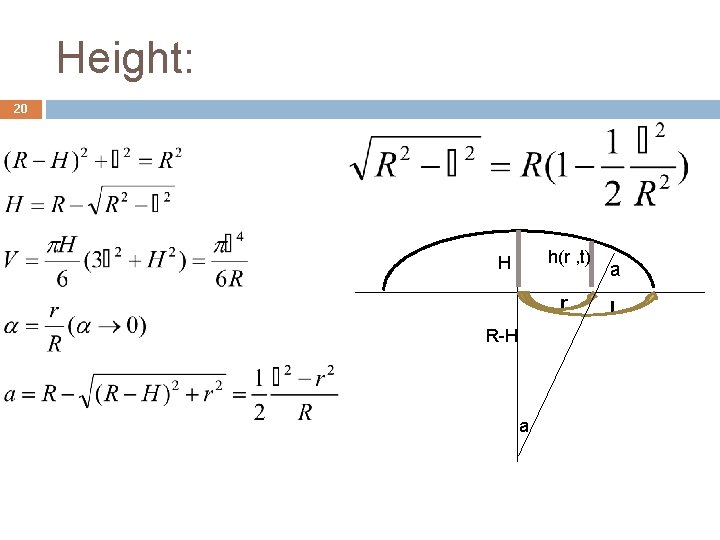
Height: 20 h(r , t) H r R-H a a l

It’s interesting: 21 How can we avoid “coffee ring effect”. The mechanism of the coffee ring effect used perfectly spherical particles, which break away from the surface of the water easily, allowing them to be transported from the middle of the droplet to the edges. Ellipsoid particles, on the other hand, are resistant to such flow. They dry much more evenly. If someone were to manufacture coffee that somehow broke into ellipsoid particles when suspended in water, it would leave nice, even-colored stains. Team „Georgians“ – Problem no. 8 – droplet

Time dependence: 22 Time dependence from: Size of droplet Angle – Subsurface/droplet θ _contact angle v _flow velocity * [Capillary flow as the cause of ring Team of Georgia – Problem no. 8 – droplet stains from dried liquid drops: Robert D. Deegan, Olgica Bakajin, Todd F. Dupont, Greg Huber, Sidney R. Nagel, Thomas A. Witten, James Franck Institute, 5640 South Ellis Ave, Chicago,

23 The contact line pinning was studied using video microscopy to measure drop radius during evaporation of different suspensions, and it was concluded that in all cases the contact line remains pinned until the final stage of evaporation.
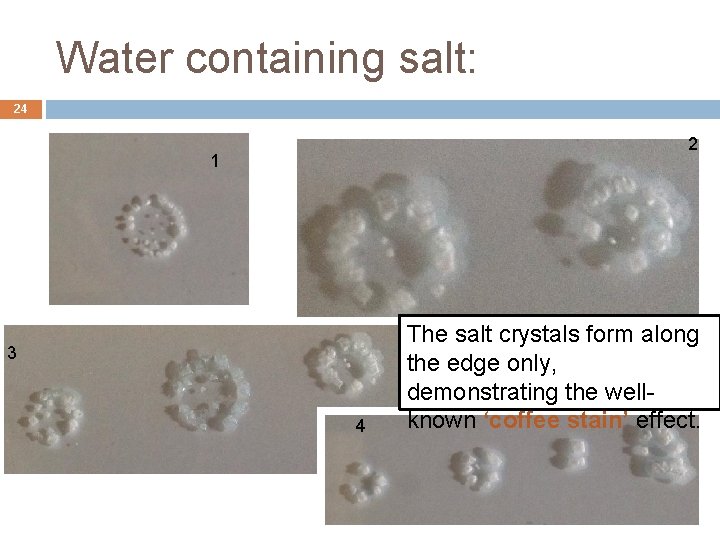
Water containing salt: 24 2 1 3 4 The salt crystals form along the edge only, demonstrating the wellknown ‘coffee stain’ effect.
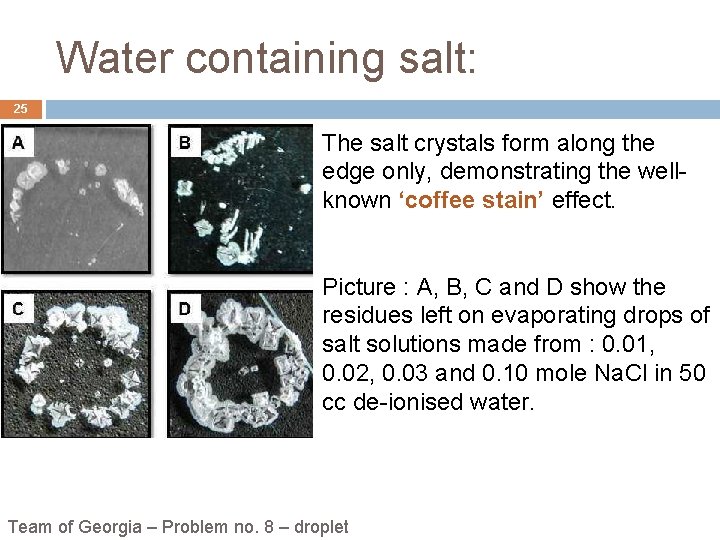
Water containing salt: 25 The salt crystals form along the edge only, demonstrating the wellknown ‘coffee stain’ effect. Picture : A, B, C and D show the residues left on evaporating drops of salt solutions made from : 0. 01, 0. 02, 0. 03 and 0. 10 mole Na. Cl in 50 cc de-ionised water. Team of Georgia – Problem no. 8 – droplet

Colloidal solutions containing salt: 26 Figure : A and B show photographs of the two films with 0. 01 mol Na. CL, 0. 5 g potato starch in 50 ml water on glass and PP. C and D show films with 0. 15 mol Na. Cl, other components remaining same on glass and PP. Team of Georgia – Problem no. 8 – droplet

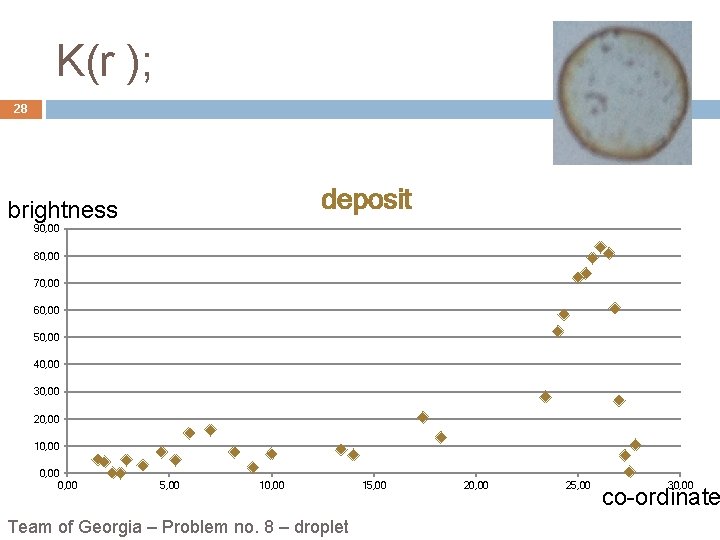
K(r ); 28 deposit brightness 90, 00 80, 00 70, 00 60, 00 50, 00 40, 00 30, 00 20, 00 10, 00 5, 00 10, 00 Team of Georgia – Problem no. 8 – droplet 15, 00 20, 00 25, 00 30, 00 co-ordinate
- Slides: 28