ITS NOT EASY BEING WHEEZY THE USE OF

IT’S NOT EASY BEING WHEEZY: THE USE OF BUDESONIDE- FORMOTEROL AS-NEEDED FOR MILD ASTHMA Alyssa N. Romaine, BS, Pharm. D PGY-1 Pharmacy Resident Erie VAMC Erie, PA

DISCLOSURE ØThe presenter has no conflicts of interest ØThe views expressed in this presentation are those of the presenter and do not necessarily reflect the position or policy of the Department of Veterans Affairs or the United States government ØThis presentation is the result of work supported with resources and the use of facilities at the Erie VA Medical Center, Erie, PA
Outline the major change in the 2019 Global Initiative for Asthma (GINA) guidelines LEARNING OBJECTIVES Discuss budesonide-formoterol’s place in therapy for mild asthma based on recent literature Recognize the dose and dosage form that budesonide-formoterol is supplied as

BACKGROUND
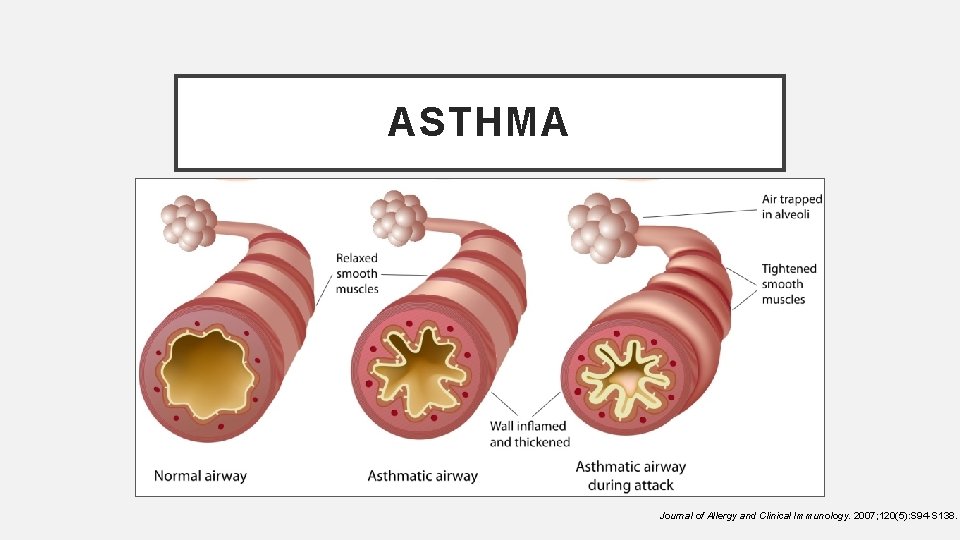
ASTHMA Journal of Allergy and Clinical Immunology. 2007; 120(5): S 94 -S 138.
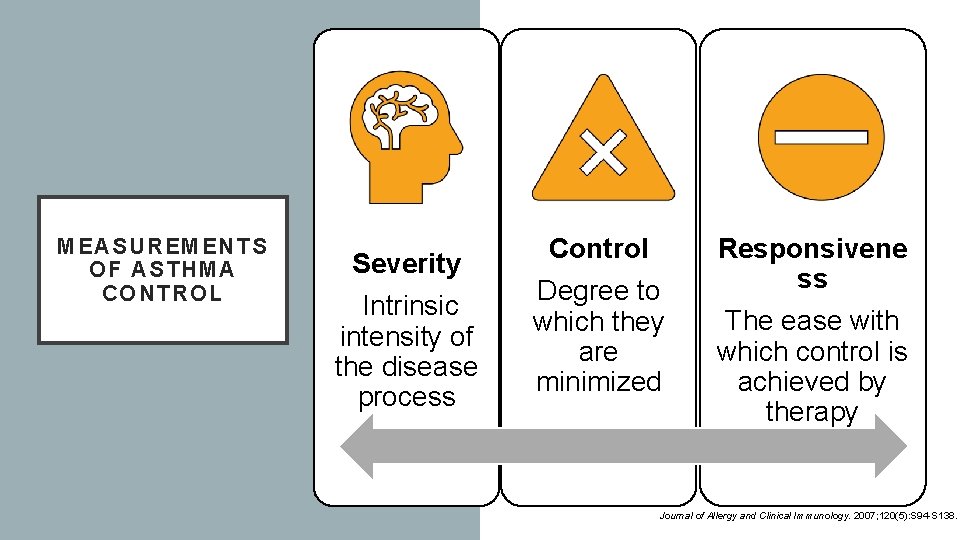
MEASUREMENTS OF ASTHMA CONTROL Severity Intrinsic intensity of the disease process Control Degree to which they are minimized Responsivene ss The ease with which control is achieved by therapy Journal of Allergy and Clinical Immunology. 2007; 120(5): S 94 -S 138.

CONSEQUENCES OF UNCONTROLLED ASTHMA Airway remodeling Scarring of the lungs Permanent decline in respiratory function Journal of Allergy and Clinical Immunology. 2007; 120(5): S 94 -S 138.
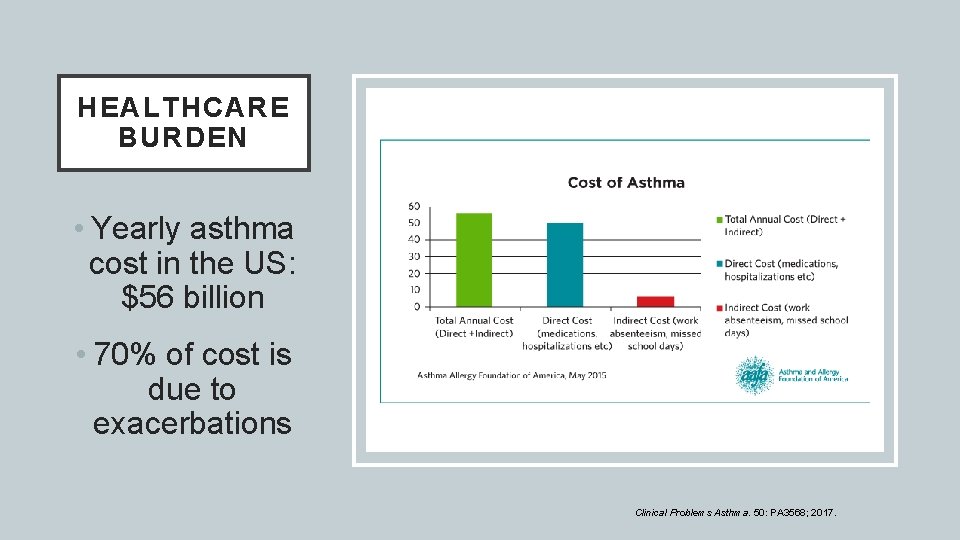
HEALTHCARE BURDEN • Yearly asthma cost in the US: $56 billion • 70% of cost is due to exacerbations Clinical Problems Asthma. 50: PA 3568; 2017.
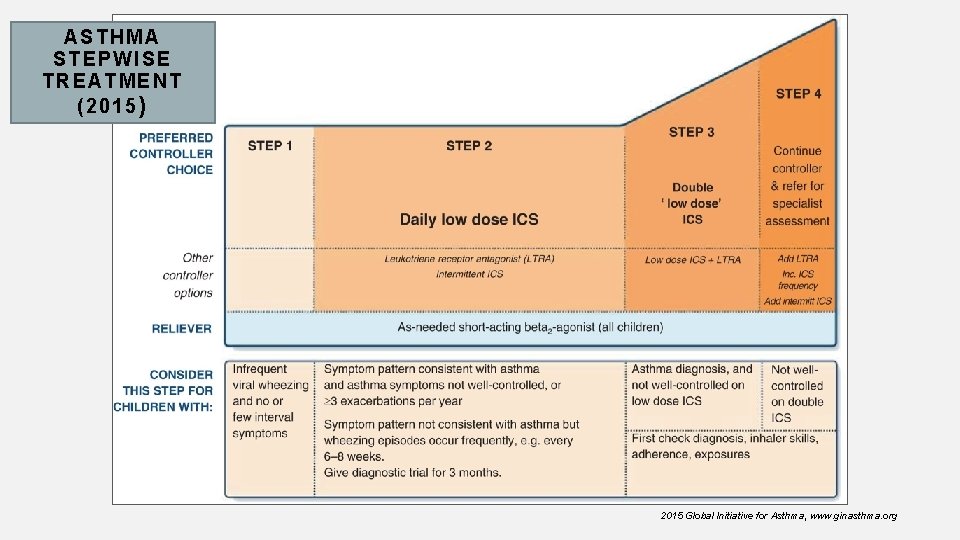
AST HMA ST EPWI SE TRE A TME NT (2015 ) 2015 Global Initiative for Asthma, www. ginasthma. org

INHALED CORTICOSTEROID CONCERNS • Systemic side effects: • Adrenal suppression • Bone metabolism • Connective Tissue Effects • Cataracts • Growth • Metabolic Effects • Psychiatric Effects Pharmaceuticals (Basel). 2010; 3(3): 514– 540.

NOVEL START TRIAL : CONTROLLED TRIAL OF BUDESONIDEFORMOTEROL AS NEEDED FOR MILD ASTHMA Beasley RD et al. NEJM. 2019; 380(21): 2020 -2030.
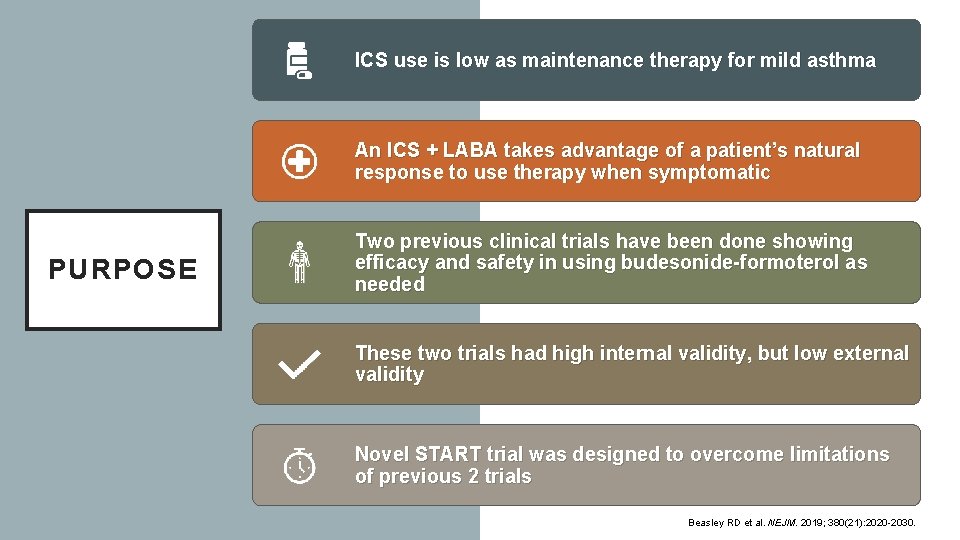
ICS use is low as maintenance therapy for mild asthma An ICS + LABA takes advantage of a patient’s natural response to use therapy when symptomatic PURPOSE Two previous clinical trials have been done showing efficacy and safety in using budesonide-formoterol as needed These two trials had high internal validity, but low external validity Novel START trial was designed to overcome limitations of previous 2 trials Beasley RD et al. NEJM. 2019; 380(21): 2020 -2030.

PREVIOUS TRIALS SYGMA-1 SYGMA-2 • Conclusion: inhaled budesonideformoterol when used as needed was superior to terbutaline, but was inferior to budesonide • Conclusion: inhaled budesonideformoterol was noninferior to the budesonide maintenance therapy for severe exacerbation • Bateman E et al. NEJM. 2018; 378(20): 1877 -1887.
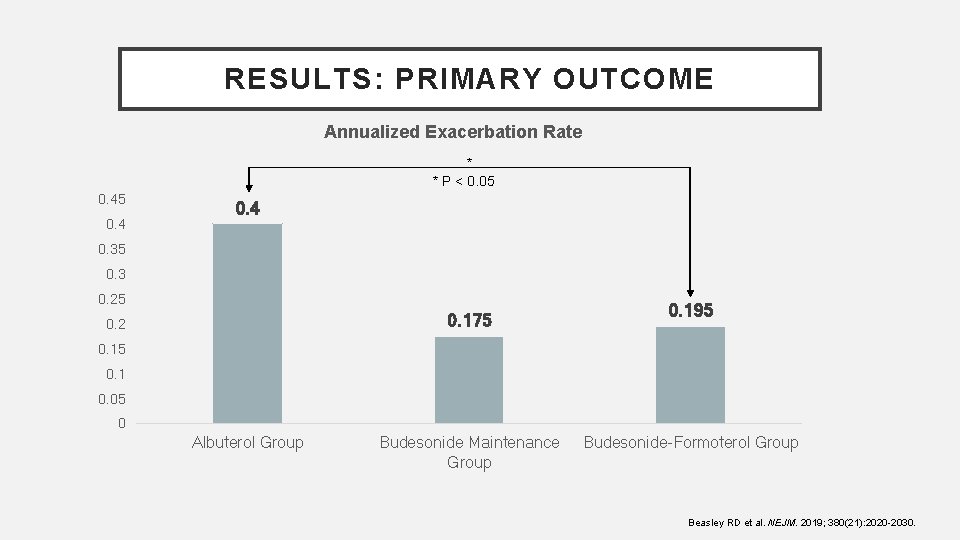
RESULTS: PRIMARY OUTCOME Annualized Exacerbation Rate * * P < 0. 05 0. 4 0. 35 0. 3 0. 25 0. 175 0. 2 0. 195 0. 1 0. 05 0 Albuterol Group Budesonide Maintenance Group Budesonide-Formoterol Group Beasley RD et al. NEJM. 2019; 380(21): 2020 -2030.

AUTHOR’S CONCLUSIONS • Among patients with mild asthma, the risk of asthma exacerbations was lower with budesonide-formoterol used as needed than with albuterol used as needed Beasley RD et al. NEJM. 2019; 380(21): 20202030.

FURTHER DISCUSSION

CURRENT BUDESONIDEFORMOTEROL FORMULATION HFA inhaler available in the US Label: NOT for use during acute bronchospasm Asthma approved dose: 80 mcg/4. 5 mcg or 160 mcg/4. 5 mcg Formoterol onset of action: 15 minutes Albuterol onset of action: 25 minutes Budesonide- Formoterol. Lexi-Drugs. Lexicomp

BUDESONIDE-FORMOTEROL BUDESONIDE 80 MCG & FORMOTEROL 4. 5 MCG BUDESONIDE 160 MCG & FORMOTEROL 4. 5 MCG Dose = 2 inhalations twice daily
MOVING FORWARD FDA approval for budesonideformoterol Turbuhaler Changes in labeling of budesonideformoterol Further trials with variety of doses and long-term effects Guideline update
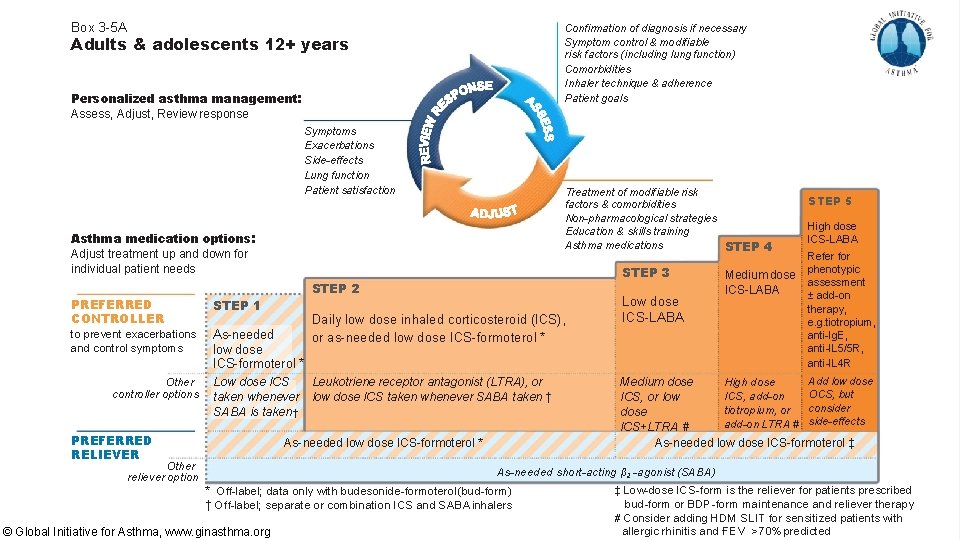
Box 3 -5 A Confirmation of diagnosis if necessary Symptom control & modifiable risk factors (including lung function) Comorbidities Inhaler technique & adherence Patient goals Adults & adolescents 12+ years Personalized asthma management: Assess, Adjust, Review response Symptoms Exacerbations Side-effects Lung function Patient satisfaction Treatment of modifiable risk factors & comorbidities Non-pharmacological strategies Education & skills training Asthma medications STEP 4 Asthma medication options: Adjust treatment up and down for individual patient needs PREFERRED CONTROLLER STEP 3 STEP 2 STEP 1 Daily low dose inhaled corticosteroid (ICS), or as-needed low dose ICS-formoterol * to prevent exacerbations As-needed and control symptoms low dose Low dose ICS-LABA ICS-formoterol * Other controller options Low dose ICS taken whenever SABA is taken † PREFERRED RELIEVER Leukotriene receptor antagonist (LTRA), or low dose ICS taken whenever SABA taken † As-needed low dose ICS-formoterol * Other reliever option STEP 5 High dose ICS-LABA Refer for phenotypic Medium dose assessment ICS-LABA ± add-on therapy, e. g. tiotropium, anti-Ig. E, anti-IL 5/5 R, anti-IL 4 R Add low dose High dose Medium dose OCS, but ICS, add-on ICS, or low consider tiotropium, or dose add-on LTRA # side-effects ICS+LTRA # As-needed low dose ICS-formoterol ‡ As-needed short-acting β 2 -agonist (SABA) * Off-label; data only with budesonide-formoterol (bud-form) † Off-label; separate or combination ICS and SABA inhalers © Global Initiative for Asthma, www. ginasthma. org ‡ Low-dose ICS-form is the reliever for patients prescribed bud-form or BDP-form maintenance and reliever therapy # Consider adding HDM SLIT for sensitized patients with 1 allergic rhinitis and FEV >70% predicted

Budesonide-formoterol as-needed lowers exacerbation rates SUMMARY 2019 GINA guidelines have been updated to include using budesonide -formoterol as-needed first line for mild asthma Budesonide-formoterol is an HFA inhaler available as 2 strengths

ASSESSMENT QUESTION #1 • Which of the following is NOT an appropriate dose of budesonideformoterol? A. One inhalation by mouth twice daily B. Two inhalations by mouth twice daily C. Two inhalations by mouth as-needed for asthma symptoms D. All of the above are appropriate

ASSESSMENT QUESTION #2 • Based on the SYGMA-1, SYGMA-2, and Novel START trial, budesonide-formoterol was superior as preventing exacerbations compared to albuterol used as needed: A. True B. False

ASSESSMENT QUESTION #3 • The 2019 GINA Guidelines were updated to include which of the following for the treatment of mild asthma? A. As-needed albuterol is no longer recommended B. As-needed low dose ICSformoterol was added as first line therapy C. Mild asthma is asthma that can be controlled with step 1 or 2 of therapy

THANK YOU Alyssa N. Romaine, BS, Pharm. D Alyssa. Romaine@va. gov
- Slides: 25