Its better to have a halflife than no

It’s better to have a half-life than no life! Radioactive Decay

What does it mean to be radioactive? Atoms that are radioactive have nuclei that spontaneously decompose to form a different nuclei and produce one or more particles These particles can be any of the following – – – Alpha particle (42 He) Beta particle (0 -1 e) Gamma particle (00γ) Positron (01 e) Cosmic rays

Radioactive (cont’d) Atoms that are radioactive have a neutron/proton ratio much greater than 1 Radioactivity can be detected by a Geiger counter

How do we measure decay? The term “half-life” is used to time radioactive decay Half-life means the time it takes for half of a sample to decay. After one half-life ½ of the nuclei have decayed to different nuclei After two half-lives ¾ of the nuclei have decayed to different nuclei 14 C = 5. 73 x 103 yr 238 U = 4. 46 x 109 yr

Nuclear Reactions? A nuclear reaction details how nuclei are changed through the emission (radioactive decay) or absorption (nuclear transformation) of alpha, beta, and gamma particles Equations are balanced by making sure the sums of the atomic numbers and mass numbers on both sides of the equation are equal

Radioactive Decay Alpha, beta, and gamma particles are emitted from radioactive nuclei Alpha particle production – EX: 222 Ra 88 42 He + 21886 Rn Beta particle production – EX: 234 Th 90 23491 Pa + 0 -1 e Gamma ray emission (usually accompanies alpha and betaparticles) – EX: 238 U 92 42 He + 23490 Th + 00γ

Nuclear Transformation Heavier elements are made from lighter ones by absorption of alpha particles, beta particles, or smaller nuclei EX: 147 N + 42 He 178 O + 11 H EX: 2713 Al + 42 He 3015 P + 10 n

Try These! Find the missing particle: – ? + 10 n 24997 Bk + 0 -1 e – 20 Ne + ? 24 Mg + 0 γ 10 12 0 Write the balanced nuclear equation for the following radioactive decays: (a) beta decay of nickel-63; (b) alpha decay of gold -185.

What use is radioactivity? Medicine – radioactive materials are used as tracers in the body Energy sources – energy can be obtained through two nuclear processes – Fission: a nucleus divides into smaller fragments – Fusion: nuclei combine to form a larger nucleus

Fission Splitting nuclei releases large quantities of energy Harnessed in nuclear power plants Reaction is controlled by 2 methods – Neutron moderation: reduces speed of neutrons – Neutron absorption: decreases number of neutrons

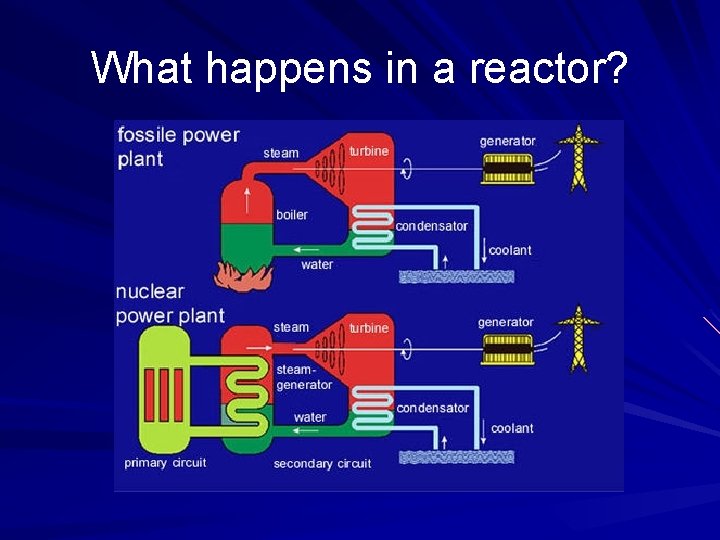
What happens in a reactor?

So what is Fusion? Fusion is much better than fission – Fuels are cheaper – Products are not radioactive Unfortunately it takes a high temperature for fusion to occur Fusion is what produces the energy for the sun.
- Slides: 13