Its a bird its a plane no its

It’s a bird, it’s a plane, no it’s a GAS!!!

Notes One Unit Five Characteristics of Gases Pressure of fluids Standard Temperature and Pressure Converting Pressures. Milwaukie High Ideal Gas Law Pages 422 -440

Importance of Gases Airbags fill with N 2 gas in an accident Gas is generated by the decomposition of sodium azide, Na. N 3 2 Na. N 3 (s) → 2 Na (s) + 3 N 2 (g)

Physical Characteristics of Gases There is a lot of “free” space in a gas Gases can be expanded infinitely Gases assume the volume and shape of their containers Gases have much lower densities than liquids and solids Gases will mix evenly and completely when confined to the same container

Properties of Gases Gas properties can be modeled using math. Model depends on: P= pressure (atmosphere) V= volume of the gas (L) T= temperature (K) n= amount, quantity of gas (moles)
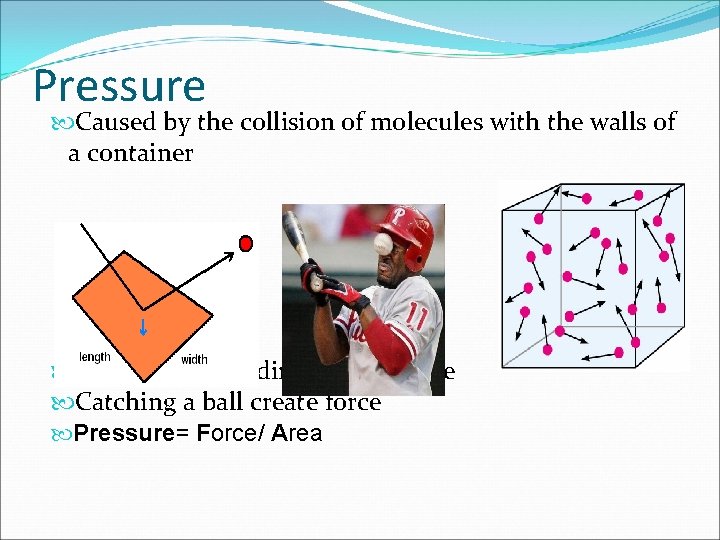
Pressure Caused by the collision of molecules with the walls of a container A molecule colliding creates force Catching a ball create force Pressure= Force/ Area

Pressure viewed as created in a fluid Created by the weight The deeper you go, the more weight
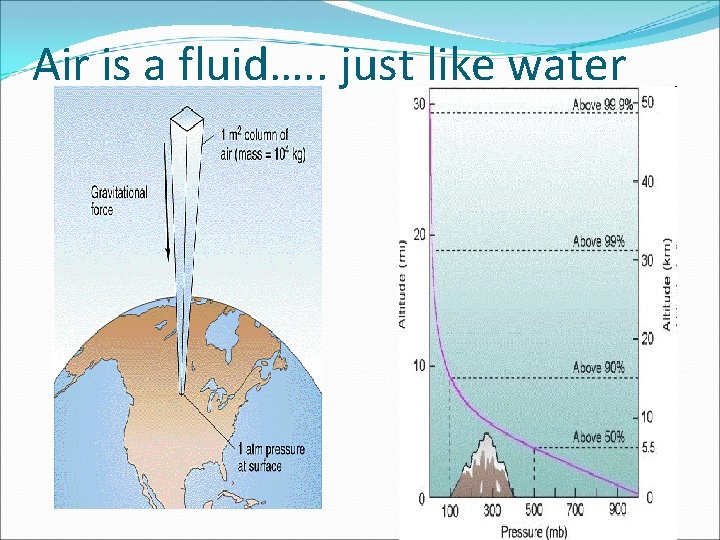
Air is a fluid…. . just like water

Measuring Pressure The first device for measuring atmospheric pressure was developed by Evangelista Torricelli during the 17 th century The device was called a “barometer” Baro = weight Meter = measure Air Pressure 760 torr Mercury

Standard Pressure, Temperature and Volume 1. 00 atm = 14. 7 psi 1. 00 atm = 29. 92 in Hg 1. 00 atm = 760 mm Hg 1. 00 atm = 760 torr 1. 00 atm = 101, 325 Pa 1. 00 atm = 101. 325 k. Pa 1. 00 atm = 1. 01325 bar Standard Temperature 273 K or 0 o. C K=o. C+273 Standard Volume: 22. 4 Liter/mole for any gas at STP pp 427

Converting Pressures Convert 25 lb/in 2 to torr Convert 75 Kpa to in Hg

Starter: Pressure Conversions The pressure of a gas is measured as 49 torr. Represent this pressure in atmospheres, Pascals, and mm. Hg.

The Ideal Gas Law PV = n. RT

Ideal Gases An “ideal” gas exhibits certain theoretical properties. Specifically, an ideal gas … Obeys all of the gas laws under all conditions. Does not condense into a liquid when cooled. Shows perfectly straight lines when its V and T & P and T relationships are plotted on a graph. In reality, there are no gases that fit this definition perfectly. We assume that gases are ideal to simplify our calculations.

THE KINETIC THEORY OF GASES Large number of particles 6. 022 x 1023 atoms/mole pp 426
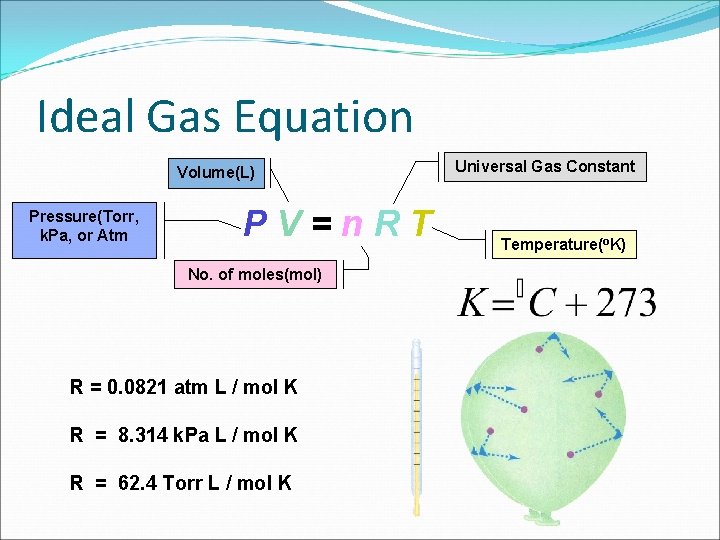
Ideal Gas Equation Volume(L) Pressure(Torr, k. Pa, or Atm PV=n. RT No. of moles(mol) R = 0. 0821 atm L / mol K R = 8. 314 k. Pa L / mol K R = 62. 4 Torr L / mol K Universal Gas Constant Temperature(o. K)

PV=n. RT Calcuation What is the volume that 2. 00 moles of iodine will occupy under the conditions: Temp = 300. o. C and Pressure = 740. Torr ? Step 1) Write down given information. n = 2. oo moles iodine T = 300. o. C = 573 K P = 740. Torr R = 62. 4 Torr. L / mol. K Step 2) Equation: PV = n. RT Step 3) Solve for variable V = Step 4) Substitute in numbers and solve 2. 00 mol x 62. 4 Torr-L/mol. K x 573. o. K V = 740. Torr = 96. 6 L n. RT P

Unit 5 Notes 2: Graham’s Law

Graham’s Law Describes how speed compares between gas molecules with different masses. Two different gases: 1)Same Temperature 2)Different Masses Kinetic energy ½ M 1 V 12= ½ M 2 V 22 pp 442

Graham’s Equation ½ M 2 V 22= ½ M 1 V 12 M 2 V 2 2 M 1 V 1 2 = M 1 M 2 V 22 V 122 V = 12 2 M 1 V 2 ÷ by M 1 ÷ by V 22 Square root of both sides pp 442

Grahams’ Law Problem One At a high temperature molecules of chlorine gas travel 15. 90 cm. What is the mass of vaporized metal (gas) under the same conditions, if the metal travels 8. 97 cm? E # Mass Cl 2 x 35. 5 = 71. 0 g/m (15. 90 cm) 8. 97 cm = M 2= 223 g/m = 14. 9 2 pp 442

Grahams’ Law Problem Two At a certain temperature molecules of chlorine gas travel at 0. 450 km/s. What is the speed of sulfur dioxide gas under the same conditions? E # Mass Cl 2 x 35. 5 = 71. 0 g/m E # Mass S 1 x 32. 1 = 32. 1 O 2 x 16. 0 = 32. 0 64. 1 g/m = V 1= 0. 474 Km/s pp 442

Unit 5 Notes #3 Gas Laws Allows us to examine the relationships between volume, temperature, pressure, and amount of gas

Boyle’s Law: P and V P α 1/V inversely proportional if moles and temperature are constant ( do not change). as one increases, the other decreases pressure is caused by moving molecules hitting container walls If V is decreased and the # of molecules stays constant, there will be more molecules hitting the walls per unit

Boyle’s Law: P and V
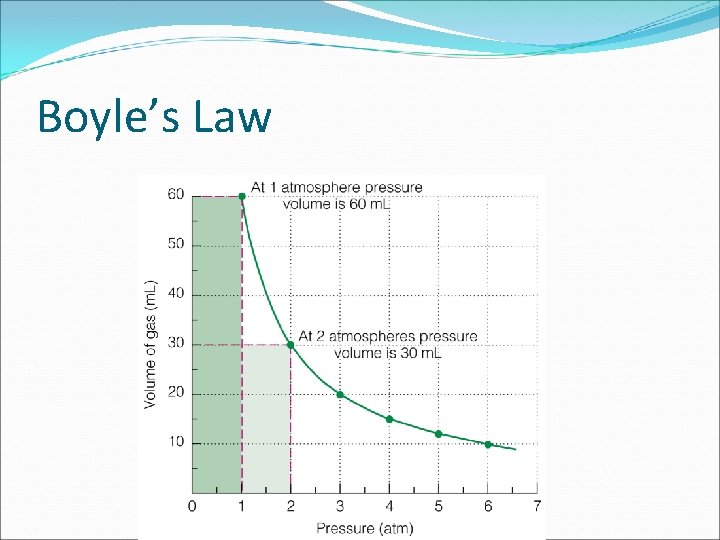
Boyle’s Law

Example: Boyle’s Law Consider a 1. 53 -L sample of gaseous SO 2 at a pressure of 5. 6 x 103 Pa. If the pressure is changed to 1. 5 x 104 Pa at constant temperature, what will be the new volume of the gas?

Charles’ Law: V and T If n and P are constant, then V α T When T increases, gas molecules move faster and collide with the walls more often and with greater force. The gas will expand To keep the P constant, the V must increase

Charles’ Law: V and T
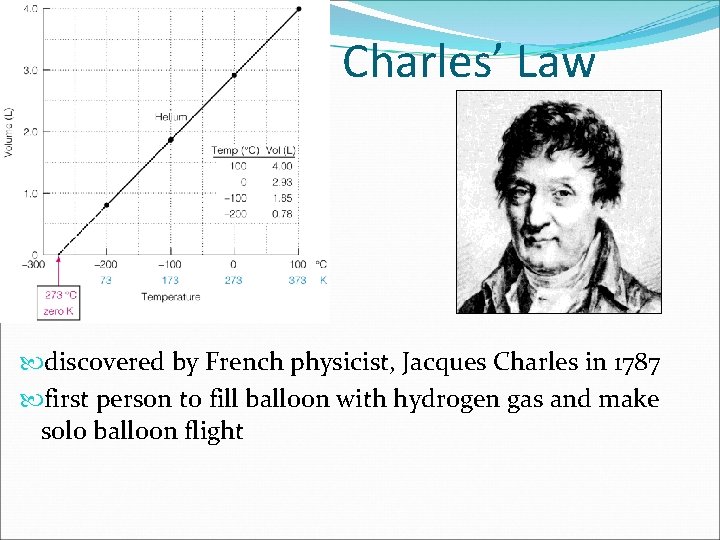
Charles’ Law discovered by French physicist, Jacques Charles in 1787 first person to fill balloon with hydrogen gas and make solo balloon flight

Charles’ Law: V and T Problem: if we use Celsius, we could end up with negative values from calculations in gas laws for volumes we need a T system with no negative values: Kelvin Temperature Scale starts at -273 ° C = absolute zero = 0 K lowest possible temperature balloon going into liquid nitrogen

Example: Charles’ Law & Temp. A sample of gas at 15°C and 1 atm has a volume of 2. 58 L. What volume will this gas occupy at 38°C and 1 atm?

T h e C o m b in e d Ga s L a w

Combining the gas laws So far we have seen two gas laws: Robert Boyle Jacques Charles V 1 V 2 = T 1 T 2 These are all subsets of a more encompassing law: the combined gas law P 1 V 1 = P 2 V 2 Joseph Louis Gay-Lussac P 1 T 1 P 2 = T 2 P 1 V 1 P 2 V 2 = T 1 T 2

Combined Gas Law Equations P 2 T 1 V 2 P 1 = T 2 V 1 T 1 P 1 T 2 V 1 = P 2 V 2 P 2 T 1 V 2 V 1 = T 2 P 1 P 1 T 2 V 1 P 2 = T 1 V 2 T 2 P 2 T 1 V 2 = P 1 V 1 P 1 T 2 V 1 V 2 = P 2 T 1

Combined Gas Law Equation Is……. .
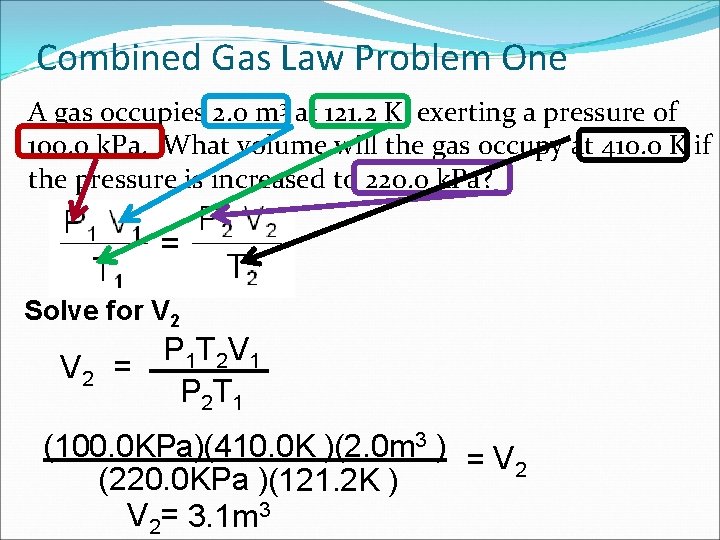
Combined Gas Law Problem One A gas occupies 2. 0 m 3 at 121. 2 K, exerting a pressure of 100. 0 k. Pa. What volume will the gas occupy at 410. 0 K if the pressure is increased to 220. 0 k. Pa? Solve for V 2 P 1 T 2 V 1 V 2 = P 2 T 1 (100. 0 KPa)(410. 0 K )(2. 0 m 3 ) = V 2 (220. 0 KPa )(121. 2 K ) V 2= 3. 1 m 3
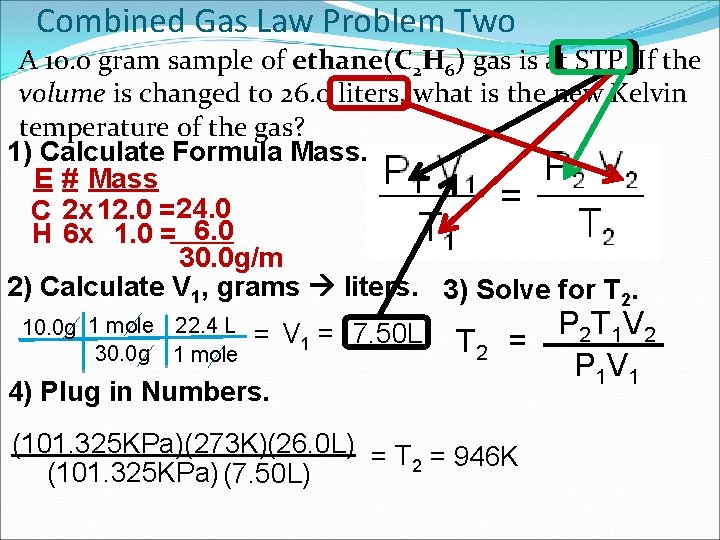
Combined Gas Law Problem Two A 10. 0 gram sample of ethane(C 2 H 6) gas is at STP. If the volume is changed to 26. 0 liters, what is the new Kelvin temperature of the gas? 1) Calculate Formula Mass. E # Mass C 2 x 12. 0 = 24. 0 H 6 x 1. 0 = 6. 0 30. 0 g/m 2) Calculate V 1, grams liters. 3) Solve for T 2. 10. 0 g 1 mole 22. 4 L 30. 0 g 1 mole = V 1 = 7. 50 L 4) Plug in Numbers. T 2 P 2 T 1 V 2 = P 1 V 1 (101. 325 KPa)(273 K)(26. 0 L) = T = 946 K 2 (101. 325 KPa) (7. 50 L)

Unit Five Notes 4 Review Mass-Mass Calculation Mass-Volume Calculation @STP Volume-Mass Calculation @STP Pages 441 -450

Let’s Put All The Steps Together! (Review of Stoich!) Factor label Method: it uses UNITS to cancel and work your way through the problem Given Info (g) 1 Mole Given (mol) MM Given (g) Given Moles Requested Moles Given Mole to Mole MM Requested (g) of 1 Mole Requested = Requested The given converts grams of given to moles of given. The middle converts moles of given to moles of requested. The requested converts moles of requested to grams of requested. All in one multistep process !!! THIS IS A MASS PROBLEM. Requested (g)

Mass to Volume Problem Info (g) 1 Mole Given (mol) MM Given (g) Given Moles Requested Moles Given 22. 4 l of Requested = Requested (l) 1 Mole Requested Mole to Mole Requested Volume to Volume Problem Info (l) 1 Mole Given (mol) 22. 4 l Given Moles Requested Moles Given Mole to Mole 22. 4 l of Requested = Requested (l) 1 Mole Requested

Mass-Mass Calculation #1 How many grams of carbon dioxide will be made by burning 6. 00 grams of Isovanillin? 1 C 8 H 8(l)+ 15 O 2(g) 8 CO 2(g) + 4 H 2 O(g) 6. 00 g C 8 H 8 1 mol C 8 H 8 8 mol CO 2 104. 0 g C 8 H 8 1 mol C 8 H 8 44. 0 g CO 2 1 mol CO 2 = 20. 3 g CO 2

Mass-Mass Calculation #2 How many grams of silver will be made by decomposing 8. 00 grams of Silver oxide? 2 Ag 2 O(s) 1 O 2(g)+ 4 Ag(s) 4 mol Ag 8. 00 g Ag. O 1 mol Ag. O 231. 8 g Ag. O 2 mol Ag. O 107. 9 g Ag 1 mol Ag = 7. 45 g Ag

Molar Volume @STP 1 mol. N 2(g) = 28. 0 g/mol 22. 4 L/mol 1 mol. O 2(g) = 32. 0 g/mol 22. 4 L/mol 1 mol. Ar(g)= 39. 9 g/mol 22. 4 L/mol

Mass-Volume Calculation How many liters of ammonia will be made reacting 56. 0 grams of nitrogen with hydrogen @ STP? 1 N 2(g) 56. 0 g N 2 + 3 H 2(g) 2 NH 3(l) 1 mol N 2 2 mol NH 3 22. 4 l NH 3 28. 0 g N 2 1 mol NH 3 = 89. 6 L NH 3

Volume-Volume Calculation How many liters of ammonia will be made reacting 33. 6 liters of nitrogen with hydrogen @STP? 1 N 2(g) + 3 H 2(g) 2 NH 3(l) 33. 6 l N 2 1 mol N 2 2 mol NH 3 22. 4 l N 2 1 mol NH 3 = 67. 2 L NH 3

Notes Four Unit Five Molar volume @ Non-STP Conditions R is Universal Gas Constant Pages 452 -459

Gas Stoich Probs NON-STP How many liters of hydrogen gas at 650. torr and 20. C will be formed by reacting 13. 0 g of zinc with excess HCl? 1 Zn + 2 HCl 1 Zn. Cl 2 + 1 H 2 Use: PV = n. RT to determine the volume of one mole of any gas will occupy at this temperature and Pressure. V = n. RT = (1. 00 mol)(62. 4 L-torr/mol. K)(293 K) = 28. 1 L P 650. Torr 13. 0 g Zn 1 mol H 2 28. 1 L H 2 65. 4 g Zn 1 mol H 2 = 5. 59 L H 2

Gas Stoich Probs NON-STP What volume of hydrogen gas at 1000. torr and 500. C would be needed to form 112 g iron? 1 Fe 2 O 3 + 3 H 2 2 Fe + 3 H 2 O Use: PV = n. RT to determine the volume of one mole of any gas will occupy at this temperature and Pressure. V = n. RT = (1. 00 mol)(62. 4 L-torr/mol. K)(773 K) = 48. 2 L P 1000. Torr 112 g Fe 1 mol Fe 3 mol H 2 48. 2 L H 2 55. 8 g Fe 2 mol Fe 1 mol H 2 = 145 L H 2

Gas Stoich Probs NON-STP What volume of oxygen gas at 1. 25 atm and 18 C would be formed by heating 10. 0 g of potassium chlorate? 2 KCl. O 3 2 KCl + 3 O 2 Use: PV = n. RT to determine the volume of one mole of any gas will occupy at this temperature and Pressure. V = n. RT = (1. 00 mol)(0. 0821 L-atm/mol. K)(291 K) = 19. 1 L P 1. 25. Atm 10. 0 g KCl. O 3 1 mol KCl. O 3 3 mol O 2 19. 1 L O 2 122. 6 g KCl. O 3 2 mol KCl. O 3 1 mol O 2 = 2. 34 L O 2

Gas Stoich Probs NON-STP What volume of methane gas (CH 4) at 98. 5 k. Pa and 24 C would be needed to form 9. 58 L of methyl bromide (CH 3 Br)? 1 CH 4 + 1 Br 2 1 CH 3 Br + 1 HBr Use: PV = n. RT to determine the volume of one mole of any gas will occupy at this temperature and Pressure. V = n. RT = (1. 00 mol)(8. 314 L-kpa/mol. K)(297 K) = 25. 1 L P 98. 5 kpa 9. 58 LCH 3 Br 1 mol CH 4 25. 1 L CH 3 Br 1 mol CH 4 = 9. 58 L CH 4

Gas Stoich Probs NON-STP What volume of hydrogen gas at. 857 atm and 23 C would be formed by reacting 2. 76 g of aluminum metal? 2 Al + 3 H 2 SO 4 1 Al 2(SO 4)3 + 3 H 2 Use: PV = n. RT to determine the volume of one mole of any gas will occupy at this temperature and Pressure. V = n. RT = (1. 00 mol)(0. 0821 L-atm/mol. K)(300. K) = 28. 7 L P. 857 Atm 2. 76 g Al 1 mol Al 3 mol H 2 28. 7 L H 2 27. 0 g Al 2 mol Al 1 mol H 2 = 4. 40 L H 2
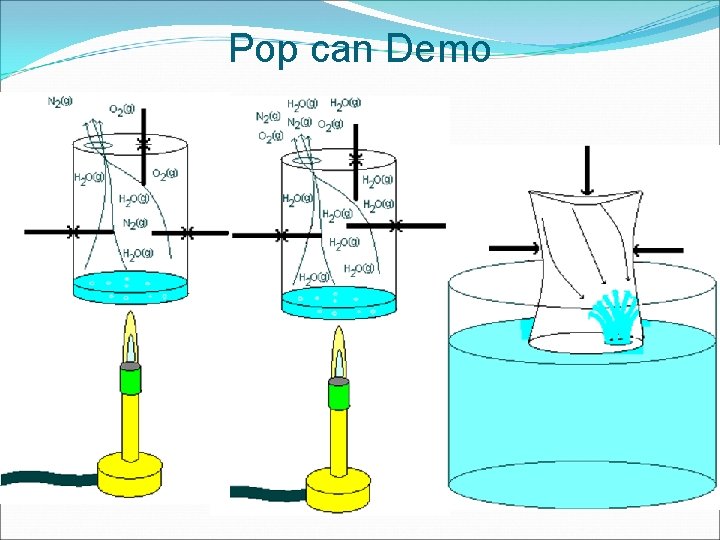
Pop can Demo

Molar Volume Lab Data Molar Volume Lab Calculations
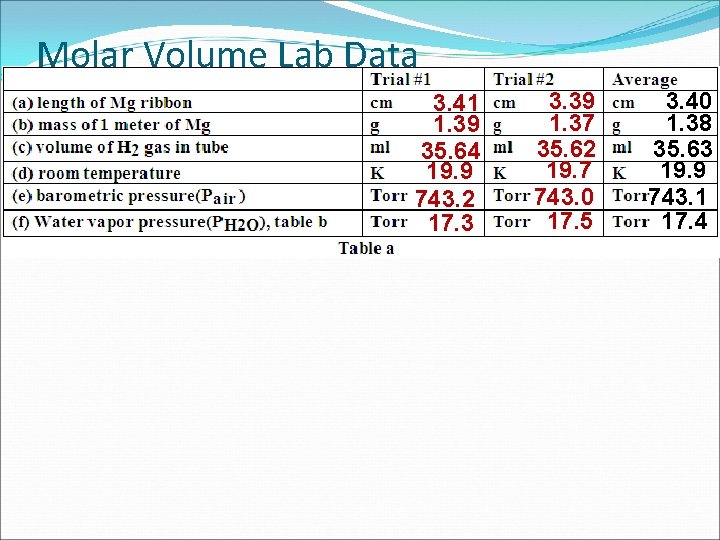
Molar Volume Lab Data 3. 41 1. 39 35. 64 19. 9 743. 2 17. 3 3. 39 1. 37 35. 62 19. 7 743. 0 17. 5 3. 40 1. 38 35. 63 19. 9 743. 1 17. 4

Molar Volume Lab Calculations 1. Calculate the grams of magnesium reacted. (1. 38 g Mg) = 0. 0469 g 3. 40 cm X (100. 00 cm) 2. Calculate the moles of magnesium reacted. 0. 0469 g ÷ 24. 3 g/mol = 0. 00193 moles 3. Calculate the pressure of the dry hydrogen gas. PH 2 = Pair- PH 2 O PH 2 = 743. 1 - 17. 4 = (725. 7 T) 4. Calculate the volume (V 2) at STP from data. (760 T) V 2 P 1 V 1 P 2 V 2 (725. 7 T)(35. 63 m. L) = = o T 1 T 2 (19. 9 C+273. 15) (273. 15 K) V 2= 31. 70 m. L 5. What is the molar volume of hydrogen at STP. Volume of H 2 at STP 31. 70 m. L = 0. 00193 m =16400 m. L/m Moles of H

Mass-Volume Calculation @STP How many liters of CO 2 will form when 6. 00 g of E # Mass Al 2(CO 3)3 decomposes? 54. 0 Al 2 x 27. 0 = ____L 1. 72 C 3 x 12. 0 = 36. 00 g ÷ 234. 0 g/m O 9 x 16. 0 =144. 0 1 Al 2(CO 3)3(s) 1 Al 2 O 3(s)+ 3 CO 2(g) 234. 0 g/m ______m 0. 0256 ______m 0. 0769 X 22. 4 g/m 1) grams Al 2(CO 3)3 to moles Al 2(CO 3)3 6. 00 g÷ 234. 0 g/m= 0. 0256 m 2) moles Al 2(CO 3)3 to moles CO 2 0. 0256 mx (3 m CO 2) =0. 0769 m 3) moles CO 2 to(1 liters CO 2 Al 2(CO 3)3 ) 0. 0769 m. CO 2 x 22. 4 L/m = 1. 72 LCO 2 pp 449

Combined gas Law Quiz Example One A gas occupies 4. 0 m 3 at 135. 1 K, exerting a pressure of 101. 3 k. Pa. What volume will the gas occupy at 390. 0 K if the pressure is increased to 150. 0 k. Pa? Assign variables and calculate V 2. (101. 3 KPa)(4. 0 m 3 ) = (150. 0 KPa )(V 2 ) (135. 1 K ) (390. 0 K ) (101. 3 KPa)(4. 0 m 3 )(390. 0 K ) = V 2 (135. 1 K )(150. 0 KPa ) V 2= 7. 8 M 3
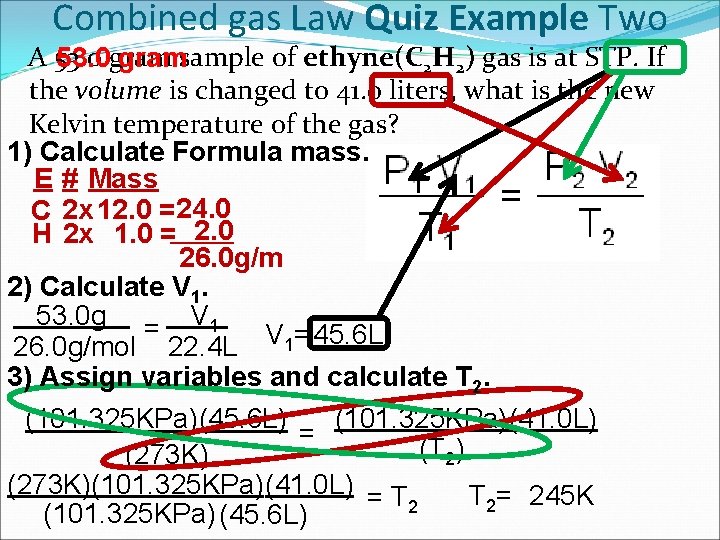
Combined gas Law Quiz Example Two A 53. 0 gramsample of ethyne(C 2 H 2) gas is at STP. If the volume is changed to 41. 0 liters, what is the new Kelvin temperature of the gas? 1) Calculate Formula mass. E # Mass C 2 x 12. 0 = 24. 0 H 2 x 1. 0 = 2. 0 26. 0 g/m 2) Calculate V 1. 53. 0 g = V 1 26. 0 g/mol 22. 4 L V 1=45. 6 L 3) Assign variables and calculate T 2. (101. 325 KPa)(41. 0 L) (101. 325 KPa)(45. 6 L) = (T 2) (273 K)(101. 325 KPa)(41. 0 L) = T T 2= 245 K 2 (101. 325 KPa) (45. 6 L)

Mass-Volume Calculation @STP How many liters of ozone will react with 4. 00 grams of nitrogen to make dinitrogen pentoxide? 5. 33 4. 00 g ÷ 28. 0 g/m ____L 3 N 2(g) _____m 0. 143 + 5 O 3(g) 3 N 2 O 5(l) pp 449 0. 238 ____m X 22. 4 g/m 1) grams N 2 to moles N 2 4. 00 g÷ 28. 0 g. N 2 /m= 0. 143 m N 2 2) moles N 2 to moles O 3 E # Mass 0. 143 m N 2 x N 2 x 14. 0 =28. 0 g/m (5 m. O 3) 3) moles O 3 to liters O 3 =0. 238 m. O 3 (3 m. N 2) E # Mass 0. 238 m. O 3 x 22. 4 L/m = 5. 33 L O 3 x 16. 0 = 48. 0 g/m

Volume-Mass Calculation @STP How many grams of ozone will react with 56. 0 liters of nitrogen to make dinitrogen pentoxide? 200. 56. 0 L ÷ 22. 4 L/m ____g 3 N 2(g) ____m 2. 50 + 5 O 3(g) 3 N 2 O 5(l) pp 449 ____m 4. 17 X 48. 0 g/m 1) Liters N 2 to moles N 2 56. 0 L÷ 22. 4 LN 2 /m= 2. 50 m N 2 2) moles N 2 to moles O 3 E # Mass 2. 50 m N 2 x N 2 x 14. 0 =28. 0 g/m (5 m. O 3) =4. 17 m. O 3) moles O 3 to grams O 3 (3 m. N 2) 3 E # Mass 4. 17 m. O 3 x 48. 0 g/m = 200. g O 3 x 16. 0 = 48. 0 g/m

Grahams’ Law Problem Three If a sulfur hexafluoride molecule has a speed of 128 m/s, what would helium’s speed be at the same temperature? M 2 = M 1 4. 0 g/m = 146. 1 g/m V 1 V 2 128 m/s E # Mass S 1 x 32. 1 = 32. 1 F 6 x 19. 0 = 114. 0 146. 1 g/m E # Mass He 1 x 4. 0 = 4. 0 g/m V 2 146. 1 g/m x(128 m/s) 4. 0 g/m = V 2 =770 m/s pp 442

Combined Gas Law: Problems 11 and 12 A gas occupies 7. 1 m 3 at 162. 3 K, exerting a pressure of 121. 5 k. Pa. What volume will the gas occupy at 250. 0 K if the pressure is increased to 172. 1 k. Pa? Assign variables and calculate V 2. (121. 5 KPa)(7. 1 m 3 ) = (172. 1 KPa )(V 2 ) (162. 3 K ) (250. 0 K ) (121. 5 KPa)(7. 1 m 3 )(250. 0 K ) = V 2 (162. 3 K )(172. 1 KPa ) V 2= 7. 7 M 3

Volume at Non-STP: Items 13. 1 and 13. 2 Oxygen is made decomposing 12. 13 g sodium peroxide at 129. 5 Kpa and 22. 1 o. C. b) What is the volume of gas at reaction conditions? PV=n. RT n. R T =V P -1 • K-1)(295. 3 K) (0. 0778 m)(8. 314 L • Kpa • m V= (129. 5 KPa) V= 1. 47 L

Grahams’ Law: Item 14 If oxygen molecules have a speed of 168 m/s, what would the speed of hydrogen molecules be at the same temperature? E # Mass O 2 x 16. 0 = 32. 0 g/m E # Mass H 2 x 1. 0 = 2. 0 g/m = V 2= 670 m/s pp 442
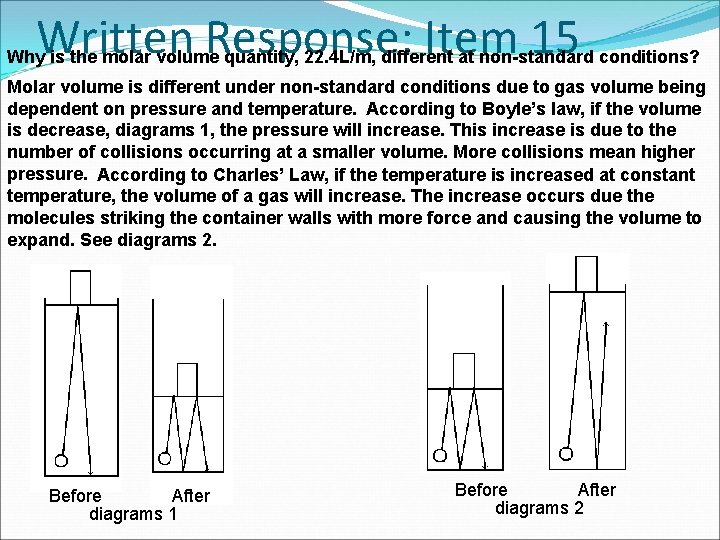
Written Response: Item 15 Why is the molar volume quantity, 22. 4 L/m, different at non-standard conditions? Molar volume is different under non-standard conditions due to gas volume being dependent on pressure and temperature. According to Boyle’s law, if the volume is decrease, diagrams 1, the pressure will increase. This increase is due to the number of collisions occurring at a smaller volume. More collisions mean higher pressure. According to Charles’ Law, if the temperature is increased at constant temperature, the volume of a gas will increase. The increase occurs due the molecules striking the container walls with more force and causing the volume to expand. See diagrams 2. Before After diagrams 1 Before After diagrams 2
- Slides: 66