ITP 210 Kimia Pangan PROTEIN Bagian Kimia Pangan

ITP 210 Kimia Pangan PROTEIN Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Materi Kuliah Pokok Bahasan Protein TIK Sub Pokok bahasan • Struktur kimia asam Mahasiswa mampu amino dan protein menjelaskan struktur • Sifat fisikokimia asam serta fungsi asam amino dan protein, • Klasifikasi protein termasuk enzim • Denaturasi protein • Sifat fungsional protein • Enzim (tatanama dan spesifisitas enzim) Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Protein • Makromolekul (polipeptida) yang tersusun dari asam amino yang dihubungkan satu sama lain dengan ikatan peptida. • Sumber: – Nabati (kedelai, kacang-kacangan, dsb) – Hewani (daging, ikan, unggas, dsb) • Merupakan molekul yang besar, mengandung lebih dari 100 residu asam amino. • Jenis: globular, fibrous, conjugated Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Protein Fungsi bagi tubuh: • Pengatur • Pembangun • Aktivitas biologis (hormon, enzim dll) 1 g/kg BB/hari Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Protein Function Enzymes Catalytic activity A B Transport Proteins Bind & carry ligand molecules (hemoglobins) Storage Proteins Ovalbumin, ferretin, casein Contractile Proteins Can contract, change shape (actin & myosins) make up elements of cytoskeleton & muscles Structural Proteins Provide support: collagen fibers of tendons, elastin of ligaments, keratin of hair & feathers, fibroin of silk & spider webs Defensive Proteins Provide protection: antibodies (Ig. G), fibrinogen, thrombin, and snake venoms Regulatory Proteins Regulate metabolic processes: includes hormones, transcription factors & enhancers Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
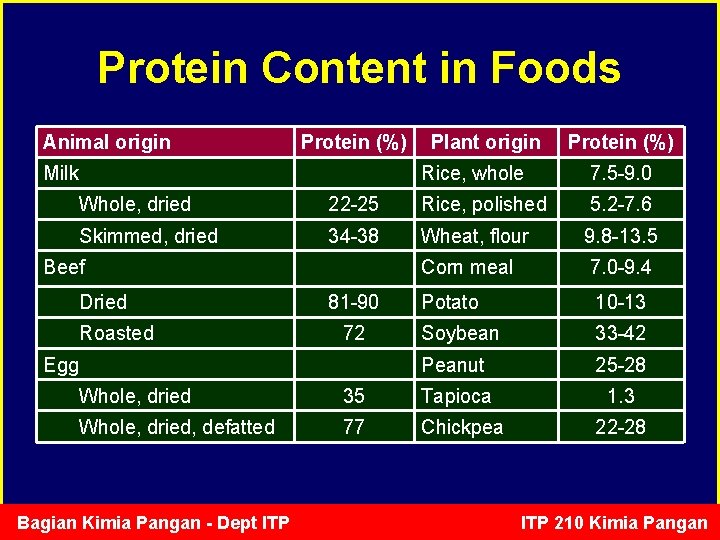
Protein Content in Foods Animal origin Protein (%) Milk Plant origin Protein (%) Rice, whole 7. 5 -9. 0 Whole, dried 22 -25 Rice, polished 5. 2 -7. 6 Skimmed, dried 34 -38 Wheat, flour 9. 8 -13. 5 Corn meal 7. 0 -9. 4 Potato 10 -13 Soybean 33 -42 Peanut 25 -28 1. 3 Beef Dried Roasted 81 -90 72 Egg Whole, dried 35 Tapioca Whole, dried, defatted 77 Chickpea Bagian Kimia Pangan - Dept ITP 22 -28 ITP 210 Kimia Pangan

Asam amino • Senyawa organik yang mengandung 2 gugus fungsional: – Amin (-NH 2): Bersifat basa – Karboksil (-COOH): Bersifat asam • Kedua gugus fungsional tersebut terikat pada karbon ( karbon) • -C bersifat asimetrik (kecuali glisin): bersifat optik aktif • Struktur dapat dinyatakan sebagai struktur ion dipolar. • Bersifat amfoterik: dapat berperilaku sebagai asam atau basa. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

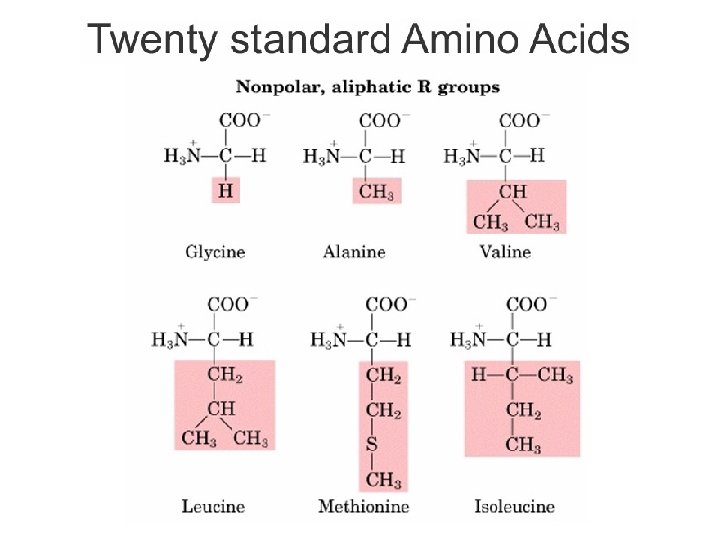
Asam Amino • Terdapat 20 jenis asam amino, yang berbeda satu sama lain pada gugus R yang terikat pada -karbon • Gugus R dapat bersifat gugus alifatik nonpolar, gugus alifatik polar, gugus aromatik, dan bermuatan positif/negatif. • Asam amino yang paling sederhana: glisin (gugus R adalah H) Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
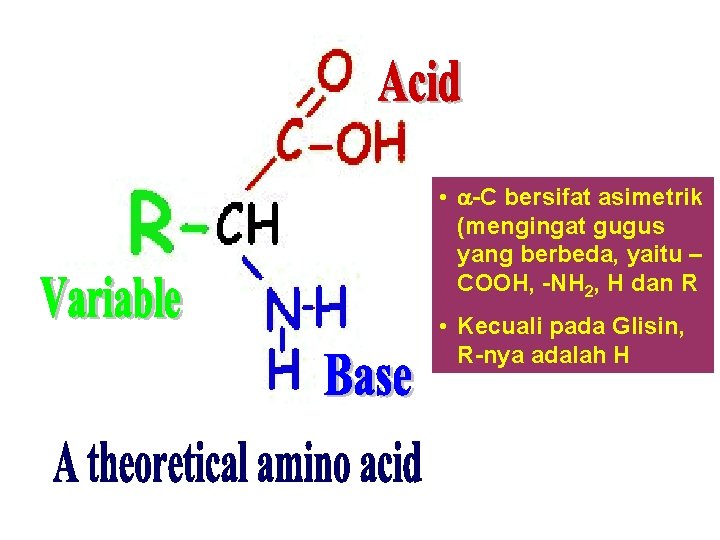
• -C bersifat asimetrik (mengingat gugus yang berbeda, yaitu – COOH, -NH 2, H dan R • Kecuali pada Glisin, R-nya adalah H Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
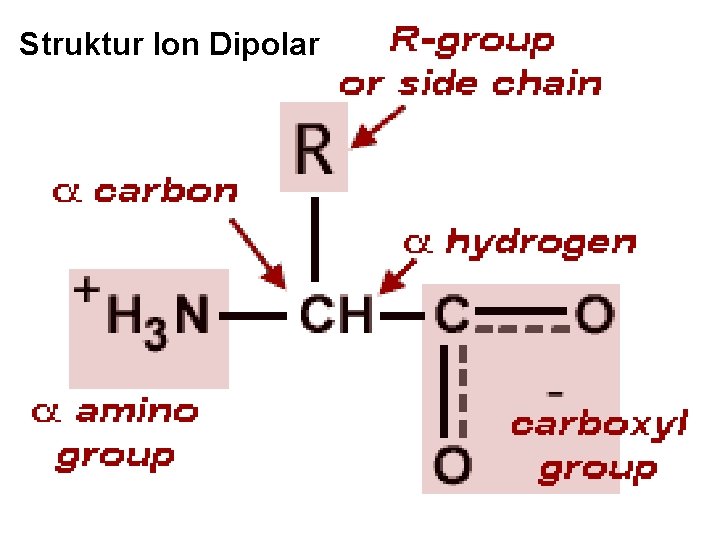
Struktur Ion Dipolar Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

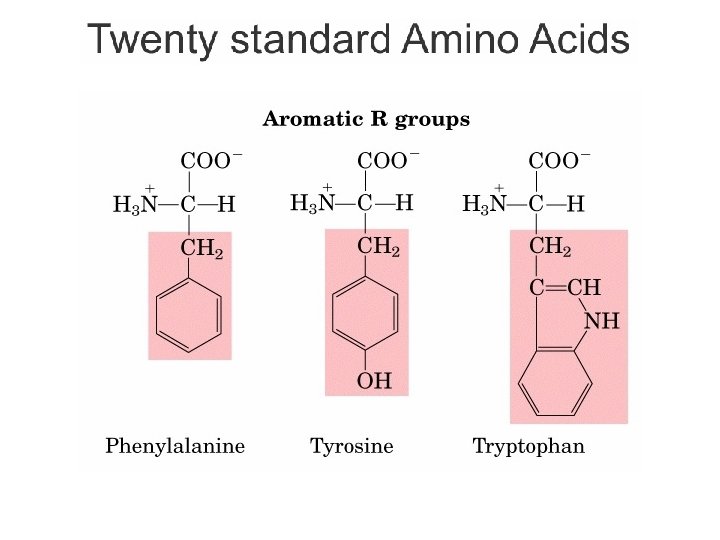
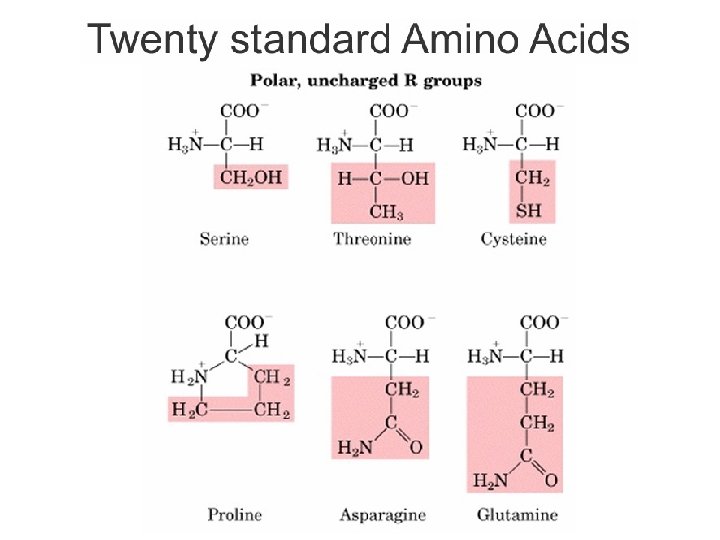
Kelompok Asam amino Berdasarkan Sifat Kepolaran • Non-polar, R gugus alifatik – Gugus R tersusun dari gugus hidrokarbon yang bersifat hidrofobik. – Glisin, alanin, valin, leusin, metionin, isoleusin • R gugus aromatik – Gugus R tersusun dari struktur cincin aromatik atau sulfur – Fenilalanin, tirosin, triptofan • Polar, gugus R tidak bermuatan – Gugus R mengandung gugus hidroksil atau gugus amino – Bersifat hidrofilik (dapat membentuk ikatan hidrogen) – Serin, treonin, sistein, prolin, asparagin, glutamin Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

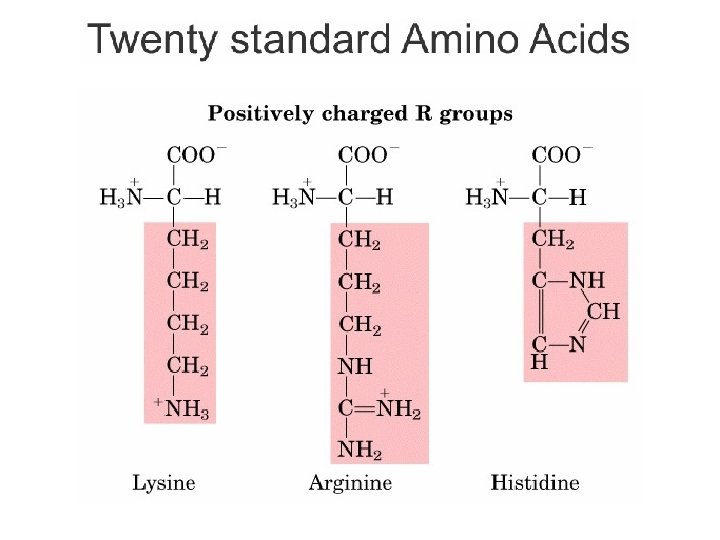
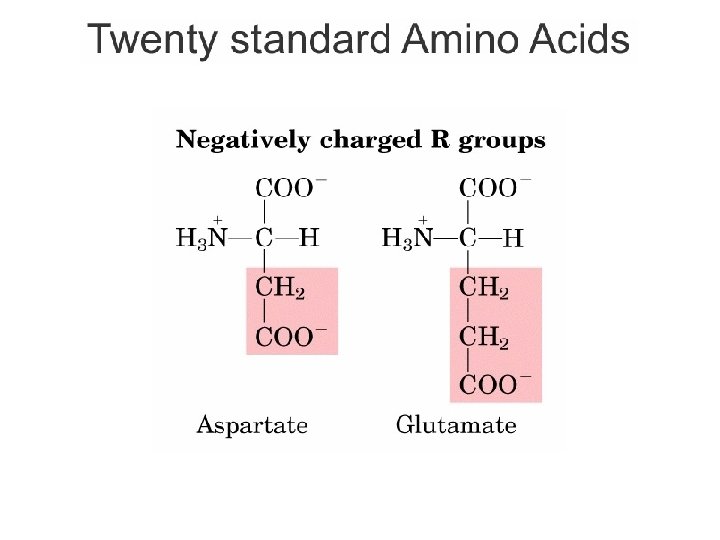
Kelompok Asam amino Berdasarkan Sifat Kepolaran • R bermuatan positif – Gugus R mempunyai gugus amide yang dapat membentuk ion positif pada p. H di bawah 7. 0 – Lisin, arginin, histidin • R bermuatan negatif – Gugus R mempunyai gugus COOH yang dapat membentuk ion negatif pada p. H di atas 7. 0 – Asam aspartat, Asam glutamat Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
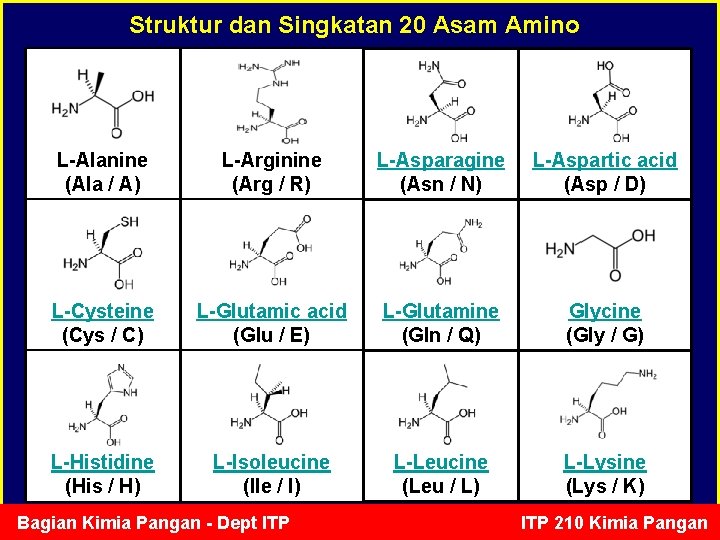
Struktur dan Singkatan 20 Asam Amino L-Alanine (Ala / A) L-Arginine (Arg / R) L-Asparagine (Asn / N) L-Aspartic acid (Asp / D) L-Cysteine (Cys / C) L-Glutamic acid (Glu / E) L-Glutamine (Gln / Q) Glycine (Gly / G) L-Histidine (His / H) L-Isoleucine (Ile / I) L-Leucine (Leu / L) L-Lysine (Lys / K) Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
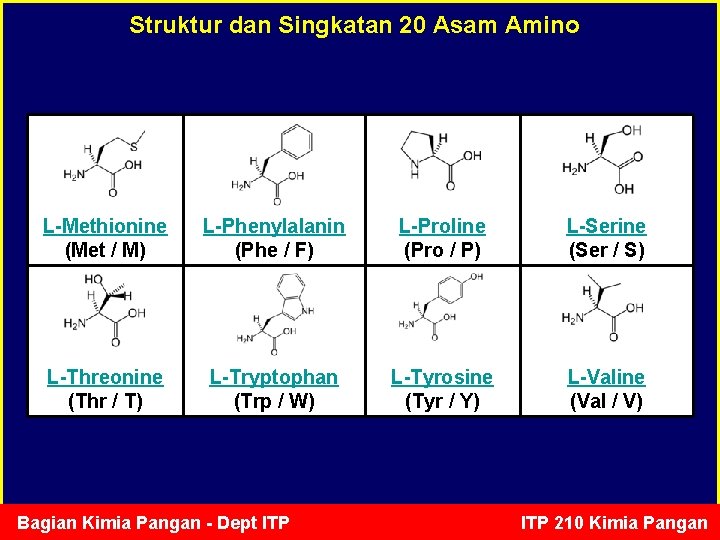
Struktur dan Singkatan 20 Asam Amino L-Methionine (Met / M) L-Phenylalanin (Phe / F) L-Proline (Pro / P) L-Serine (Ser / S) L-Threonine (Thr / T) L-Tryptophan (Trp / W) L-Tyrosine (Tyr / Y) L-Valine (Val / V) Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Sifat ionisasi Asam amino • Di dalam larutan, asam amino terionisasi dan dapat bersifat sebagai asam atau basa (bersifat amfoter). • Dalam keadaan dipolar (zwitterion), dimana gugus amin dan karboksil berionisasi, asam amino memiliki kelarutan yang minimal. • Titik isoelektrik: p. H pada saat molekul asam amino tidak bermuatan • p. K: p. H pada saat gugus amino dan karboksil 50% terionisasi dan 50% tidak terionisasi. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
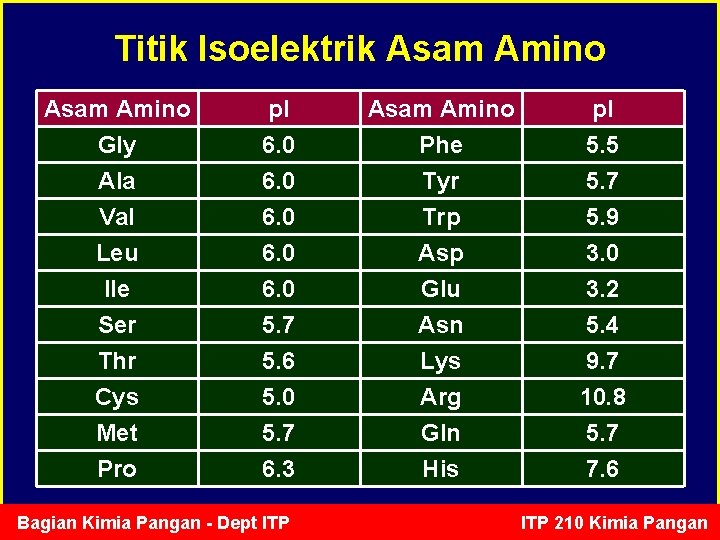
Titik Isoelektrik Asam Amino Gly Ala Val p. I 6. 0 Asam Amino Phe Tyr Trp p. I 5. 5 5. 7 5. 9 Leu Ile Ser Thr Cys Met Pro 6. 0 5. 7 5. 6 5. 0 5. 7 6. 3 Asp Glu Asn Lys Arg Gln His 3. 0 3. 2 5. 4 9. 7 10. 8 5. 7 7. 6 Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Electrical Charges • Some individual amino acid residues in a protein have the potential to be charged electrically in different ways, depending on the p. H of the medium in which the protein is found. • The individual protein molecules are said to be amphoteric, because they have the potential to function as either acid or a base, depending on the p. H. • Isoelectric point: when the number of positive and negative charges are equal. Protein has minimum solubility at the isoelectric point. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
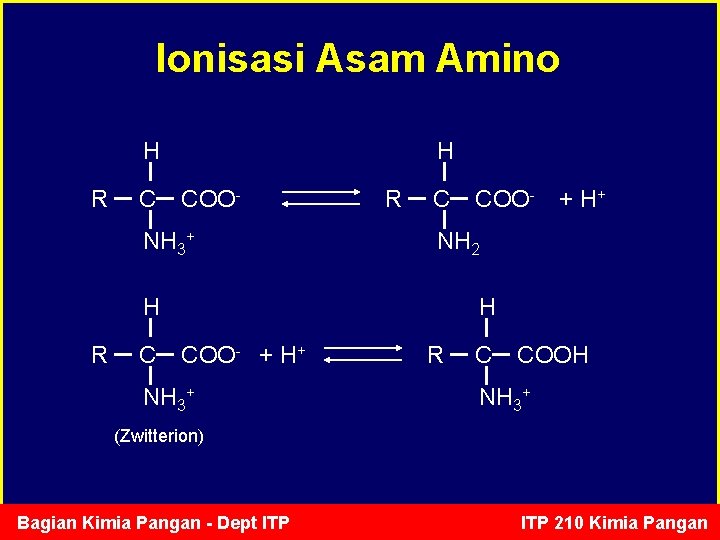
Ionisasi Asam Amino H R C H COO- NH 3+ R C C + H+ NH 2 H R COO- H COO- + H+ NH 3+ R C COOH NH 3+ (Zwitterion) Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
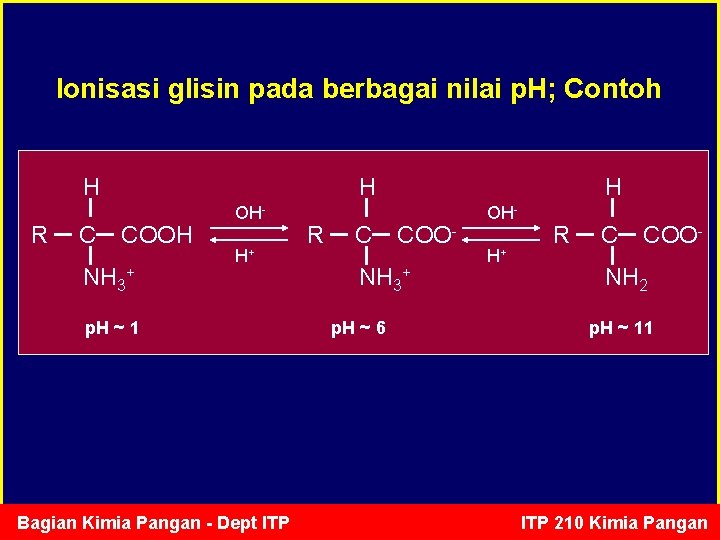
Ionisasi glisin pada berbagai nilai p. H; Contoh H R C H COOH NH 3+ OHH+ p. H ~ 1 Bagian Kimia Pangan - Dept ITP R C H COO- NH 3+ p. H ~ 6 OHH+ R C COO- NH 2 p. H ~ 11 ITP 210 Kimia Pangan
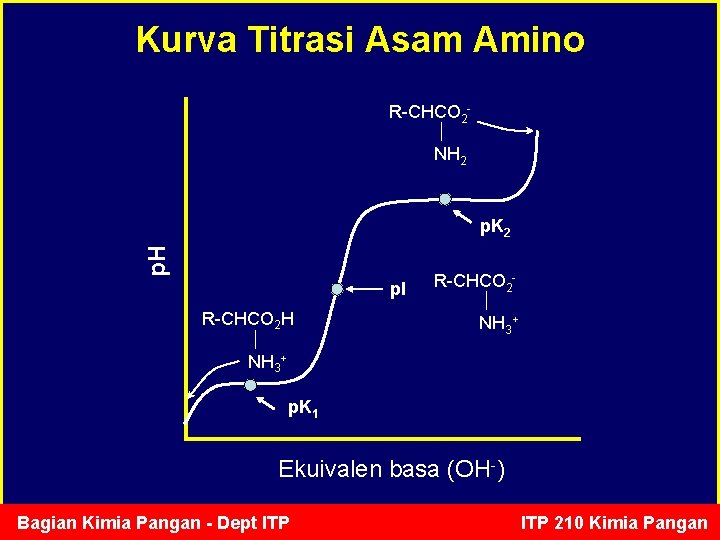
Kurva Titrasi Asam Amino R-CHCO 2 NH 2 p. H p. K 2 p. I R-CHCO 2 H R-CHCO 2 NH 3+ p. K 1 Ekuivalen basa (OH-) Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Polimerisasi • Jenis polimerisasi: – Dipeptida: 2 asam amino berikatan – Oligopeptida: – Polipeptida: – Protein: • Dihubungkan satu sama lain dengan ikatan peptida Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Ikatan peptida • Ikatan peptida merupakan ikatan kovalen yang menghubungkan antara gugus amin (-NH 2) pada AA 1 dengan gugus karboksil (-COOH) pada AA 2 • Pada saat terbentuk ikatan peptida, 1 molekul air dibebaskan (polimerisasi kondensasi) • Ikatan peptida lebih pendek dan lebih kuat daripada ikatan C-C, tetapi lebih lemah dibanding C=C. • Ikatan peptidak dapat berotasi secara bebas Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
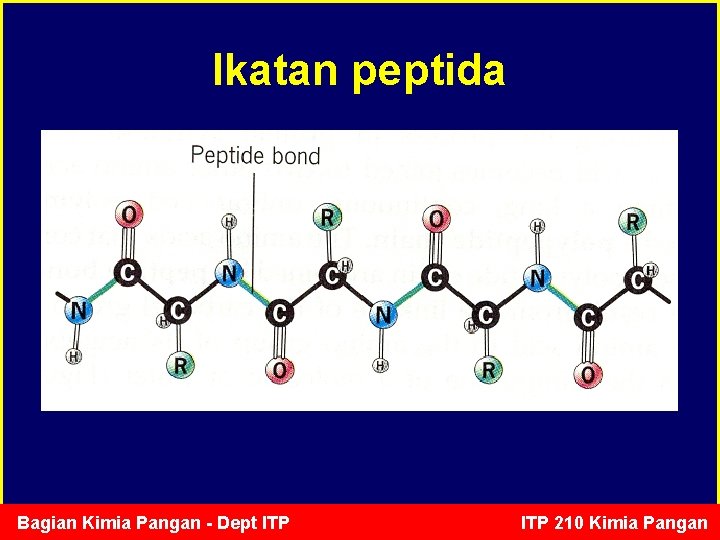
Ikatan peptida Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
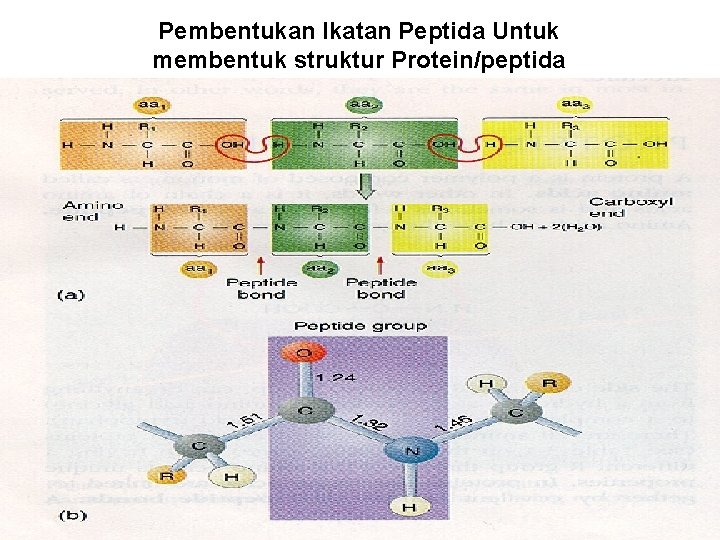
Pembentukan Ikatan Peptida Untuk membentuk struktur Protein/peptida Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
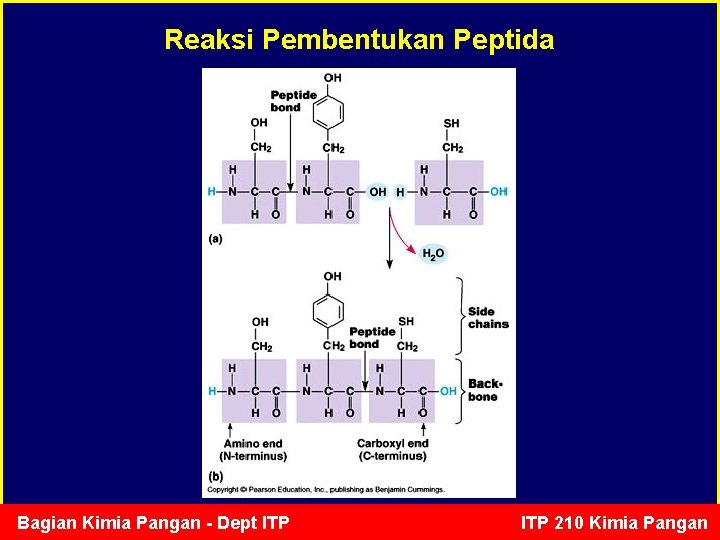
Reaksi Pembentukan Peptida Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Struktur Peptida hasil polimerisasi Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
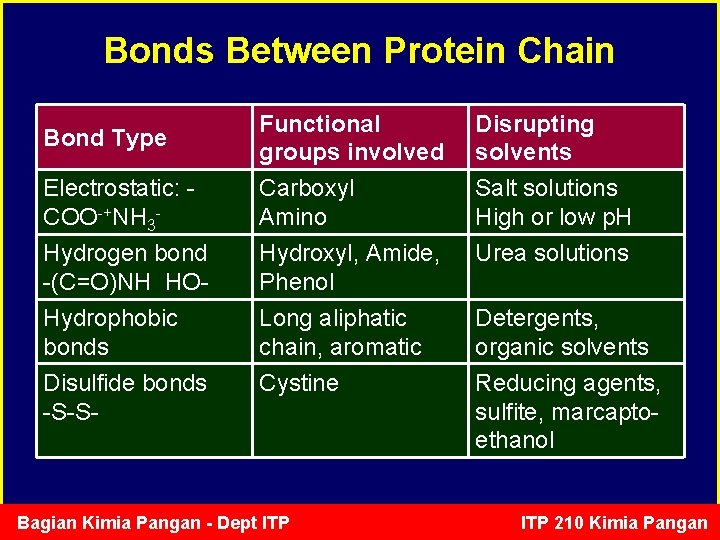
Bonds Between Protein Chain Electrostatic: COO-+NH 3 - Functional groups involved Carboxyl Amino Disrupting solvents Salt solutions High or low p. H Hydrogen bond -(C=O)NH HO- Hydroxyl, Amide, Phenol Urea solutions Hydrophobic bonds Disulfide bonds -S-S- Long aliphatic chain, aromatic Cystine Detergents, organic solvents Reducing agents, sulfite, marcaptoethanol Bond Type Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Protein Structure Primary sequence Secondary structure Tertiary Quaternary Linear sequence of AA's from N-terminal to C-terminal NCC-NCC-NCC-NCCNCC Regular, recurring orientations of AA's in a peptide chain due to H-bonds = alpha helix & beta sheets • 3 D - conformational shape due to weak electrostatic interactions with other atoms • Shape of most proteins is GLOBULAR 2 or more different polypeptides or subunits interacting to give a unique 3 D spatial relationship Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
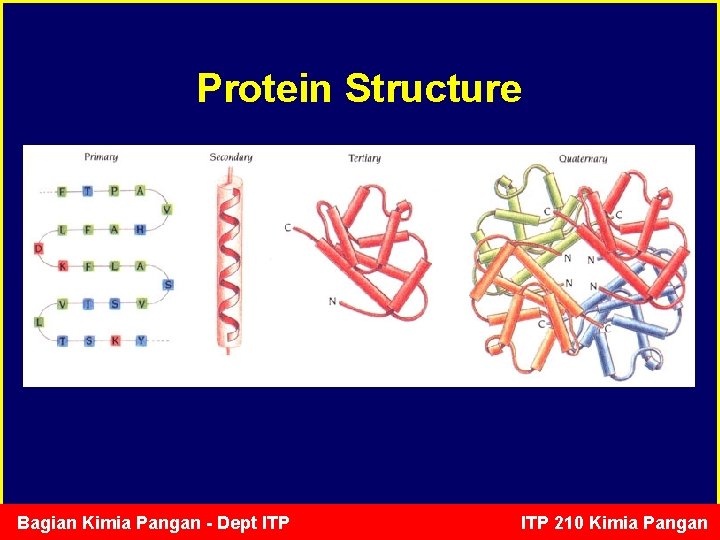
Protein Structure Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
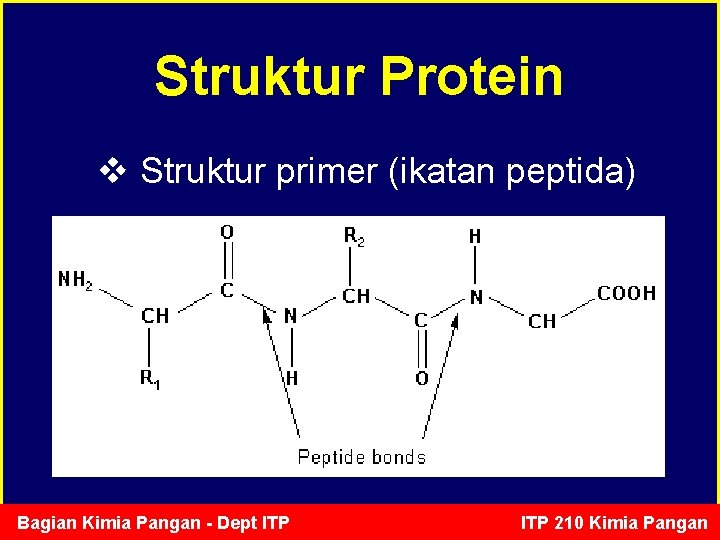
Struktur Protein v Struktur primer (ikatan peptida) Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
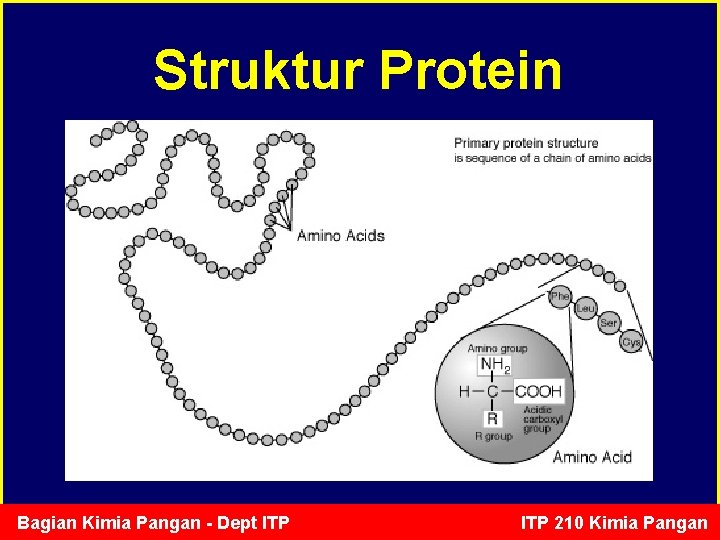
Struktur Protein Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
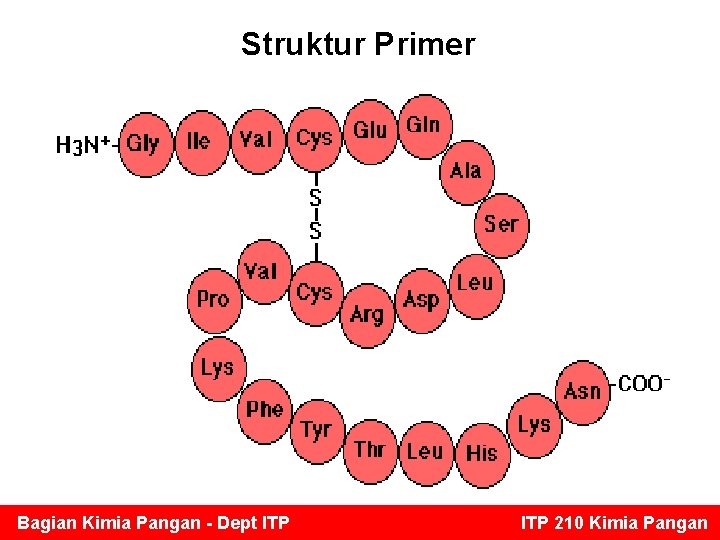
Struktur Primer Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
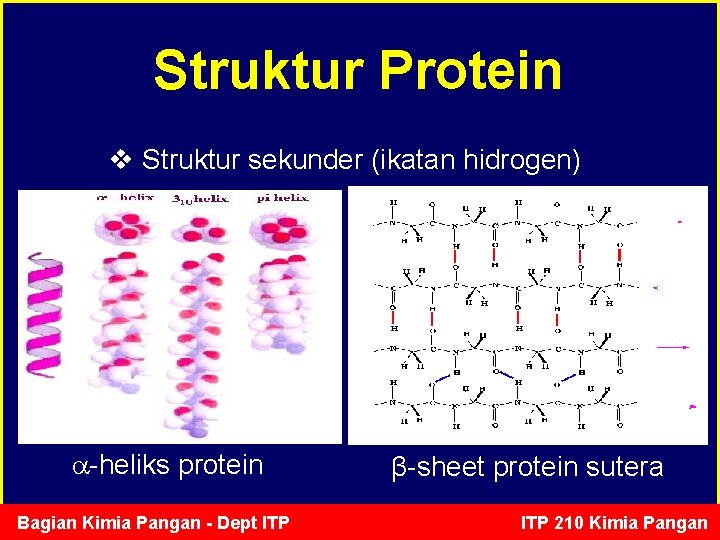
Struktur Protein v Struktur sekunder (ikatan hidrogen) -heliks protein Bagian Kimia Pangan - Dept ITP β-sheet protein sutera ITP 210 Kimia Pangan
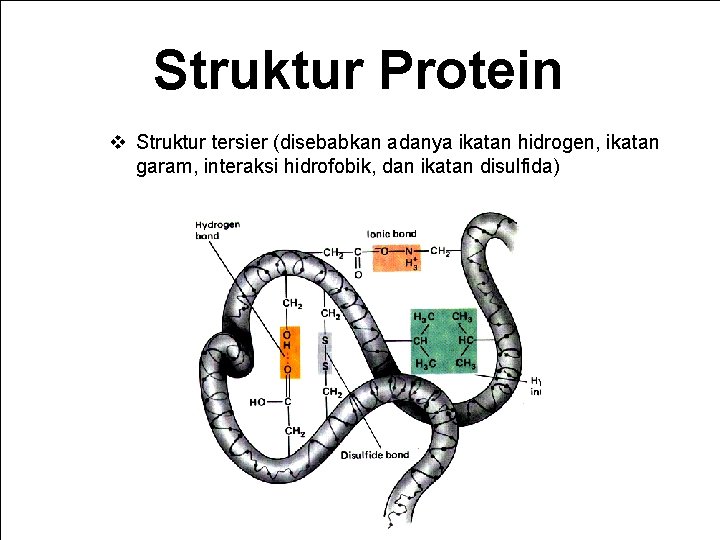
Struktur Protein v Struktur tersier (disebabkan adanya ikatan hidrogen, ikatan garam, interaksi hidrofobik, dan ikatan disulfida) Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
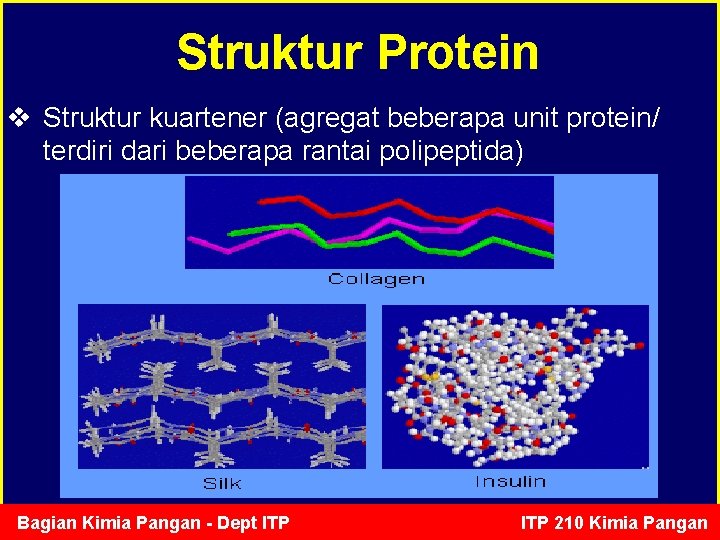
Struktur Protein v Struktur kuartener (agregat beberapa unit protein/ terdiri dari beberapa rantai polipeptida) Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
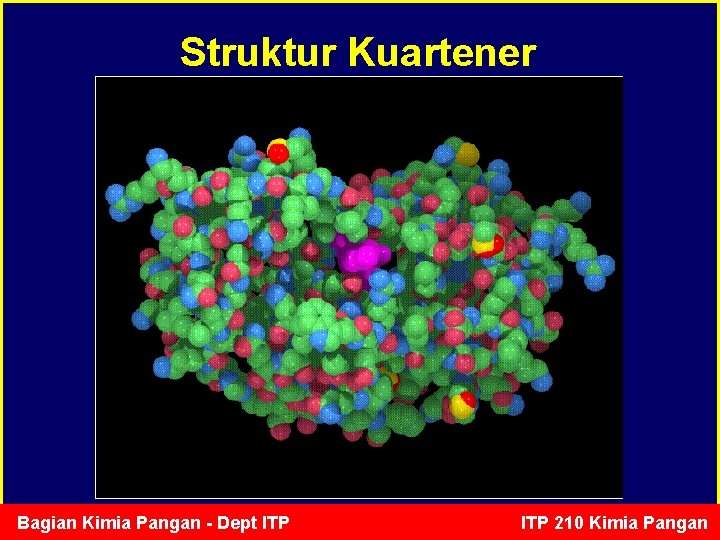
Struktur Kuartener Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Types of Protein • Globular protein: native proteins that are rather spherical in the configuration of their tertiary structure. Exp. Albumin (egg), globulin (meat, legume), histone (glandular tissue), protamines (fish sperm cells) • Fibrous protein: Insoluble, elongated protein molecules. Exp. Collagen, elastin (in meats, poultry) • Conjugated protein: proteins combined with some other type of compound, such as a carbohydrate or lipid. Exp: mucoproteins (glycoproteins), lipoprotein, metalloprotein, nucleoprotein, phosphoprotein Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
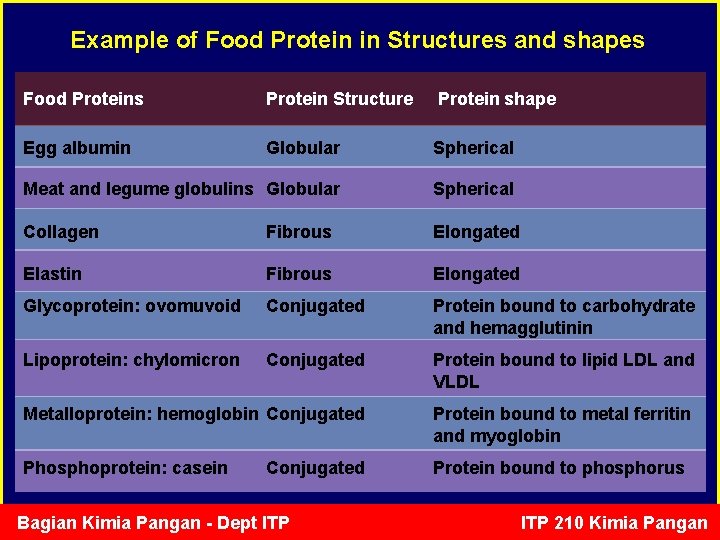
Example of Food Protein in Structures and shapes Food Proteins Protein Structure Protein shape Egg albumin Globular Spherical Meat and legume globulins Globular Spherical Collagen Fibrous Elongated Elastin Fibrous Elongated Glycoprotein: ovomuvoid Conjugated Protein bound to carbohydrate and hemagglutinin Lipoprotein: chylomicron Conjugated Protein bound to lipid LDL and VLDL Metalloprotein: hemoglobin Conjugated Protein bound to metal ferritin and myoglobin Phosphoprotein: casein Protein bound to phosphorus Conjugated Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Komponen Bahan Pangan Protein Perubahan Yang mungkin Terjadi selama Proses Pengolahan dan Penyimpanan Pangan • Denaturasi (karena panas) yang akan menyebabkan perubahan kelarutan, sehingga mempengaruhi tekstur pada bahan pangan • Penyimpangan flavor yang disebabkan karena oksidasi (dikatalisis oleh cahaya) • Degradasi enzimatik yang akan menyebabkan perubahan pada tekstur dan flavor (bisa menyebabkan terbentuknya flavor pahit) • Pembekuan dapat menyebabkan protein mengalami perubahan konformasi dan kelarutannya Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

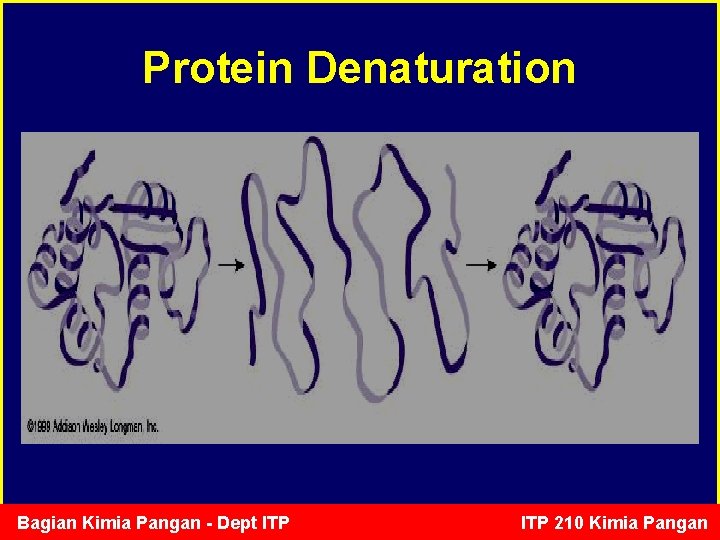
Protein Denaturation • Denaturation of proteins involves the disruption and possible destruction of both the secondary and tertiary structures. • Since denaturation reactions are not strong enough to break the peptide bonds, the primary structure (sequence of amino acids) remains the same after a denaturation process. • Denaturation disrupts the normal alpha-helix and beta sheets in a protein and uncoils it into a random shape. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Protein Denaturation • Denaturation occurs because the bonding interactions responsible for the secondary structure (hydrogen bonds to amides) and tertiary structure are disrupted. • In tertiary structure there are four types of bonding interactions between "side chains" including: hydrogen bonding, salt bridges, disulfide bonds, and non-polar hydrophobic interactions, which may be disrupted. Therefore, a variety of reagents and conditions can cause denaturation. • The most common observation in the denaturation process is the precipitation or coagulation of the protein. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Protein Denaturation • Unfolding of protein structure (due to H bonds breaking) without disrupting protein covalent bonds. • Functional properties of protein will change during denaturation (e. g. enzyme function will be stopped; solubility in water will decrease). • Example: – Thermal processing denatures the meat protein actin, myosin and myoglobin. – Cooking egg denatures egg white proteins including ovalbumin Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
Protein Denaturation Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
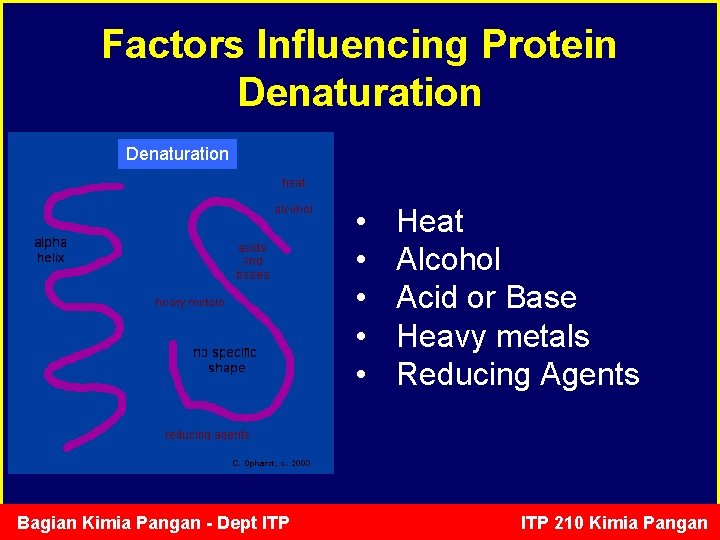
Factors Influencing Protein Denaturation • • • Bagian Kimia Pangan - Dept ITP Heat Alcohol Acid or Base Heavy metals Reducing Agents ITP 210 Kimia Pangan

Protein Denaturation • Effect of Alcohol – Hydrogen bonding occurs between amide groups in the secondary protein structure. Hydrogen bonding between "side chains" occurs in tertiary protein structure in a variety of amino acid combinations. All of these are disrupted by the addition of another alcohol. – Alcohol denatures proteins by disrupting the side chain intramolecular hydrogen bonding. New hydrogen bonds are formed instead between the new alcohol molecule and the protein side chains. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Protein Denaturation • Effect of Acids and Bases – Salt bridges result from the neutralization of an acid and amine on side chains. – The final interaction is ionic between the positive ammonium group and the negative acid group. – Any combination of the various acidic or amine amino acid side chains will have this effect. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Protein Denaturation • Effect of Acids and Bases – Acids and bases disrupt salt bridges held together by ionic charges. A type of double replacement reaction occurs where the positive and negative ions in the salt change partners with the positive and negative ions in the new acid or base added. – This reaction occurs in the digestive system, when the acidic gastric juices cause the curdling (coagulating) of milk. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Protein Denaturation • Effect of Acids and Bases – The denaturation reaction on the salt bridge by the addition of an acid results in a further straightening effect on the protein chain Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
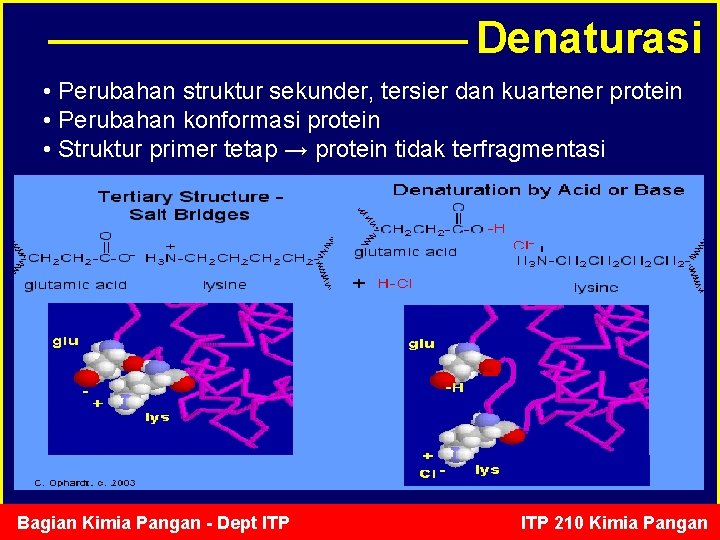
Denaturasi • Perubahan struktur sekunder, tersier dan kuartener protein • Perubahan konformasi protein • Struktur primer tetap → protein tidak terfragmentasi Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
Denaturasi Flour Process Bread Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Functional Properties of Protein • Characteristics that govern the behavior of proteins in foods during processing, storage, and preparation as they affect food quality and acceptance. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Fisiko-kimia Protein § Hidrasi (daya ikat air/water holding capacity) dan pembentuk viskositas § Kelarutan (penting untuk proses ekstraksi) § Pengental § Pembentuk gel (penting untuk produk olahan daging dan ikan) § Pembentuk tekstur § Koagulasi panas dan pembentuk film (seperti pada kembang tahu) § Texturized protein (pembentukan struktur serat seperti daging pada produk ekstrusi) Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Fisiko-kimia Protein § Pengemulsi § Pembentuk busa (busa protein putih telur melembutkan dan mengembangkan produk cake) § Pembentuk adonan (protein gluten yang bersifat viskoelastis pada adonan roti dan mie) § Bersama-sama dengan gula pereduksi membentuk warna dan aroma melalui reaksi Maillard Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
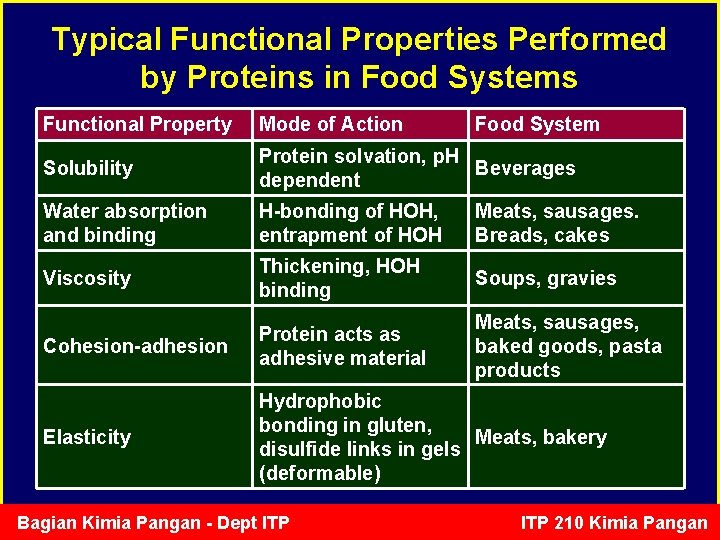
Typical Functional Properties Performed by Proteins in Food Systems Functional Property Mode of Action Solubility Protein solvation, p. H Beverages dependent Water absorption and binding H-bonding of HOH, entrapment of HOH Meats, sausages. Breads, cakes Viscosity Thickening, HOH binding Soups, gravies Cohesion-adhesion Protein acts as adhesive material Meats, sausages, baked goods, pasta products Elasticity Hydrophobic bonding in gluten, Meats, bakery disulfide links in gels (deformable) Bagian Kimia Pangan - Dept ITP Food System ITP 210 Kimia Pangan

Typical Functional Properties Performed by Proteins in Food Systems Functional Property Mode of Action Food System Gelation Protein matrix formation and setting Meats, curds, cheese Emulsification Formation and stabilization of fat emulsions Sausages, soup, cakes Fat adsorption Binding of free fat Meats, sausages, donuts Flavor binding Adsorption, entrapment, release Simulated meats, bakery, etc Foaming Forms stable film to entrap gas Whipped toppings. dessert Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Factors Influencing the Functional Properties of Food Proteins Intrinsic Environmental Factors • Composition of • Water proteins • p. H • Conformation • Temperature of proteins • Oxidizing/ • Mono- or multi reducing agents component • Lipids. Flavors, • Homogeneity – sugars heterogeneity Bagian Kimia Pangan - Dept ITP Process Treatments • Heating • p. H • Salts • Reducing/ oxidizing agents • Drying • Physical modification • Chemical modification ITP 210 Kimia Pangan
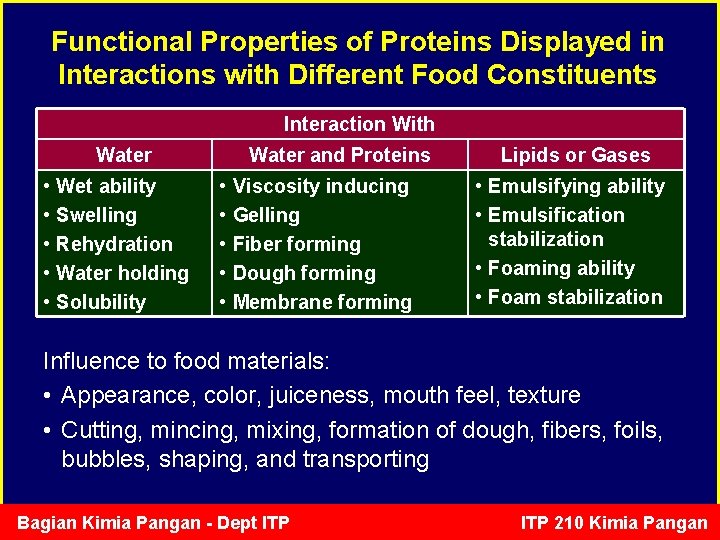
Functional Properties of Proteins Displayed in Interactions with Different Food Constituents Interaction With Water • Wet ability • Swelling • Rehydration • Water holding • Solubility Water and Proteins • Viscosity inducing • Gelling • Fiber forming • Dough forming • Membrane forming Lipids or Gases • Emulsifying ability • Emulsification stabilization • Foaming ability • Foam stabilization Influence to food materials: • Appearance, color, juiceness, mouth feel, texture • Cutting, mincing, mixing, formation of dough, fibers, foils, bubbles, shaping, and transporting Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
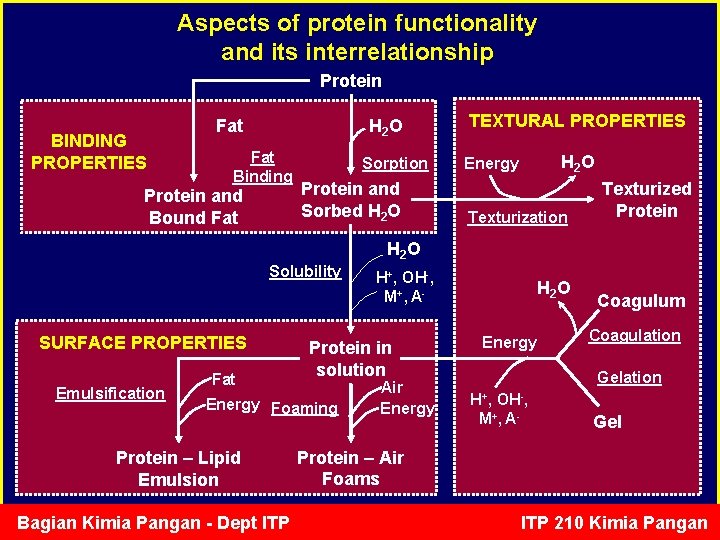
Aspects of protein functionality and its interrelationship Protein BINDING PROPERTIES Fat H 2 O Fat Binding Protein and Bound Fat Sorption Protein and Sorbed H 2 O TEXTURAL PROPERTIES H 2 O Energy Texturization Texturized Protein H 2 O Solubility SURFACE PROPERTIES Emulsification Fat Protein in solution Energy Foaming Protein – Lipid Emulsion Bagian Kimia Pangan - Dept ITP H+, OH-, M+, A- Air Energy H 2 O Energy Coagulum Coagulation Gelation H+, OH-, M+, A- Gel Protein – Air Foams ITP 210 Kimia Pangan
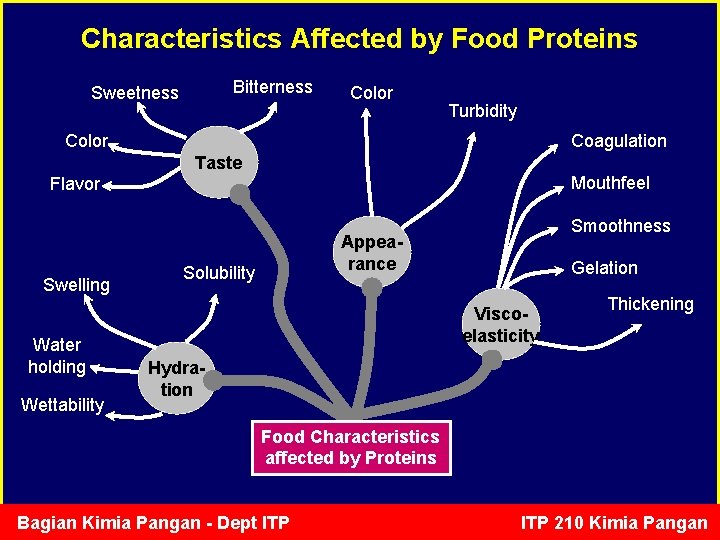
Characteristics Affected by Food Proteins Bitterness Sweetness Color Turbidity Color Coagulation Taste Mouthfeel Flavor Swelling Water holding Wettability Smoothness Appearance Solubility Gelation Viscoelasticity Thickening Hydration Food Characteristics affected by Proteins Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
Emulsification • Since protein molecules contain both hydrolytic and hydrophobic characteristics, proteins can stabilize emulsions by acting at the oil-water interface. • This functional property is important in the formation of many common food products, such as salad dressings, sauces, frankfurters, and sausages. • Foods, such as meat tissue, milk, eggs, and soy contain proteins that can be isolated as emulsifiers. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Protein Gelation • Proteins can form a well-ordered gel matrix by balancing protein-protein and protein-solvent interactions in food products. • These gel matrices can hold water, fat, and other food ingredients to produce various food products, including bread dough, communited meat products, gelatin desserts, tofu, and yoghurt. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
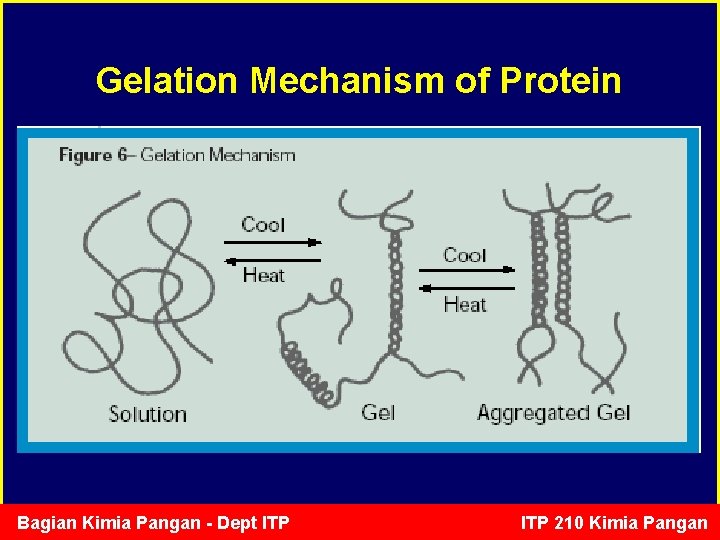
Gelation Mechanism of Protein Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Water Holding Capacity • The ability of a protein molecule to bind water has to do with the presence of hydrophylic and charged groups in its structure. This property is called water-holding capacity of protein • Example: meat retains water during application of external forces such as cutting, heating, grinding, and pressing. • Affected by: p. H, salt, and temperature. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Water Holding Capacity • When the negative and positive charges on a protein equal to each other, protein : protein interactions are at maximum. • When the protein is not electrically neutral, these interactions lessen, allowing for greater water: protein associations. • By increasing salt concentration, more Na+ and Cl- ions are available to bind the charged groups on protein fiber molecules. This reduces the protein fiber interactions with each other in favor of increase protein fiber : water associations. • As temperature increases to 80 o. C, water binding increases in proteins that form thermally induced gels, because gelations traps water inside a three-dimensional gel network in addition to creating gel surface binding of water molecules. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Protein Functionality: Fat Binding Properties • Hydrophobic proteins effectively lower surface tension and bind many lipophilic materials, such as lipids, emulsifiers, and flavor materials. • The capacity of protein to bind fat is important in the production of meat extenders and replacers, in which the absorption of fat by proteins enhances flavor retention and improves mouthfeel. • Fat is absorbed through physical entrapment. • Fat absorption can be increased if the protein is modified chemically to increase its bulk density. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Contribution of Amino Acids to Taste and Flavor Contribution to Taste/Flavor Amino Acids Sweet taste Gly, Ala, Thr, Pro, Ser, Glu Bitter taste Phy, Tyr, Arg, Leu, Val, Met, His Umami and Sour taste Glu, Asp Cheese and miso (soy sauce) are examples of fermented foods whose flavor is largely caused by their amino acid composition. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Protein Hydrolysis • Protein molecules may undergo hydrolysis to form shorter chains. • The reaction usually is the result of enzymatic action by peptidases, but sometimes collagen is cleaved by acid hydrolysis. • The result if cleavage of the peptide bond and uptake of a molecule of water • The shorter chains resulting from hydrolysis show increased solubility and decreased ability to thicken food products. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Degradasi enzimatik ü Meningkatnya kandungan peptida dan asam amino pada tempe (dikehendaki) ü Meningkatnya kandungan peptida pada keju karena pertumbuhan mikroba yang tidak dikehendaki pada saat pemeraman, menyebabkan keju berasa pahit. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Food Enzyme Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

What is enzyme? • Specialized protein molecules that speed up chemical reactions in living cells (biological catalysts). • Specific for particular substances • Composed of 200 -1000 amino acids residues covalently linked. • Molecular weight: 12. 000 – 1 million Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Sumber Enzim • Plant – Kedelai: lipoksigenase • Animal – Sapi: renin • Microbial sources – Bakteri asam laktat: memproduksi enzim proteolitik membantu degradasi protein keju Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Some Properties of Enzymes Cofactor Coenzyme Small inorganic ion that helps catalyze reactions Exp. Cu+2 ; Mg+2 ; Mn+2, etc Small, organic, non-protein ligands (such as NAD+, B-vitamins), which catalyze reaction most often by gaining/losing electron, transfers group or break bonds. Reaction path E + S <---> E + P sucrase reaction Active site Portion of enzyme protein that attaches to the substrate by means of weak chemical bonds (H -bonds, ionic bonds, hydrophobic forces, etc) Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

How important in food processing? • In foods: – Indigenous food enzymes, e. g, papain in papaya – Added enzyme: concentrate or isolates – Enzyme produced by microorganisms and present either as contaminants or added as cultures • Important in food processing because of the roles they play in the composition, processing and shelf life of foods. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

How important in food processing? Desirable: – Used in food processing: • Papain tenderizes meat • Amylase/glucoamylase is used in the production of glucose • Proteases in the production of bread – To measure adequacy of processing • Phosphatase in milk pasteurization and catalase and peroxidase in vegetable blanching. Undesirable: – Cause quality changes in food color, texture, flavor and odor during storage and use • Lipases produce rancidity • Polyphenol oxidase results in enzymatic browning • Lipoxigenase produces off-flavor in soymilk Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Examples of Several Food Enzymes Food Enzyme Reaction Amylase Hydrolysis of starch Chlorophyllase Changes color of chlorophyll Catalase Decomposition of hydrogen peroxide (H 2 O 2) Glucose oxidase Oxidation of glucose Lactase Hydrolysis of lactose Lipase Hydrolysis of triglycerides Lipoxygenase Oxidation of unsaturated lipid Pectinase Clarification of wines; modification of jam jelly texture Peptidase Hydrolysis of proteins Polyphenol oxidase (PPO) Browning of fruits and vegetables Proteases Elimination of chill haze on cold meat packaging; meat tenderizers Rennet Coagulation of milk protein Sucrase Hydrolysis of sucrose Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Bau Langu (Beany Flavor) • Disebabkan enzim lipoksigenase yang aktif dengan penghancuran jaringan • Enzim lipoksigenase menghidrolisa asam lemak tidak jenuh menghasilkan senyawa volatil. – Pencegahan : pemanasan untuk inaktivasi lipoksigenase, p. H rendah untuk penghambatan aktivitas enzim, ekstraksi lemak Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
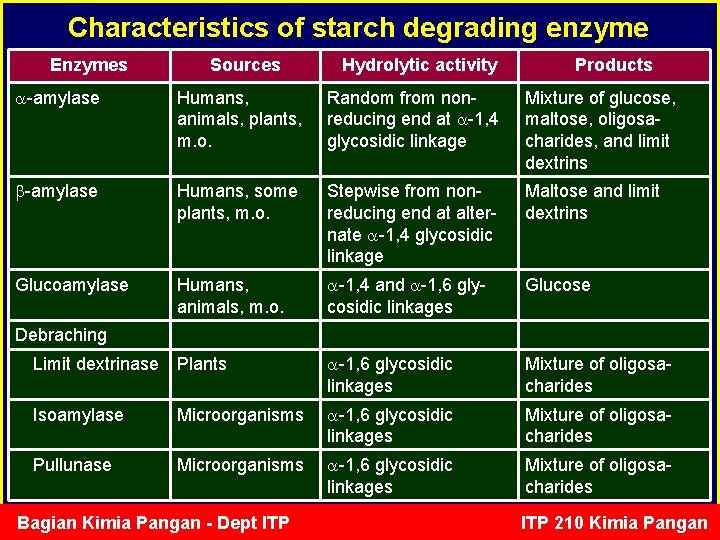
Characteristics of starch degrading enzyme Enzymes Sources Hydrolytic activity Products -amylase Humans, animals, plants, m. o. Random from nonreducing end at -1, 4 glycosidic linkage Mixture of glucose, maltose, oligosacharides, and limit dextrins -amylase Humans, some plants, m. o. Stepwise from nonreducing end at alternate -1, 4 glycosidic linkage Maltose and limit dextrins Glucoamylase Humans, animals, m. o. -1, 4 and -1, 6 glycosidic linkages Glucose Limit dextrinase Plants -1, 6 glycosidic linkages Mixture of oligosacharides Isoamylase Microorganisms -1, 6 glycosidic linkages Mixture of oligosacharides Pullunase Microorganisms -1, 6 glycosidic linkages Mixture of oligosacharides Debraching Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Pemanfaatan Enzim di industri roti • Adonan roti: kuat dan elastis yang dapat menahan gelembung gas membentuk volume roti yang besar. • Tepung terigu mengandung sedikit gula yang dapat difermentasi (0. 5% mono dan disakarida) tidak cukup untuk proses fermentasi untuk menghasilkan adonan yang baik dan volume roti yang besar. • Ditambahkan -amylase untuk menghasilkan maltosa dari pati terigu digunakan oleh Sacharomyces cereviceae untuk membentuk gas CO 2 dan etanol. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
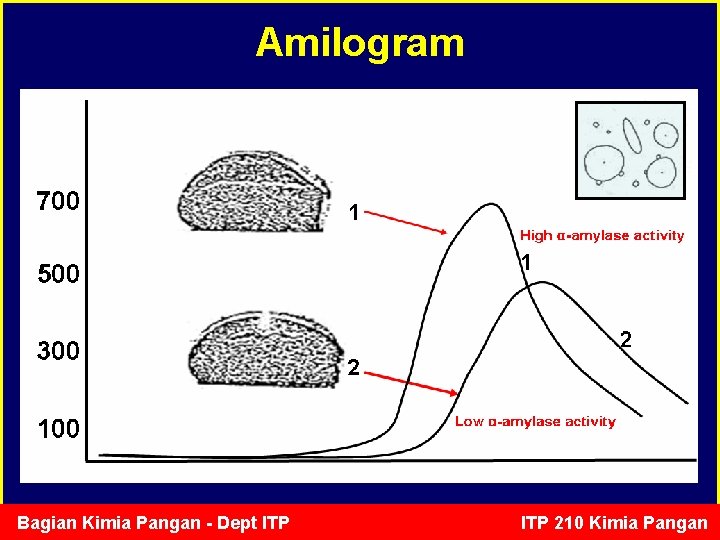
Amilogram Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
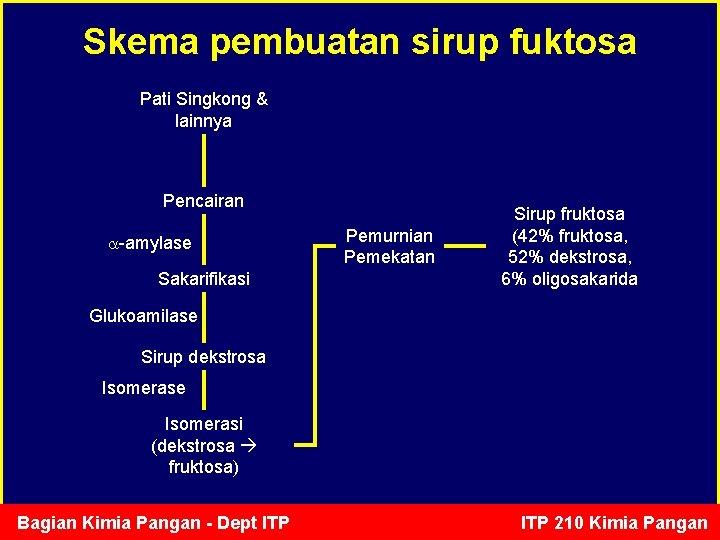
Skema pembuatan sirup fuktosa Pati Singkong & lainnya Pencairan -amylase Sakarifikasi Pemurnian Pemekatan Sirup fruktosa (42% fruktosa, 52% dekstrosa, 6% oligosakarida Glukoamilase Sirup dekstrosa Isomerase Isomerasi (dekstrosa fruktosa) Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Lysozyme • The enzyme lysozyme is present in some foods such as egg white, shellfish, milk and plant tissues • Lysozyme hydrolyzes the mucopeptide layer present in the cell wall of Gram positive bacteria and in the middle membrane of Gram negative bacteria • The anti microbial effect is manifested by the lysis of cells Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Active Site of Enzyme • Active Site: the region on the surface of enzyme where catalytic occurs • The active site of an enzyme is directly involved in binding and catalytic action • Active site is a small, three-dimensional region that contains amino acids that bind non-covalently with the substrate. – Exp. Active site of papain is adjacent histidine and cysteine residues that form non-covalent bonds with functional groups of protein substrate • The location where the substrate joins with the enzyme to form enzyme-substrate complex: transition state, reaction intermediate, or ES-complex Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

How an Enzyme Works Substrate (S) + Enzyme (E) ES Enzyme (E) + Product (P) • The green one is enzyme. Unchanged at the end of the reaction • The orange one is substrate molecules acted on by the enzyme. Changed to products. • The blue one is water. Involved in the reaction. • Lower activation energy (the amount of energy needed to convert substrate molecules from the ground or baseline energy state to the ES complex) Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
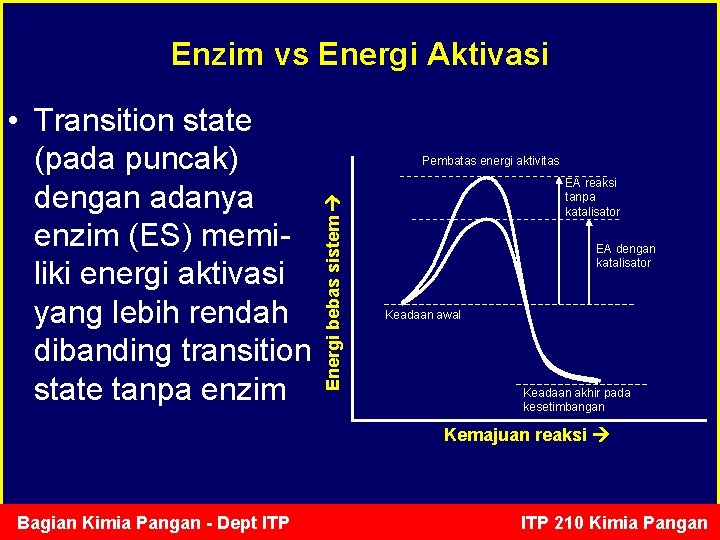
Enzim vs Energi Aktivasi Pembatas energi aktivitas Energi bebas sistem • Transition state (pada puncak) dengan adanya enzim (ES) memiliki energi aktivasi yang lebih rendah dibanding transition state tanpa enzim EA reaksi tanpa katalisator EA dengan katalisator Keadaan awal Keadaan akhir pada kesetimbangan Kemajuan reaksi Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

How an Enzyme Works • The substrate molecules (orange rectangle) diffuse in [from the left] and, as in all hydrolysis reactions, water (represented by the small blue rectangle) participates in the breakdown, with the release of products (red and yellow squares), which diffuse away [to the right]. • The process continues as long as there are substrate molecules to be converted, as the enzyme molecule (green) is unchanged at the end of the reaction. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
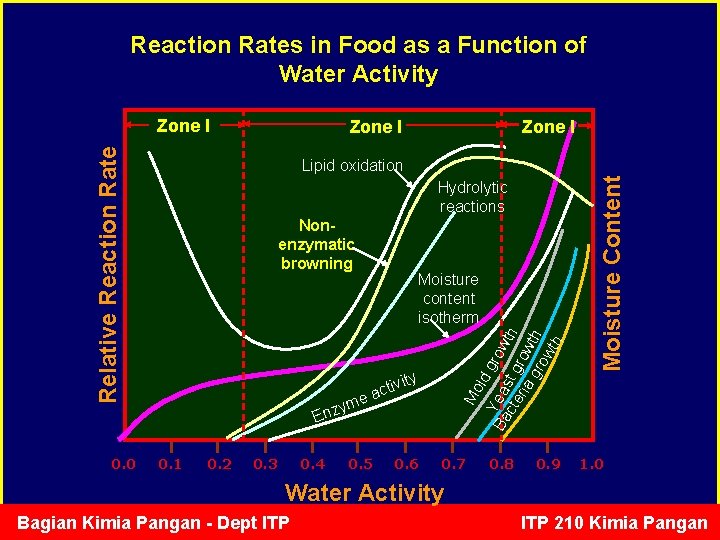
Reaction Rates in Food as a Function of Water Activity Zone I Relative Reaction Rate Zone I 0. 0 Zone I Hydrolytic reactions Nonenzymatic browning Mo ld gro Y Ba ea cte st g wth ria row gr th ow th Moisture content isotherm ity ctiv a e m y Enz 0. 1 0. 2 0. 3 0. 4 0. 5 0. 6 0. 7 0. 8 0. 9 Moisture Content Lipid oxidation 1. 0 Water Activity Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

How to name enzyme? • By adding suffix “ase” to the name of enzyme substrate or reaction. For example: – Urease: acts on urea – Glucose oxidase oxidizes glucose to gluconic acid • By adding suffix “in” to the name of enzyme source. For example: – Papain is obtained from papaya plant Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

International Commission of Enzyme • Develop a systematic nomenclature and classification scheme for enzyme. • Trivial name, systematic name, enzyme commission (EU) number, specific reaction. – For example: amylase • Trivial name: -amylase • Systematic name: -1, 4 -Glucan-4 -glucanohydrolase • EC number: 3. 2. 1. 1 • Reaction: hydrolysis of -1, 4 -glucan links • Enzyme is divided into six classes based on types of reactions they catalyze: – Oxidoreductase (1); Transferase (2); Hydrolase (3); Lyase (4); Isomerase (5); Ligase (6) Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Enzim Dalam Pengolahan Pangan • Dua kelompok enzim yang banyak digunakan dalam pengolahan pangan: – Oksidoreduktase – Hidrolase • Yang jarang digunakan dalam pengolahan pangan: – Liase – Ligase Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Oxidoreduktase (1) • Enzim yang dapat mengkatalisis reaksi oksidasi atau reduksi suatu bahan. • Yang utama: – Oksidase: mengkatalisis reaksi antara substrat dengan molekul oksigen. • Katalase, peroksidase, tirosinase, asam askorbat oksidase, lipoxidase (lipoxygenase) – Dehidrogenase: enzim yang aktif dalam pengambilan atom hidrogen dari substrat. • Suksinat dehidrigenase: memecah asam glutamat menjadi asam ketoglutarat dan NH 3 • Laktat dehidrogenase: memecah asam laktat menjadi asam piruvat. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Transferase (2) • Enzim yang ikut serta dalam reaksi pemindahan (transfer) suatu radikal atau gugus (AB + C A + CB). • Yang utama: – Fosforilase: memecah ikatan glikosida -1, 4 pati dengan pertolongan ion fosfat membentuk -Dglukosa-1 -fosfat. – Transfosforilase: memindahkan gugus fosfat dari suatu molekul ke molekul lainnya, misal memindahkan gugus fosfat dari ATP kepada heksosa menghasilkan heksosa-monofosfat dan ADP. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Transferase (2) – Transaminase: mengkatalisis reaksi antara asam amino dan asam -keto menghasilkan asam amino baru dan asam keto baru. Misal reaksi antara asam glutamat dan asam oksaloasetat menghasilkan asam -ketoglutarat dan asam aspartat. – Transmetilase: mengkatalisis pemindahan gugus metil dari suatu mole-kul ke molekul lainnya. Contoh: memindahkan gugus metil dari metionin kepada asam guanidoasetat membentuk homosistein dan kreatin. – Transasetilase: membentuk molekul bergugus asetil. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Hidrolase (3) • Mengkatalisis reaksi hidrolisis suatu substrat atau pemecahan substrat dengan pertolongan molekul air. • Yang utama: – Lipase: menghidrolisis ikatan ester pada lemak alami menjadi gliserol dan asam lemak. – Glikosidase: menghidrolisis ikatan glikosida – Aminopeptidase (tripsin): menghidrolisis ikatan peptida. – Urease: menghidrolisis urea menjadi amonia dan CO 2. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Liase (4) • Enzim yang aktif dalam pemecahan ikatan C-C dan ikatan C-O dengan tidak menggunakan molekul air. • Yang utama: – Dekarboksilase: memecah ikatan C-C – Karbonat anhidrase: memecah ikatan C-O Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Isomerase (5) • Enzim yang mengkatalisis reaksi perubahan konfigurasi molekul dengan cara pengaturan kembali atom-atom dalam molekul substrat, sehingga dihasilkan molekul baru yang merupakan isomer dari substrat, misal merubah aldosa menjadi ketosa. • Yang utama: – Fosfoheksosa isomerase: mengubah glukosa 6 -P (glukosa 6 -fosfat) menjadi fruktosa 6 -P – Fosfomanosa isomerase: merubah glukosa 6 -P menjadi manosa 6 -P Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Ligase (6) • Mengkatalisis pembentukan ikatan-ikatan tertentu, misalnya pembentukan ikatan CO, C-C dan C-S dalam biosintesis koenzim A serta pembentukan ikatan C-N dalam sintesis glutamin. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Nomor Kode Enzim (EC) • Terdiri dari 4 digit: – Digit 1: Nomor urut dari salah satu 6 pembagian umum. – Digit 2: Subklas, substrat, atau jenis reaksi – Digit 3: Subklas, jenis reaksi yang lebih detail lagi, biasanya memerlukan NAD dan ko-enzim – Digit 4: nomor seri dalam subklas tersebut. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Pedoman Klasifikasi Enzim • Oksidoreduktase – 1. 1. Bekerja pada gugus -CHOH – 1. 2. Bekerja pada gugus keton atau aldehida – 1. 3. Bekerja pada gugus -CH-CH– 1. 4. Bekerja pada gugus –CH-NH 2 – 1. 5. Bekerja pada gugus –C-NH– 1. 11. Bekerja pada gugus H 2 O 2 Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Pedoman Klasifikasi Enzim • Tranferase – 2. 1. Pemindahan (transfer) gugus satu carbon seperti metil atau karboksil – 2. 2. Pemindahan gugus aldehida dan keton – 2. 3. Pemindahan gugus asil – 2. 4. Pemindahan gugus glikosil – 2. 5. Pemindahan gugus alkil – 2. 6. Pemindahan gugus nitrogen – 2. 7. Pemindahan gugus yang mengandung fosfor – 2. 8. Pemindahan gugus yang mengandung sulfur Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Pedoman Klasifikasi Enzim • Hidrolase – 3. 1. Bekerja pada ikatan ester – 3. 2. Bekerja pada senyawa glikosil – 3. 3. Bekerja pada ikatan ester-tio – 3. 4. Bekerja pada ikatan peptida – 3. 5. Bekerja pada ikatan C-N yang bukan peptida – 3. 6. Bekerja pada ikatan asam anhidrida – 3. 7. Bekerja pada ikatan C-C – 3. 8. Bekerja pada ikatan halida Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Pedoman Klasifikasi Enzim • Liase – 4. 1. Bekerja pada –C=C– 4. 2. Bekerja pada –C=O – 4. 3. Bekerja pada C=N– 4. 4. Bekerja pada –C=S – 4. 5. Bekerja pada C-halida Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Pedoman Klasifikasi Enzim • Isomerase – 5. 1. Rasemase dan epimerase – 5. 2. Cis-trans isomerase – 5. 3. Oksidoreduktase intramolekul – 5. 4. Transferase intramolekul – 5. 5. Liase intramolekul Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Pedoman Klasifikasi Enzim • Ligase – 6. 1. Pembentukan ikatan C-O – 6. 2. Pembentukan ikatan C-S – 6. 3. Pembentukan ikatan C-N – 6. 4. Pembentukan ikatan C-C Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Nomenklatur enzim yang penting dalam pengolahan pangan No. EC Nama sistematiknya Nama trivial 1. 1. 3. 4. -D-glukosa: O 2 oksidoreduktase Glukosa oksidase 1. 10. 3. 1 -difenol: 02 oksidoreduktase Katekol oksidase 1. 1. 6 H 2 O 2: H 2 O 2 oksidoreduktase Katalase 1. 1. 7 Donor: H 2 O 2 oksidoreduktase Lipoksigenase/lipoksidase 1. Oksidoreduktase Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

No. EC Nama sistematiknya Nama trivial 2. 1. 1. 5. -1, 6 -glucan; D-fruktosa 2 glukosil transferase Dekstran sukrase 2. 4. 1. 19 -1, 4 -glukan-4 gliosil transferase Enzim B. macerans 3. 1. 1. 1. Karbolik ester hidrolase Karboksil esterase 3. 1. 1. 3. Gliserol ester hidrolase Lipase 3. 1. 1. 11 Pektin pektil hidrolase Pektin esterase 3. 2. 1. 1. -1, 4 -glukanohidrolase -amilase 3. 2. 1. 2. -1, 4 -glukan maltohidrolase -amilase D-glukosa-6 -fosfat ketol isomerase Glukosa isomerase 2. Transferase 3. Hidrolase 4. Isomerase 5. 3. 1. 9. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Faktor yang mempengaruhi aktivitas enzim • Suhu – Mula-mula dengan meningkat suhu, aktifitas enzim meningkat. Tetapi, pada suhu tertentu, enzim mengalami inaktivasi. – Pada suhu rendah, laju inaktivasi enzim sangat lambat • p. H – Aktivitas maksimum: 4. 5 -8. 0 (tergantung jenis enzim) – Pada kisaran p. H di luar kisaran tersebut, umumnya enzim mengalami inaktivasi. • Aktivitas air – Pada aw yang rendah, enzim tidak aktif. – Pada aw tinggi, enzim aktif • Garam – Kadar elektrolit yang tinggi umumnya mempengaruhi kelarutan protein mempengaruhi aktifitas enzim. Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
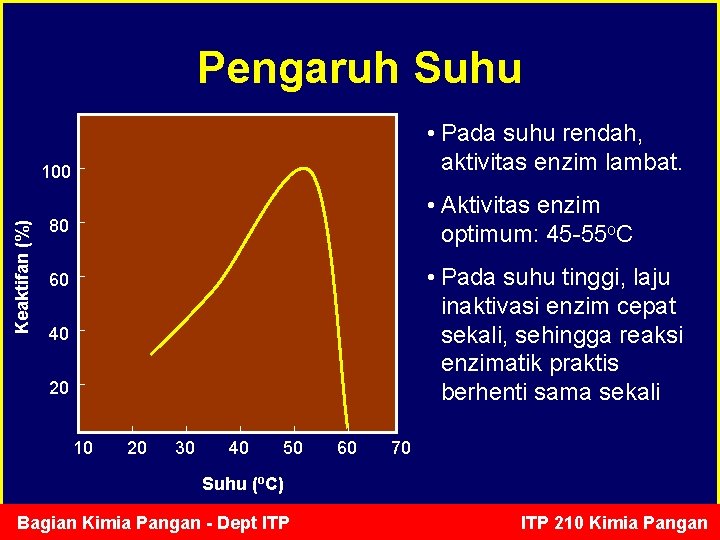
Pengaruh Suhu • Pada suhu rendah, aktivitas enzim lambat. Keaktifan (%) 100 • Aktivitas enzim optimum: 45 -55 o. C 80 • Pada suhu tinggi, laju inaktivasi enzim cepat sekali, sehingga reaksi enzimatik praktis berhenti sama sekali 60 40 20 10 20 30 40 50 60 70 Suhu (o. C) Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
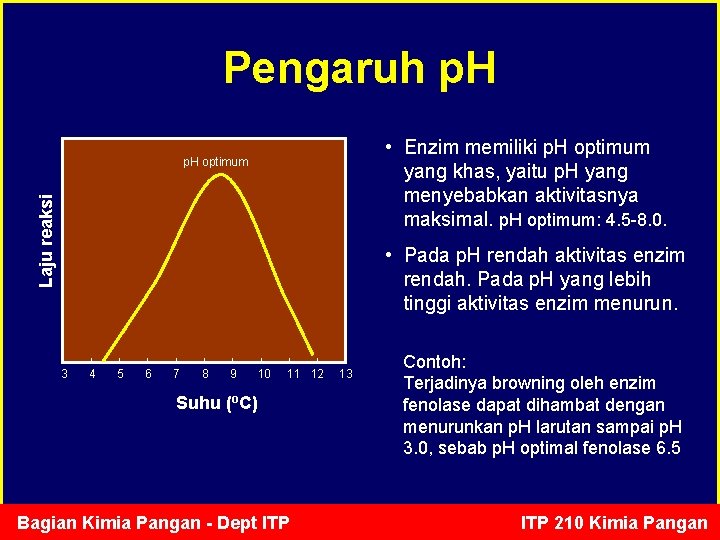
Pengaruh p. H • Enzim memiliki p. H optimum yang khas, yaitu p. H yang menyebabkan aktivitasnya maksimal. p. H optimum: 4. 5 -8. 0. Laju reaksi p. H optimum • Pada p. H rendah aktivitas enzim rendah. Pada p. H yang lebih tinggi aktivitas enzim menurun. 3 4 5 6 7 8 9 10 11 12 Suhu (o. C) Bagian Kimia Pangan - Dept ITP 13 Contoh: Terjadinya browning oleh enzim fenolase dapat dihambat dengan menurunkan p. H larutan sampai p. H 3. 0, sebab p. H optimal fenolase 6. 5 ITP 210 Kimia Pangan

p. H optimum beberapa enzim • • • Pepsin : 1. 5 Tripsin : 7, 7 Katalase : 7. 6 Arginase : 9, 7 Fumarase : 7, 8 Ribonuklease : 7, 8 Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Enzymatic reaction vs aw • At low Aw (<0. 2), freely mobile water is not available to carry out the reaction, and so enzymatic reactions tend to be surpressed in the lower regions of the sorption isotherm. • At high Aw, free water is available for the enzymatic reaction. • For example: lipoxygenase starts to be active after dry soybean (MC=14%) is soaked in water off-flavor Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
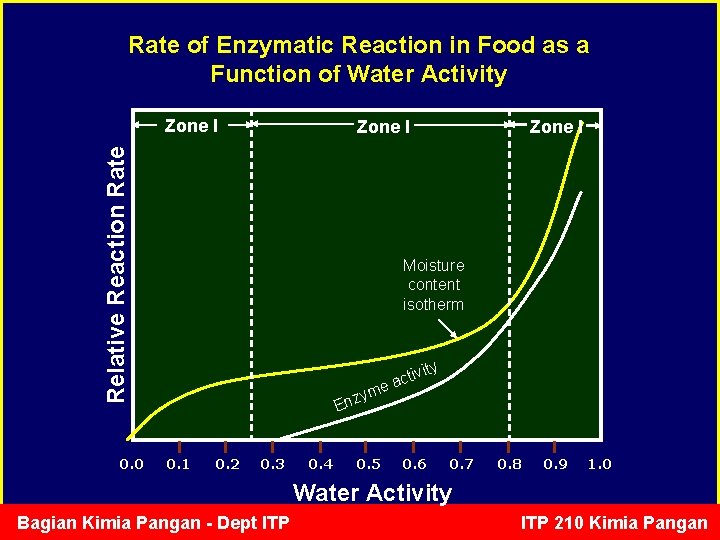
Rate of Enzymatic Reaction in Food as a Function of Water Activity Zone I Relative Reaction Rate Zone I 0. 0 Zone I Moisture content isotherm vity i t c a yme Enz 0. 1 0. 2 0. 3 0. 4 0. 5 0. 6 0. 7 0. 8 0. 9 1. 0 Water Activity Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan

Pengaruh Konsentrasi Substrat terhadap kecepatan reaksi enzimatik • Pada konsentrasi substrat yang amat rendah, kecepatan reaksi pun amat rendah. • Kecepatan reaksi akan meningkat dengan meningkatnya konsentrasi substrat. • Pada titik tertentu, peningkatkan konsentrasi substrat tidak dapat meningkatkan kecepatan reaksi (enzim menjadi jenuh oleh substratnya dan tidak dapat berfungsi lebih cepat). • Pada saat kecepatan reaksi tidak berubah lagi disebut mencapai kecepatan Maksimum (Vmax) Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
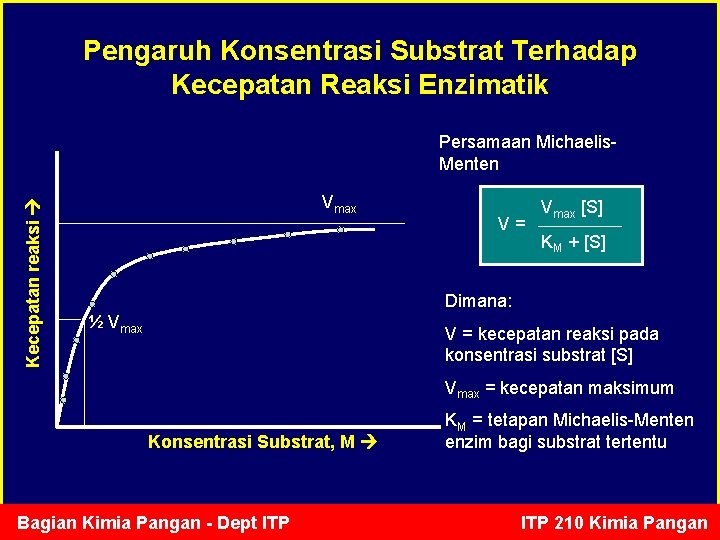
Pengaruh Konsentrasi Substrat Terhadap Kecepatan Reaksi Enzimatik Kecepatan reaksi Persamaan Michaelis. Menten Vmax V = Vmax [S] KM + [S] Dimana: ½ Vmax V = kecepatan reaksi pada konsentrasi substrat [S] Vmax = kecepatan maksimum Konsentrasi Substrat, M Bagian Kimia Pangan - Dept ITP KM = tetapan Michaelis-Menten enzim bagi substrat tertentu ITP 210 Kimia Pangan
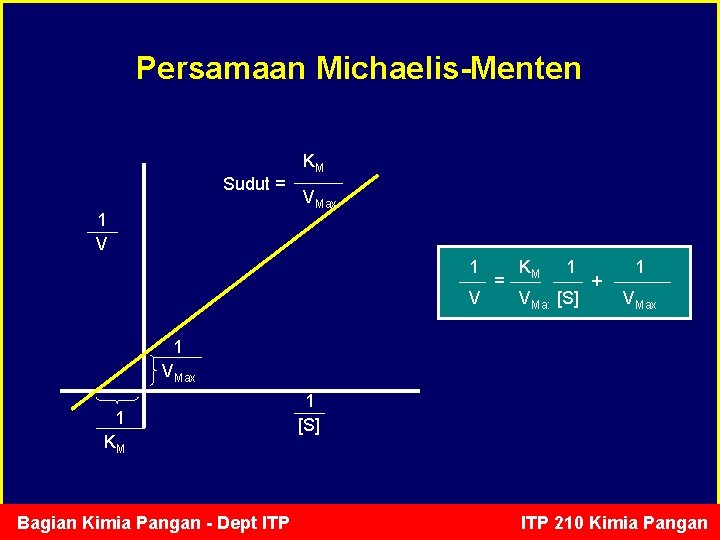
Persamaan Michaelis-Menten Sudut = 1 V KM VMax 1 V = KM 1 VMax [S] + 1 VMax 1 KM Bagian Kimia Pangan - Dept ITP 1 [S] ITP 210 Kimia Pangan

Tugas Mandiri • Buat artikel (per kelompok, max 3 orang) – Aplikasi enzim dalam industri pengolahan pangan yang lain (cara penggunaan, tujuan penggunaan, mutu akhir produk yang diinginkan) : yang diinginkan – Menghilangkan enzim dalam proses pengolahan pangan (mengapa, caranya, mutu akhir produk yang diinginkan): yang tidak diinginkan Bagian Kimia Pangan - Dept ITP 210 Kimia Pangan
- Slides: 121