IT Phsae transformation of metals Rate of Phase




- Slides: 33

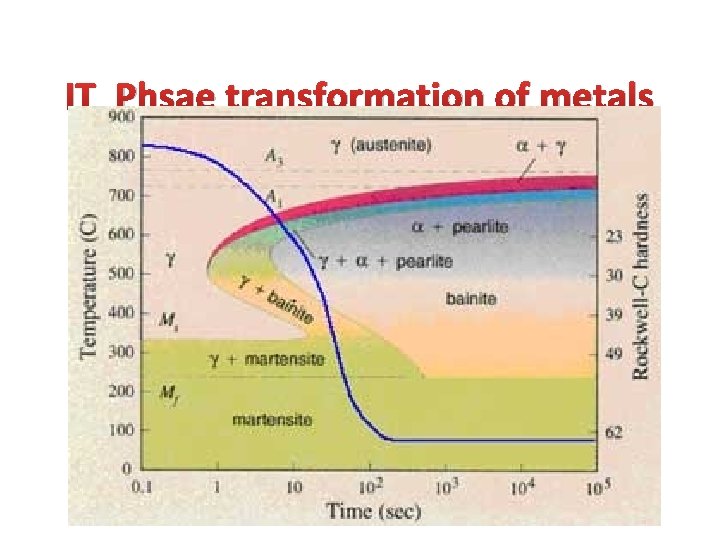
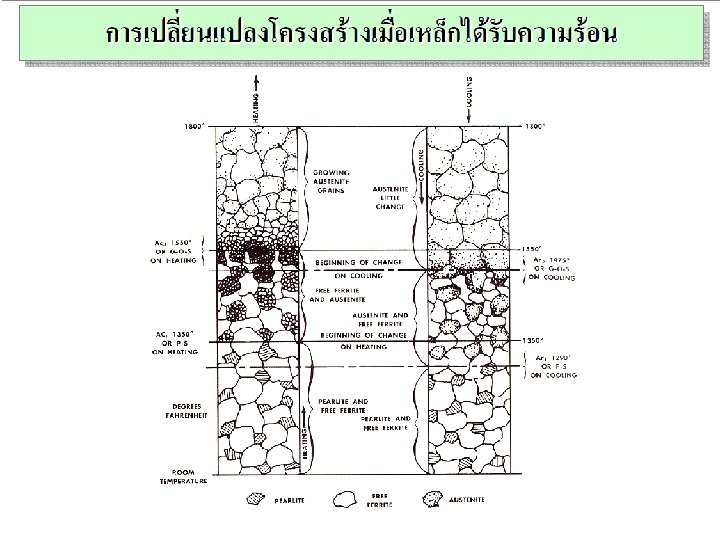
IT Phsae transformation of metals


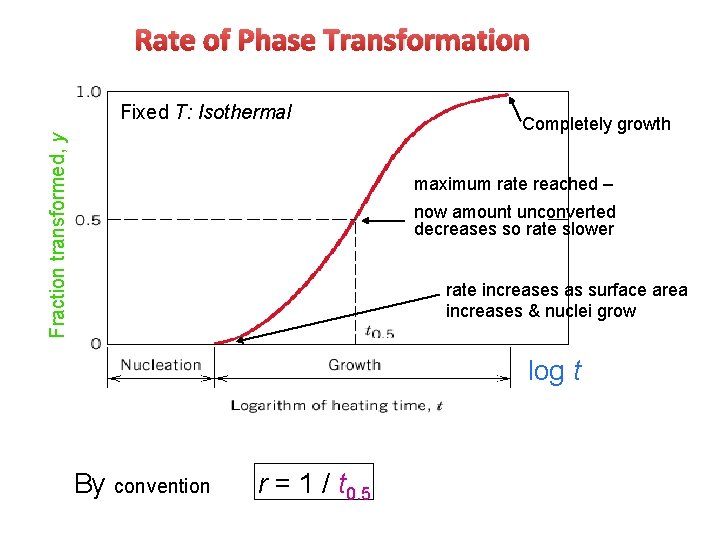
Rate of Phase Transformation Fraction transformed, y Fixed T: Isothermal Completely growth maximum rate reached – now amount unconverted decreases so rate slower rate increases as surface area increases & nuclei grow log t By convention r = 1 / t 0. 5

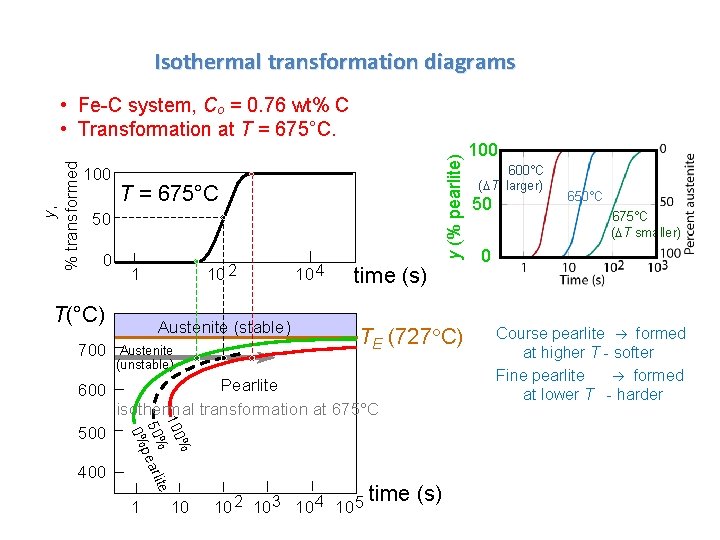
Isothermal transformation diagrams 100 y (% pearlite) y, % transformed • Fe-C system, Co = 0. 76 wt% C • Transformation at T = 675°C 50 0 10 2 1 T(°C) Austenite (stable) 10 4 time (s) TE (727 C) Austenite (unstable) 600 Pearlite isothermal transformation at 675°C 500 400 % 100 te 50%pearli 0% 700 1 10 10 2 10 3 10 4 10 5 time (s) 100 600°C (DT larger) 50 650°C 675°C (DT smaller) 0 Course pearlite formed at higher T - softer Fine pearlite formed at lower T - harder

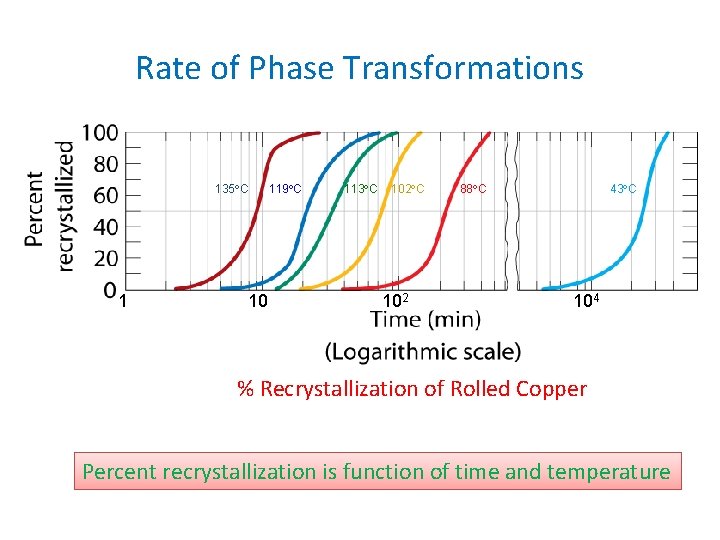
Rate of Phase Transformations 135 C 1 10 119 C 113 C 102 88 C 43 C 104 % Recrystallization of Rolled Copper Percent recrystallization is function of time and temperature

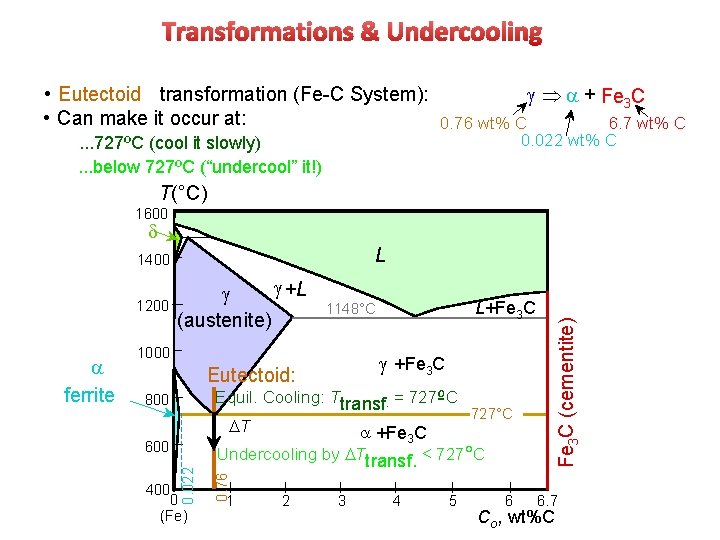
Transformations & Undercooling • Eutectoid transformation (Fe-C System): Þ + Fe 3 C • Can make it occur at: 0. 76 wt% C 6. 7 wt% C 0. 022 wt% C . . . 727ºC (cool it slowly). . . below 727ºC (“undercool” it!) T(°C) 1600 d 1200 L+Fe 3 C 1148°C 1000 +Fe 3 C Eutectoid: Equil. Cooling: Ttransf. = 727ºC 800 DT 400 0 (Fe) +Fe 3 C 727°C Undercooling by DTtransf. < 727 C 0. 76 600 0. 022 ferrite +L (austenite) 1 2 3 4 5 6 Fe 3 C (cementite) L 1400 6. 7 Co , wt%C

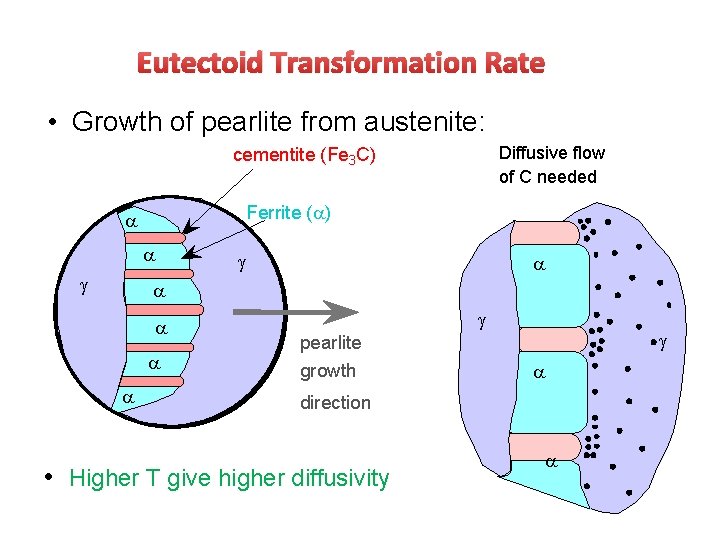
Eutectoid Transformation Rate • Growth of pearlite from austenite: Diffusive flow of C needed cementite (Fe 3 C) Ferrite ( ) pearlite growth direction • Higher T give higher diffusivity

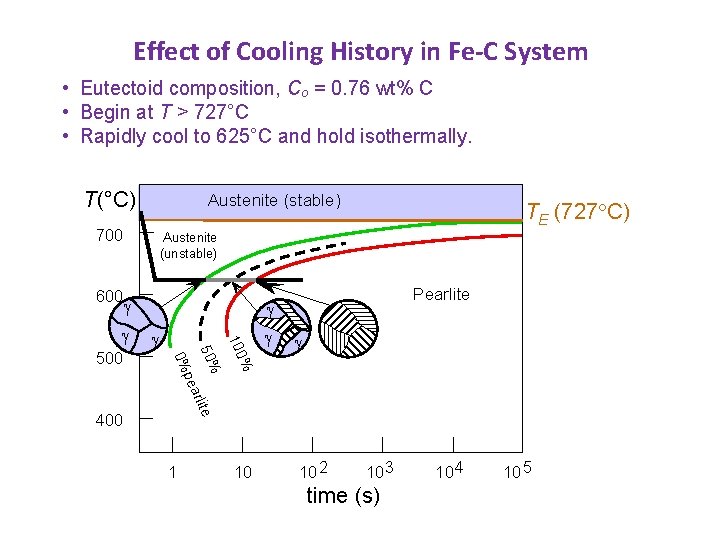
Effect of Cooling History in Fe-C System • Eutectoid composition, Co = 0. 76 wt% C • Begin at T > 727°C • Rapidly cool to 625°C and hold isothermally. T(°C) Austenite (stable) 700 600 TE (727 C) Austenite (unstable) % 100 pea 50% 500 Pearlite 400 1 10 10 2 10 3 time (s) 10 4 10 5

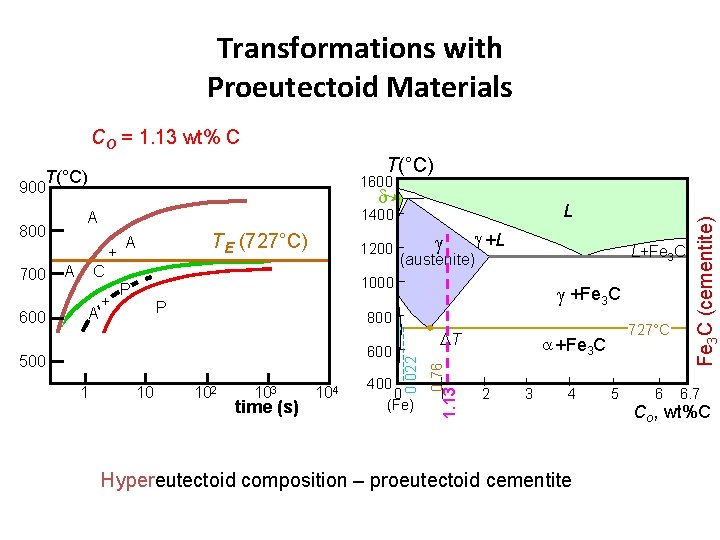
Transformations with Proeutectoid Materials CO = 1. 13 wt% C T(°C) 900 d 600 A C A + +L L+Fe 3 C (austenite) 1000 P P 500 10 102 103 time (s) 104 +Fe 3 C 800 600 1 DT 400 0 (Fe) 0. 76 700 1200 1 1. 13 + TE (727°C) A 0. 022 800 L 1400 A 727°C +Fe 3 C 2 3 4 Hypereutectoid composition – proeutectoid cementite 5 6 Fe 3 C (cementite) 1600 6. 7 Co , wt%C

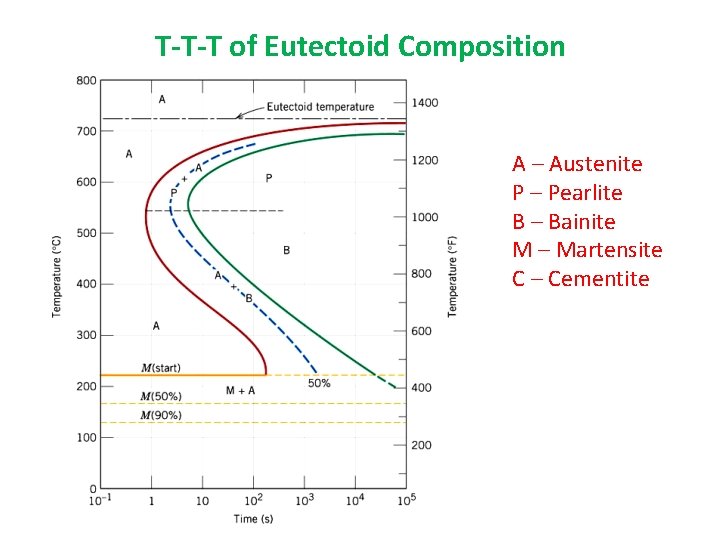
T-T-T of Eutectoid Composition A – Austenite P – Pearlite B – Bainite M – Martensite C – Cementite

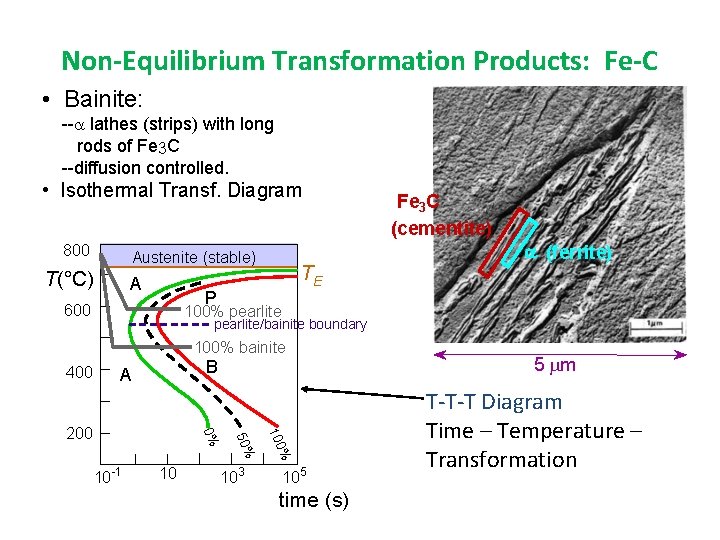
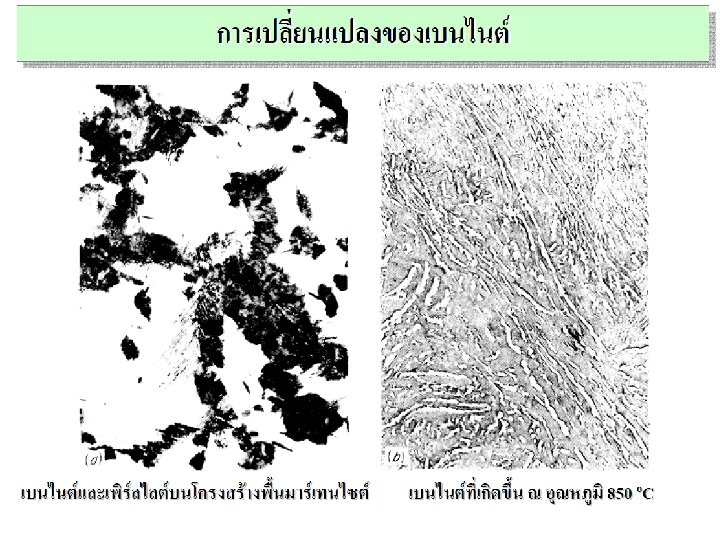
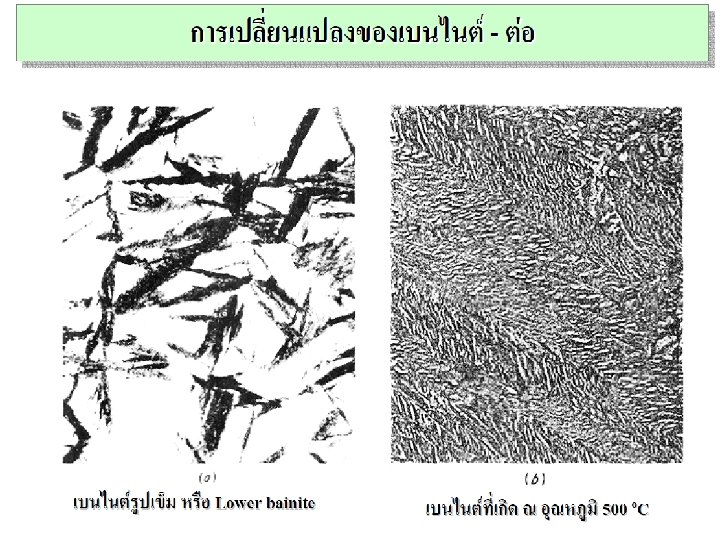
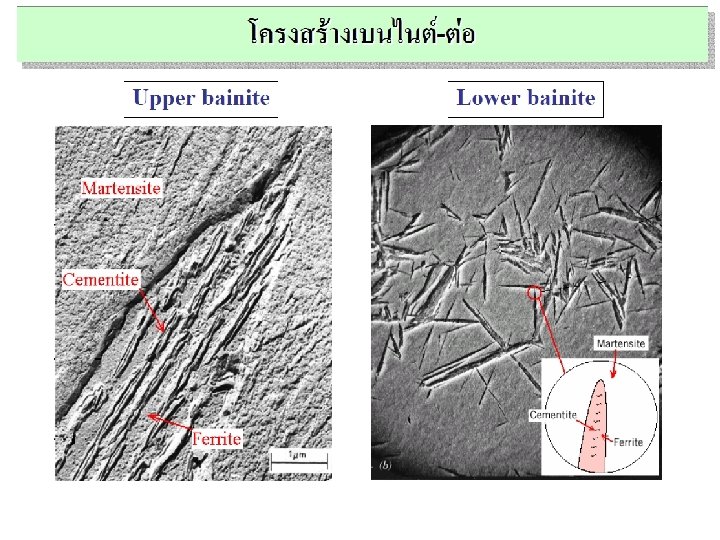
Non-Equilibrium Transformation Products: Fe-C • Bainite: -- lathes (strips) with long rods of Fe 3 C --diffusion controlled. • Isothermal Transf. Diagram 800 Austenite (stable) T(°C) A TE P 600 (ferrite) 100% pearlite/bainite boundary 100% bainite 400 Fe 3 C (cementite) B A 10 103 % 100 10 -1 50% 0% 200 105 time (s) 5 m T-T-T Diagram Time – Temperature – Transformation







Spheroidite: Fe-C System • Spheroidite: -- grains with spherical Fe 3 C -- diffusion dependent. -- heat bainite or pearlite for long times near TE (ferrite) Fe 3 C (cementite) 60 m

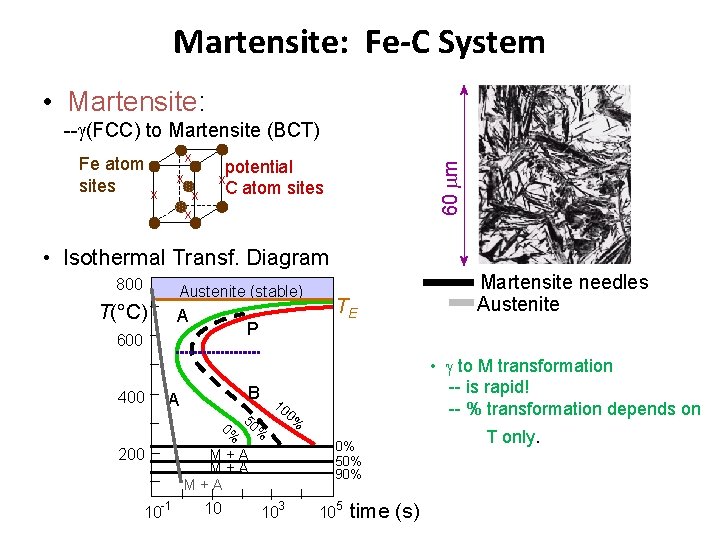
Martensite: Fe-C System • Martensite: -- (FCC) to Martensite (BCT) x x x potential C atom sites 60 m Fe atom sites x x x • Isothermal Transf. Diagram 800 Austenite (stable) T(°C) A P 600 400 A 200 10 -1 B 5 0% 0% • to M transformation -- is rapid! -- % transformation depends on 10 0% 0% 50% 90% M+A M+A 10 TE 103 Martensite needles Austenite 105 time (s) T only.

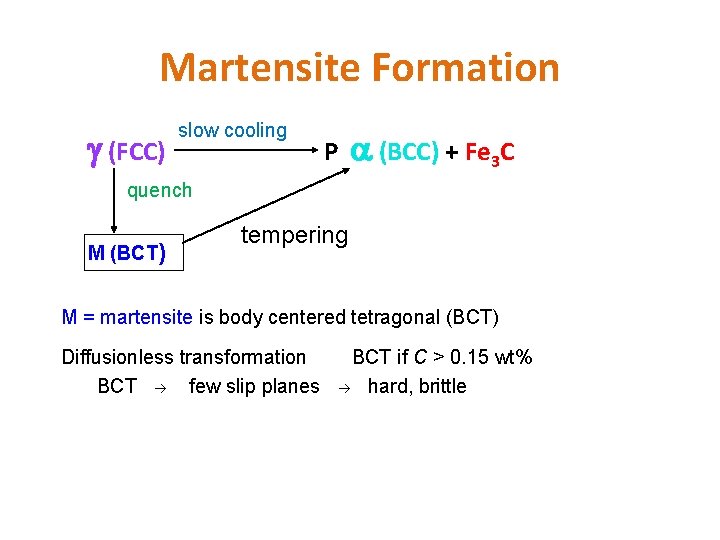
Martensite Formation (FCC) slow cooling P (BCC) + Fe 3 C quench M (BCT) tempering M = martensite is body centered tetragonal (BCT) Diffusionless transformation BCT few slip planes BCT if C > 0. 15 wt% hard, brittle

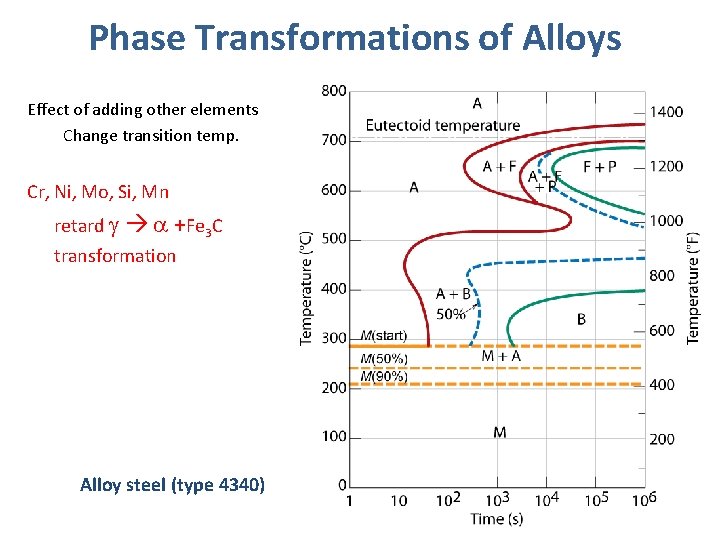
Phase Transformations of Alloys Effect of adding other elements Change transition temp. Cr, Ni, Mo, Si, Mn retard +Fe 3 C transformation Alloy steel (type 4340)

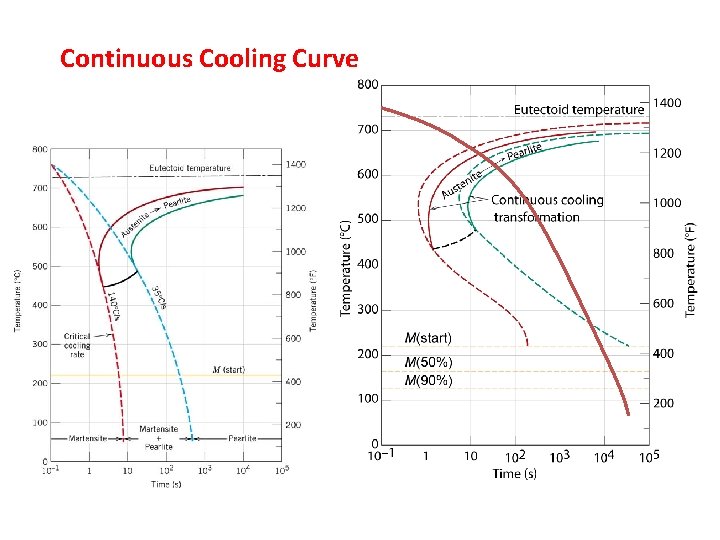
Continuous Cooling Curve

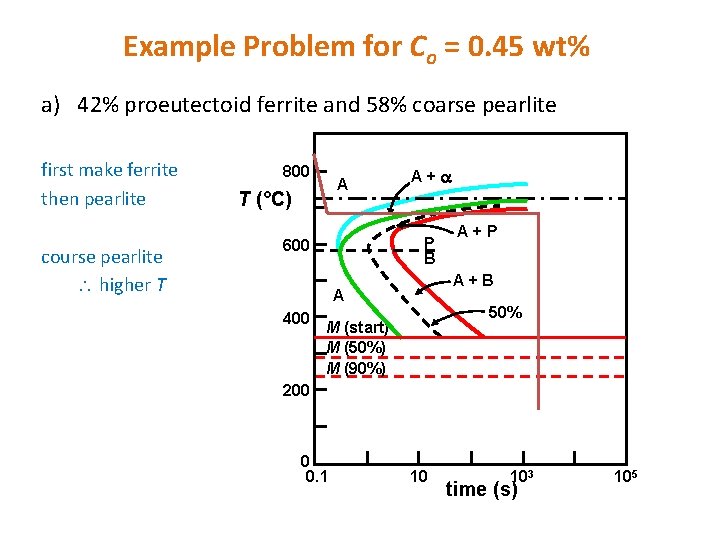
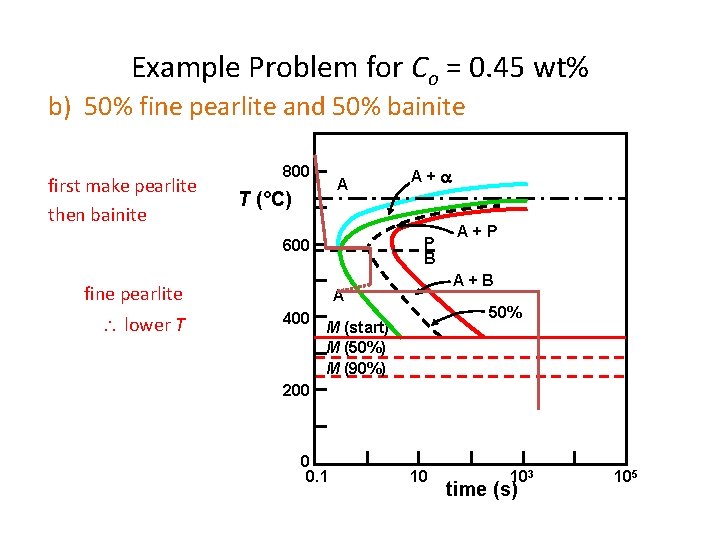
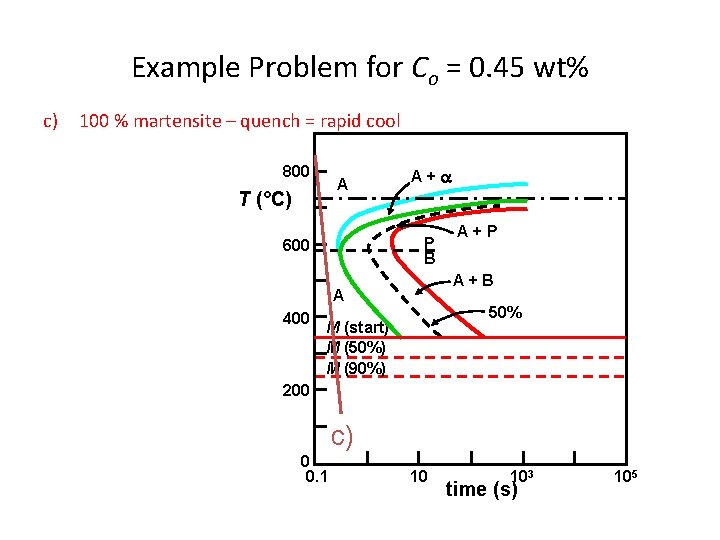
Dynamic Phase Transformations On the isothermal transformation diagram for 0. 45 wt% C Fe-C alloy, sketch and label the timetemperature paths to produce the following microstructures: a) 42% proeutectoid ferrite and 58% coarse pearlite b) 50% fine pearlite and 50% bainite c) 100% martensite

Example Problem for Co = 0. 45 wt% a) 42% proeutectoid ferrite and 58% coarse pearlite first make ferrite then pearlite course pearlite higher T 800 A T (°C) A+ P B 600 A+B A 400 A+P 50% M (start) M (50%) M (90%) 200 0 0. 1 10 103 time (s) 105

Example Problem for Co = 0. 45 wt% b) 50% fine pearlite and 50% bainite first make pearlite then bainite 800 A T (°C) P B 600 fine pearlite lower T A+ A+B A 400 A+P 50% M (start) M (50%) M (90%) 200 0 0. 1 10 103 time (s) 105

Example Problem for Co = 0. 45 wt% c) 100 % martensite – quench = rapid cool 800 A T (°C) A+ P B 600 A+B A 400 A+P 50% M (start) M (50%) M (90%) 200 c) 0 0. 1 10 103 time (s) 105

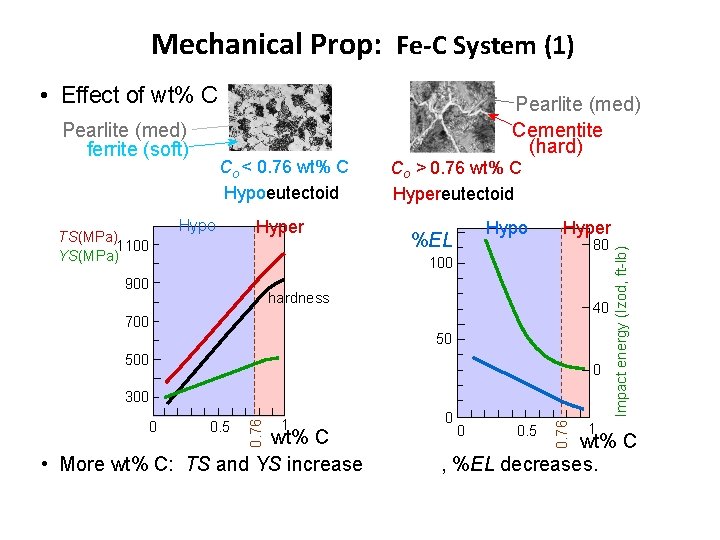
Mechanical Prop: Fe-C System (1) • Effect of wt% C TS(MPa) 1100 YS(MPa) Co < 0. 76 wt% C Hypoeutectoid Hypo Hyper Co > 0. 76 wt% C Hypereutectoid Hypo %EL Hyper 80 100 900 hardness 40 700 50 500 0 0. 5 1 wt% C • More wt% C: TS and YS increase 0 0 0. 5 0. 76 0 0. 76 300 1 Impact energy (Izod, ft-lb) Pearlite (med) ferrite (soft) Pearlite (med) Cementite (hard) wt% C , %EL decreases.

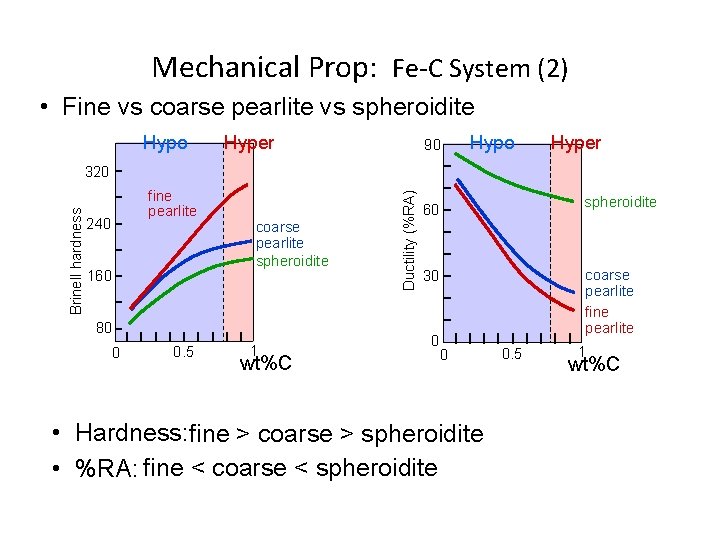
Mechanical Prop: Fe-C System (2) • Fine vs coarse pearlite vs spheroidite Hypo Hyper 90 Hypo Hyper fine pearlite 240 160 coarse pearlite spheroidite 80 0 0. 5 1 wt%C Ductility (%RA) Brinell hardness 320 spheroidite 60 coarse pearlite fine pearlite 30 0 0 • Hardness: fine > coarse > spheroidite • %RA: fine < coarse < spheroidite 0. 5 1 wt%C

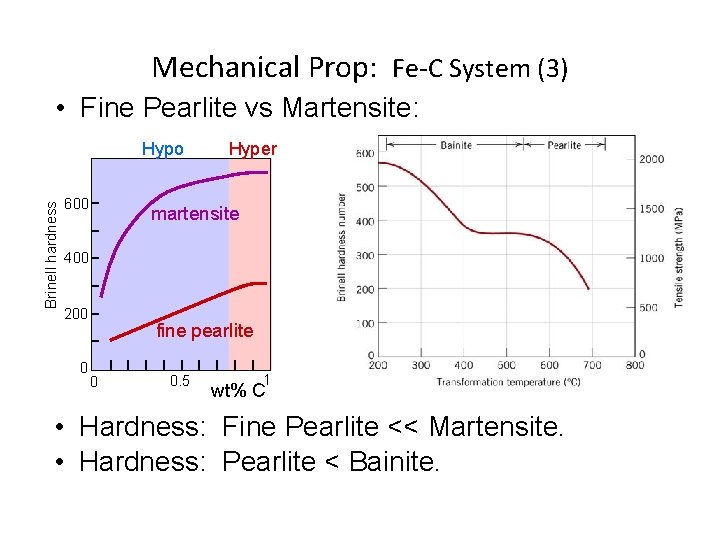
Mechanical Prop: Fe-C System (3) • Fine Pearlite vs Martensite: Brinell hardness Hypo 600 Hyper martensite 400 200 0 fine pearlite 0 0. 5 1 wt% C • Hardness: Fine Pearlite << Martensite. • Hardness: Pearlite < Bainite.

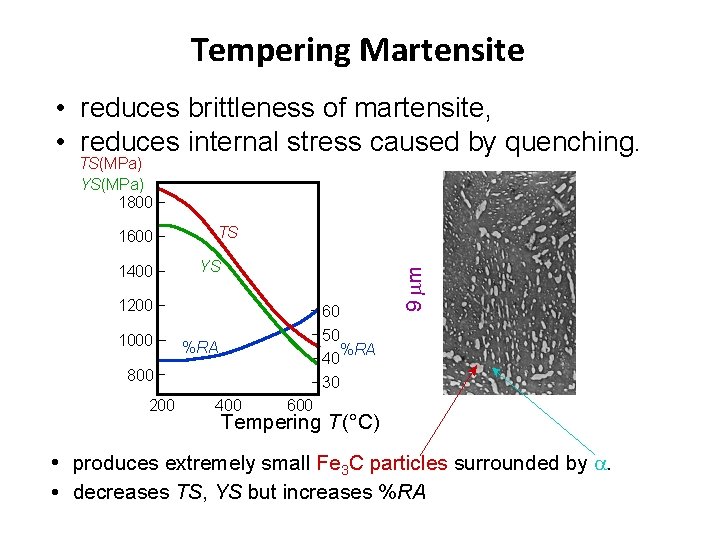
Tempering Martensite • reduces brittleness of martensite, • reduces internal stress caused by quenching. TS(MPa) YS(MPa) 1800 1400 TS YS 1200 1000 60 50 %RA 40 30 %RA 800 200 400 9 m 1600 Tempering T (°C) • produces extremely small Fe 3 C particles surrounded by . • decreases TS, YS but increases %RA

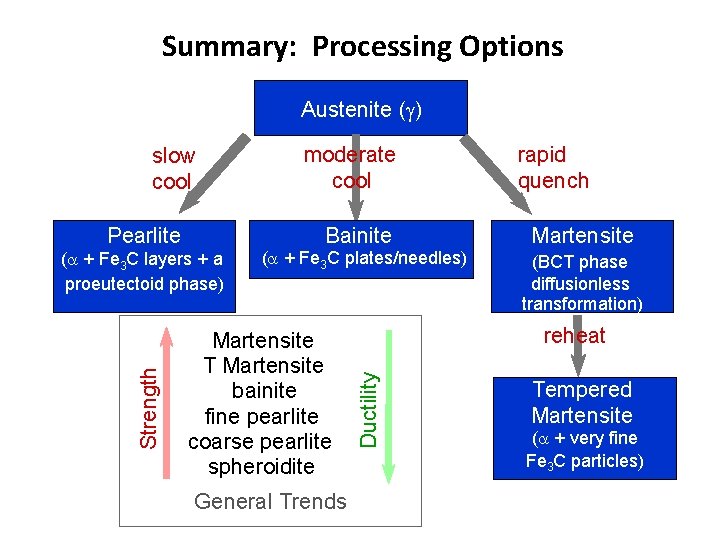
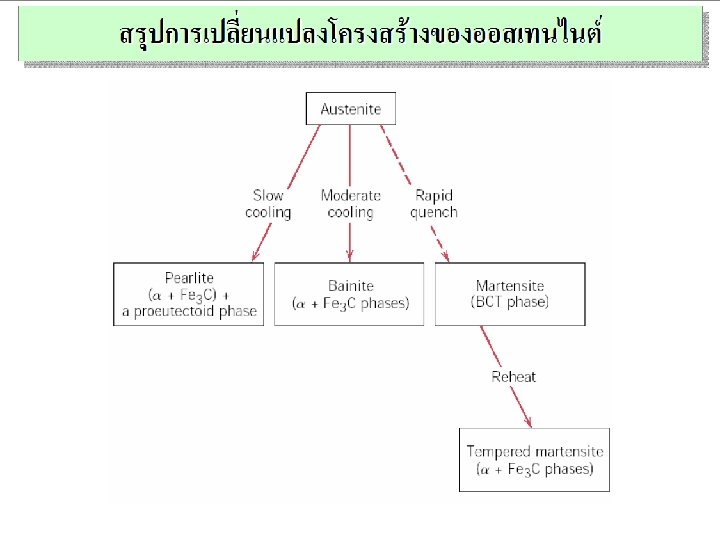
Summary: Processing Options Austenite ( ) slow cool moderate cool rapid quench Bainite Martensite ( + Fe 3 C layers + a proeutectoid phase) ( + Fe 3 C plates/needles) (BCT phase diffusionless transformation) Martensite T Martensite bainite fine pearlite coarse pearlite spheroidite General Trends reheat Ductility Strength Pearlite Tempered Martensite ( + very fine Fe 3 C particles)

