ISTITUTO DISTRUZIONE SUPERIORE DON LORENZO MILANI CLIL Content





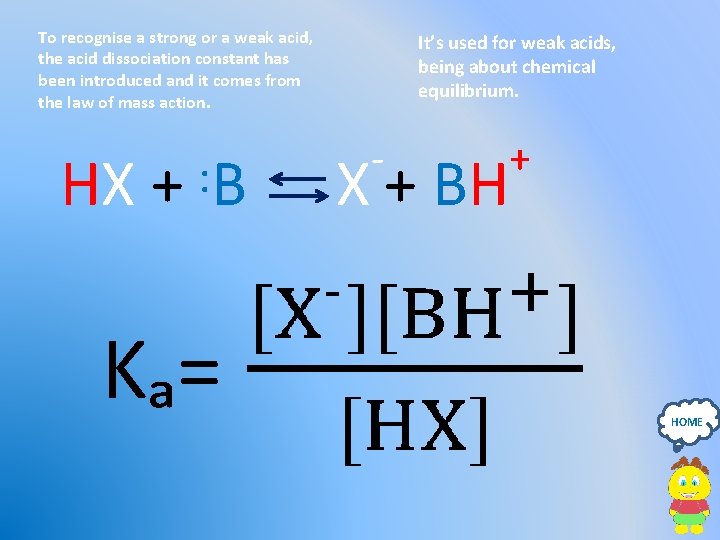



- Slides: 16

ISTITUTO D’ISTRUZIONE SUPERIORE DON LORENZO MILANI CLIL Content and language integrated learning III A L. S. Anno scolastico 2013/2014 Docente Rita Cuomo

ACIDS & BASES Hi guys! My name is Lyecid and I’m gonna help you to discover acids and bases.

Who got interested in acids and bases? What are acids and bases? What is p. H scale?

Acids and bases are both electrolytes in aqueous solution. Electrolytes are substances able to conduct electricity in water. Acids have a sour taste HOME Bases have a bitter taste

AThe lotfirst of people gotwho scientist interested inaacids and worked out theory bases, forand most of time about but acids bases they couldn’t find an was Arrhenius. accurate definition. ARRHENIUS S. A. Arrhenius An acid releases H+ ions in water. A base releases OH- ions in water. ++ - - Me. OH Me(aq)+ + OH HX H X(aq) HH 22 OO HOME

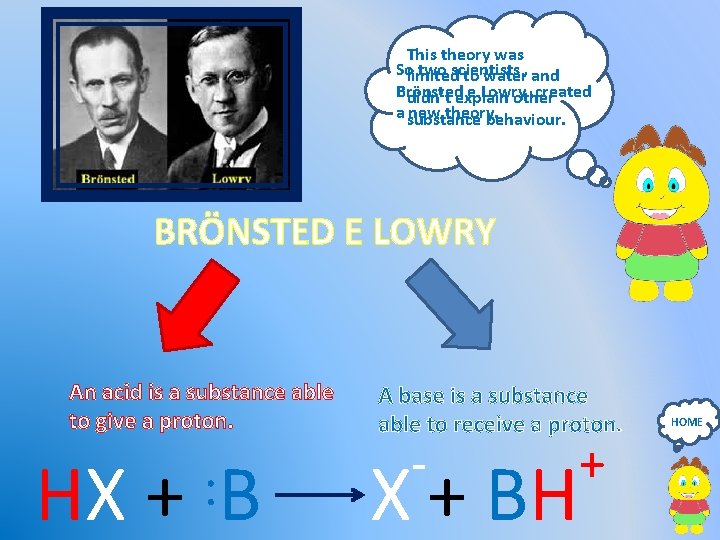
This theory was Solimited two scientists to water, and Brönsted e Lowry , created didn’t explain other a substance new theory. behaviour. BRÖNSTED E LOWRY An acid is a substance able to give a proton. A base is a substance able to receive a proton. - HX + : B X + BH + HOME

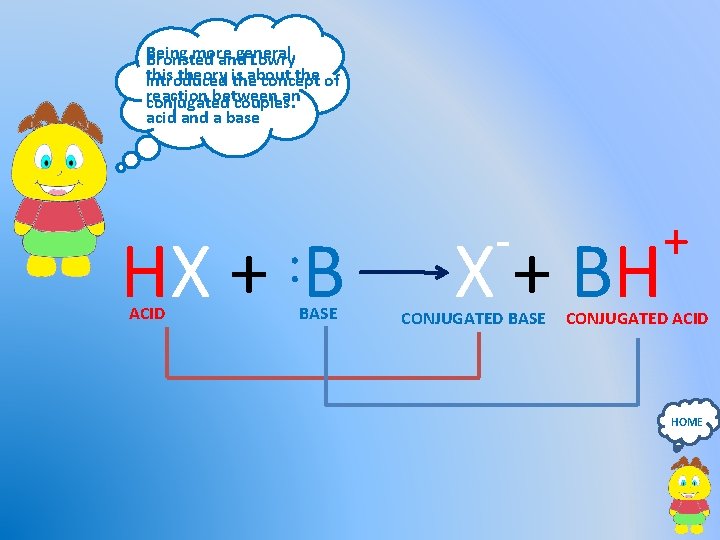
Being more general, Bronsted and Lowry this theory is about the of introduced the concept reaction between an conjugated couples. acid and a base - : HX + BH ACID BASE CONJUGATED BASE + CONJUGATED ACID HOME

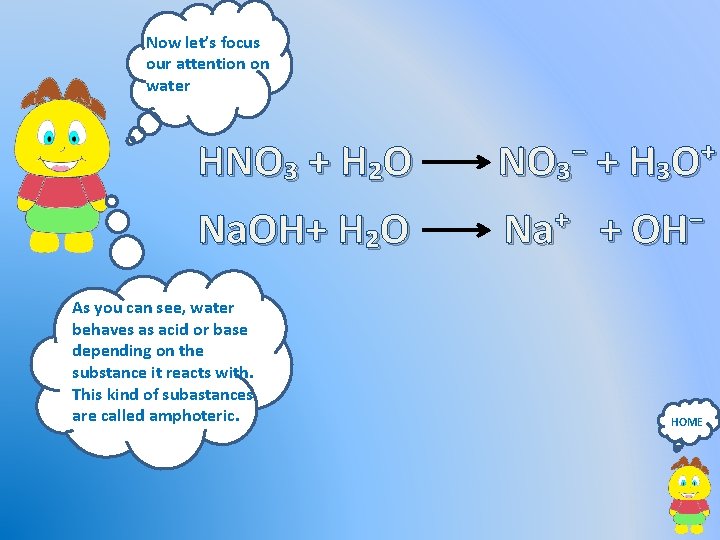
Now let’s focus our attention on water HNO₃ + H₂O NO₃⁻ + H₃O⁺ Na. OH+ H₂O Na⁺ + OH⁻ As you can see, water behaves as acid or base depending on the substance it reacts with. This kind of subastances are called amphoteric. HOME

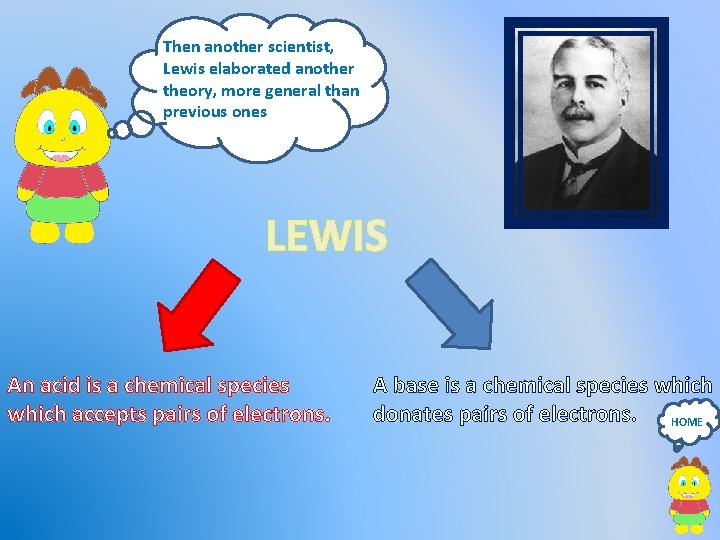
Then another scientist, Lewis elaborated another theory, more general than previous ones LEWIS An acid is a chemical species which accepts pairs of electrons. A base is a chemical species which donates pairs of electrons. HOME

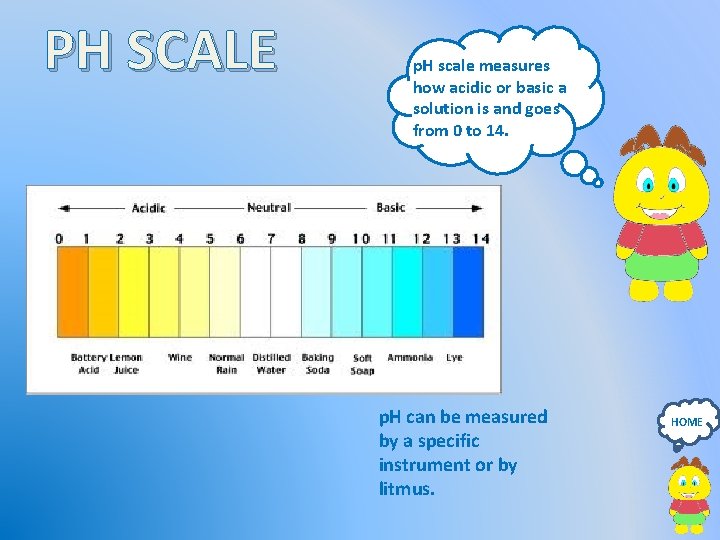
PH SCALE p. H scale measures how acidic or basic a solution is and goes from 0 to 14. p. H can be measured by a specific instrument or by litmus. HOME

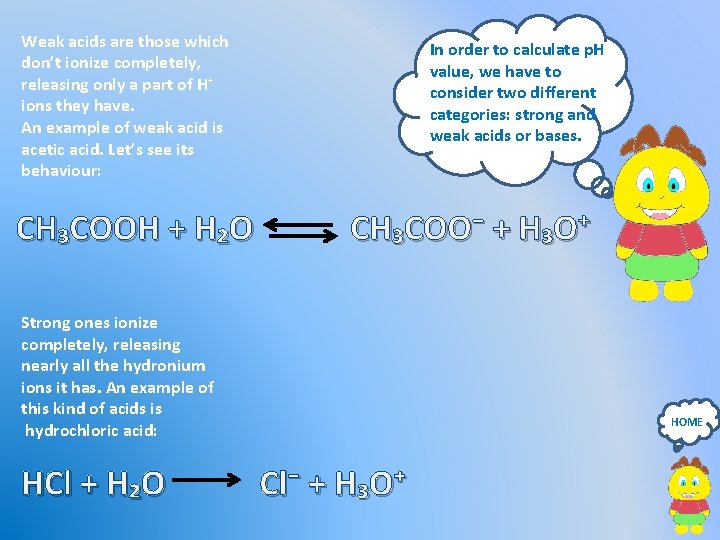
Weak acids are those which don’t ionize completely, releasing only a part of H⁺ ions they have. An example of weak acid is acetic acid. Let’s see its behaviour: CH₃COOH + H₂O In order to calculate p. H value, we have to consider two different categories: strong and weak acids or bases. CH₃COO⁻ + H₃O⁺ Strong ones ionize completely, releasing nearly all the hydronium ions it has. An example of this kind of acids is hydrochloric acid: HCl + H₂O HOME Cl⁻ + H₃O⁺
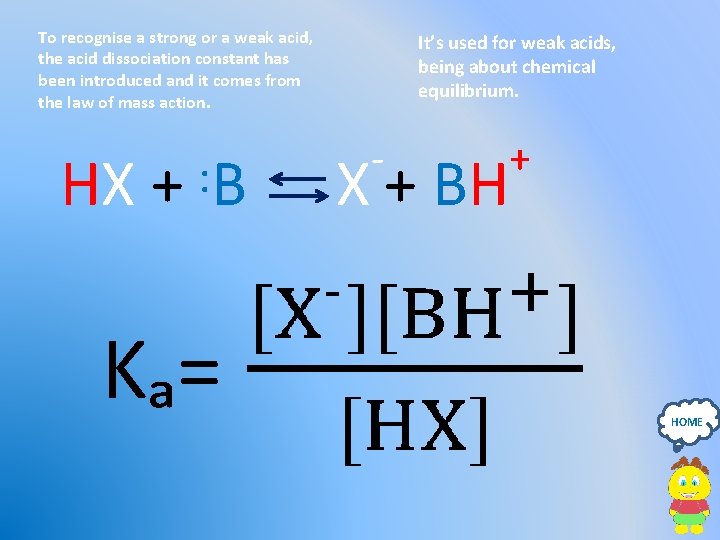
To recognise a strong or a weak acid, the acid dissociation constant has been introduced and it comes from the law of mass action. It’s used for weak acids, being about chemical equilibrium. - HX + : B X + BH Kₐ= + HOME

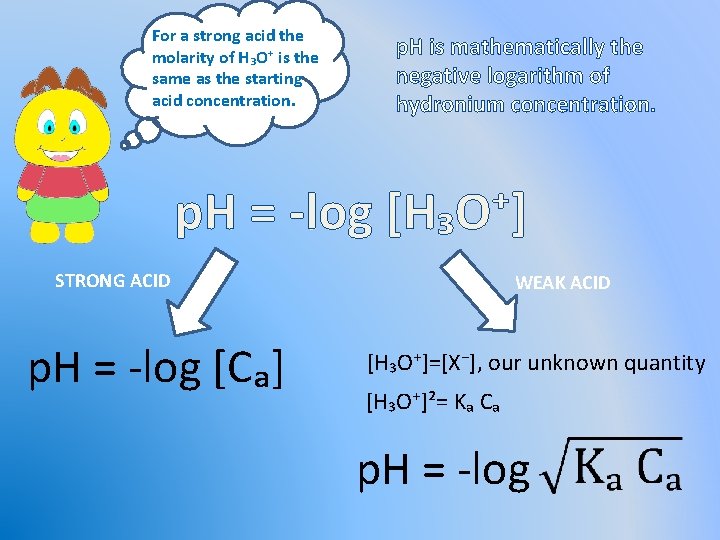
For a strong acid the molarity of H₃O⁺ is the same as the starting acid concentration. p. H is mathematically the negative logarithm of hydronium concentration. p. H = -log [H₃O⁺] STRONG ACID p. H = -log [Cₐ] WEAK ACID [H₃O⁺]=[X⁻], our unknown quantity [H₃O⁺]²= Kₐ Cₐ p. H = -log

AND NOW… IN THE LAB!


Hanno elaborato la seguente presentazione: Alvigi Luigi D’Auria Antonio Gentile Alberto Marinaro Graziano Sorrentino Antonio Todisco Claudia Vitale Daniele