Issues Surrounding Informed Consent to Research with Substance

Issues Surrounding Informed Consent to Research with Substance Abusing Participants Karen L. Dugosh, Ph. D. Treatment Research Institute Philadelphia, PA 3/3/2021 ©Treatment Research Institute, 2012

Overview • This presentation will: • Provide a brief history of informed consent • Review the primary tenets of informed consent • Discuss ways in which the informed consent process may be compromised among substance abusers 3/3/2021 ©Treatment Research Institute, 2012

Introduction • Many important medical and behavioral advancements required research on humans • Frequently conducted with marginal and highly vulnerable populations • Revelations of unethical investigations heightened public awareness of the potential for research misconduct 3/3/2021 ©Treatment Research Institute, 2012

Nazi Medical Experiments (1942 -1945) • Experimental Surgery - Ravensbrook • Sterilization Research - Auschwitz • Typhus Experiments - Buchenwald • Poison Experiments - Buchenwald • Sea Water Experiments - Dachau • High Altitude Experiments - Dachau • Hypothermia Experiments - Dachau 3/3/2021 ©Treatment Research Institute, 2012

Tuskegee Syphilis Experiment (1932 -1972) • Longest non-therapeutic experiment on human beings in medical history • U. S. Public Health Service (PHS) • 399 black men in the late stages of syphilis • Never told what disease they were suffering from or of its seriousness • 28 died directly of syphilis, 100 of complications, 40 wives infected, & 19 of their children born with congenital syphilis 3/3/2021 ©Treatment Research Institute, 2012

Nuremberg Code • Adopted by UN General Assembly in 1948 • First international document to provide guidelines on research ethics • Made voluntary consent a requirement in clinical research studies • Emphasized that consent can be voluntary only if participants: • 1) are able to consent • 2) are free from coercion • 3) comprehend all risks and benefits 3/3/2021 ©Treatment Research Institute, 2012

Helsinki Declaration • Established in 1964 at the 18 th World Medical Assembly in Helsinki, Finland • World Medical Association adopted a set of principles to guide physicians on ethical considerations related to biomedical research • Emphasized the distinction between medical care that directly benefits the patient and research which may have no direct benefit 3/3/2021 ©Treatment Research Institute, 2012

Belmont Report (1979) • National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research • Set forth three principles underlying ethical conduct of research: • Respect for Persons • • Beneficence • • Recognizing autonomy and dignity Protection from harm by maximizing benefits and minimizing risks Justice • Fair distribution of benefits and burdens 3/3/2021 ©Treatment Research Institute, 2012

Basic Principles of Informed Consent • Intelligent • Must be capable of understanding • Knowing • Must be understood and retained • Voluntary • Must be autonomous 3/3/2021 ©Treatment Research Institute, 2012

Vulnerabilities • Intrinsic • Attributes of the individual that limit capacity or freedoms • Extrinsic • Situational factors that may limit capacity or freedoms • Relational • Occur as a result of the a relationship with another individual or group 3/3/2021 ©Treatment Research Institute, 2012

Intelligence • Refers to one’s intrinsic capacity to understand, appreciate, and express a choice • May be compromised in substance abusers due to a host of factors • Severe neurological effects of chronic drug use • Traumatic brain injury 3/3/2021 ©Treatment Research Institute, 2012

Knowingness • Refers to one’s accurate understanding and appreciation of the study and their involvement • Substance abusers may experience impaired attention, cognition, or recall as a result of: • Acute intoxication or withdrawal • Long term effects of drug use on the brain • Developmental and environmental factors • Limited education, poor nutrition, and comorbid health and mental health problems 3/3/2021 ©Treatment Research Institute, 2012

Consent recall • Rounsaville et al. (2008) • Examined consent recall among marijuana users • Presented consent information and tested participants on recall • 55% correctly answered all four multiple choice questions • Kiluk et al. (2010) • Immediate recall related to intelligence, attention, educational attainment 3/3/2021 ©Treatment Research Institute, 2012

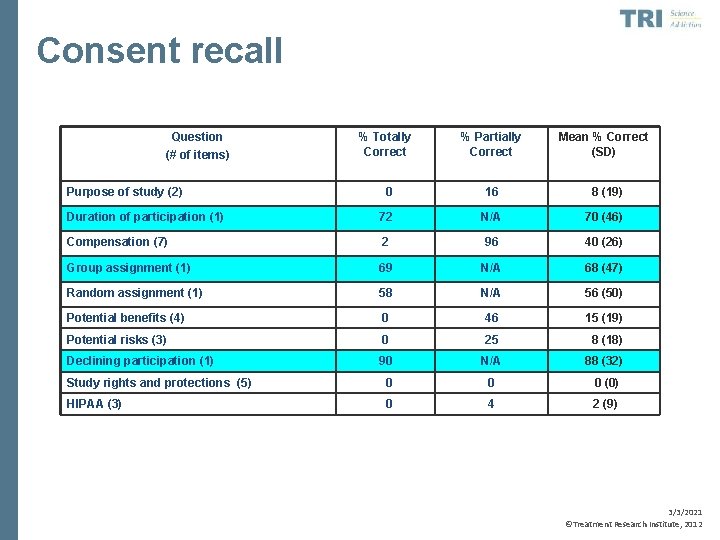
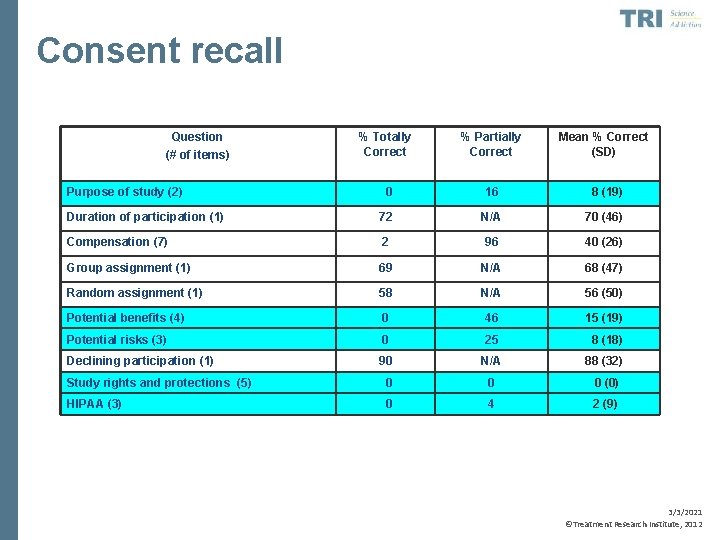
Consent recall • Festinger, Dugosh et al. , 2007 • Examined consent recall among substance abusing offenders • Tested delayed (2 week post-consent) recall of consent information 3/3/2021 ©Treatment Research Institute, 2012
Consent recall Question (# of items) % Totally Correct % Partially Correct Mean % Correct (SD) 0 16 8 (19) Duration of participation (1) 72 N/A 70 (46) Compensation (7) 2 96 40 (26) Group assignment (1) 69 N/A 68 (47) Random assignment (1) 58 N/A 56 (50) Potential benefits (4) 0 46 15 (19) Potential risks (3) 0 25 8 (18) Declining participation (1) 90 N/A 88 (32) Study rights and protections (5) 0 0 0 (0) HIPAA (3) 0 4 2 (9) Purpose of study (2) 3/3/2021 ©Treatment Research Institute, 2012
Consent recall Question (# of items) % Totally Correct % Partially Correct Mean % Correct (SD) 0 16 8 (19) Duration of participation (1) 72 N/A 70 (46) Compensation (7) 2 96 40 (26) Group assignment (1) 69 N/A 68 (47) Random assignment (1) 58 N/A 56 (50) Potential benefits (4) 0 46 15 (19) Potential risks (3) 0 25 8 (18) Declining participation (1) 90 N/A 88 (32) Study rights and protections (5) 0 0 0 (0) HIPAA (3) 0 4 2 (9) Purpose of study (2) 3/3/2021 ©Treatment Research Institute, 2012

Consent recall • Overall, clients recalled only 34% of information on average just two weeks post-consent • Replicated in several subsequent trials (Festinger, Dugosh, et al. , 2009, 2011) • Delayed recall related to intelligence, reading level, memory, and attention 3/3/2021 ©Treatment Research Institute, 2012
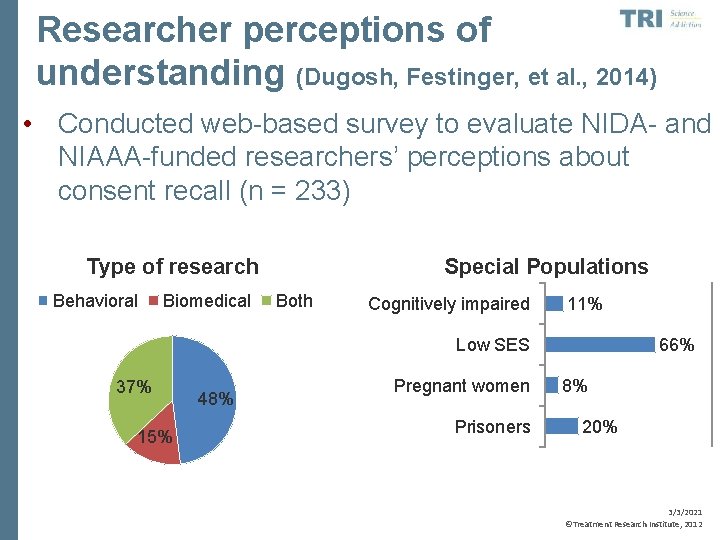
Researcher perceptions of understanding (Dugosh, Festinger, et al. , 2014) • Conducted web-based survey to evaluate NIDA- and NIAAA-funded researchers’ perceptions about consent recall (n = 233) Type of research Behavioral Biomedical Special Populations Both Cognitively impaired 11% Low SES 37% 15% 48% Pregnant women Prisoners 66% 8% 20% 3/3/2021 ©Treatment Research Institute, 2012
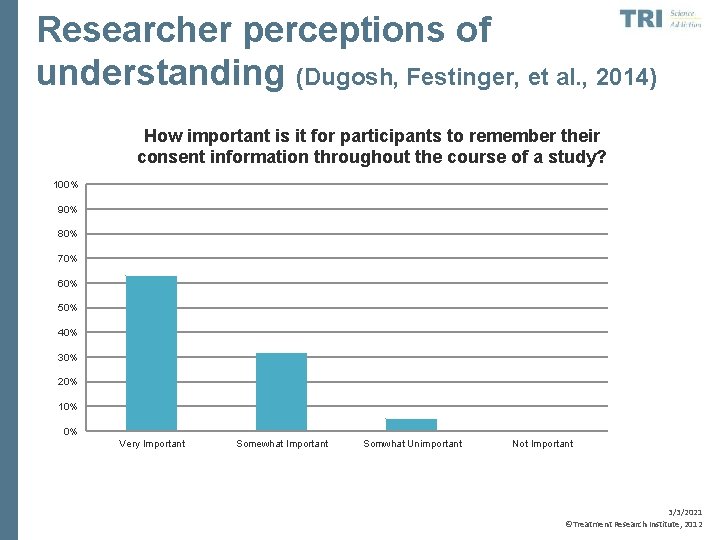
Researcher perceptions of understanding (Dugosh, Festinger, et al. , 2014) How important is it for participants to remember their consent information throughout the course of a study? 100% 90% 80% 70% 60% 50% 40% 30% 20% 10% 0% Very Important Somewhat Important Somwhat Unimportant Not Important 3/3/2021 ©Treatment Research Institute, 2012
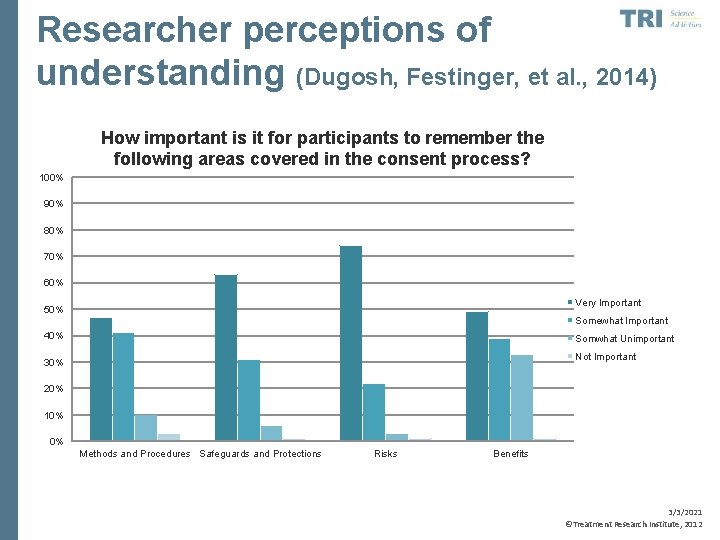
Researcher perceptions of understanding (Dugosh, Festinger, et al. , 2014) How important is it for participants to remember the following areas covered in the consent process? 100% 90% 80% 70% 60% Very Important 50% Somewhat Important 40% Somwhat Unimportant Not Important 30% 20% 10% 0% Methods and Procedures Safeguards and Protections Risks Benefits 3/3/2021 ©Treatment Research Institute, 2012
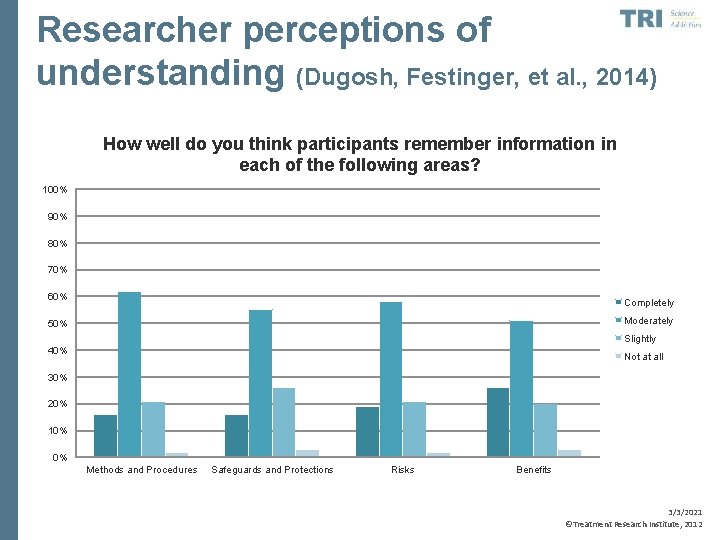
Researcher perceptions of understanding (Dugosh, Festinger, et al. , 2014) How well do you think participants remember information in each of the following areas? 100% 90% 80% 70% 60% Completely Moderately 50% Slightly 40% Not at all 30% 20% 10% 0% Methods and Procedures Safeguards and Protections Risks Benefits 3/3/2021 ©Treatment Research Institute, 2012

Voluntariness • Participation free from coercion and undue influence • Substance abusers often have certain situational factors that may interfere with their ability to make autonomous decisions • Often recruited from settings that are implicitly coercive (e. g. , inpatient units, detoxification facilities, prisons) • May perceive, correctly or incorrectly, that cooperation is essential for their well being 3/3/2021 ©Treatment Research Institute, 2012
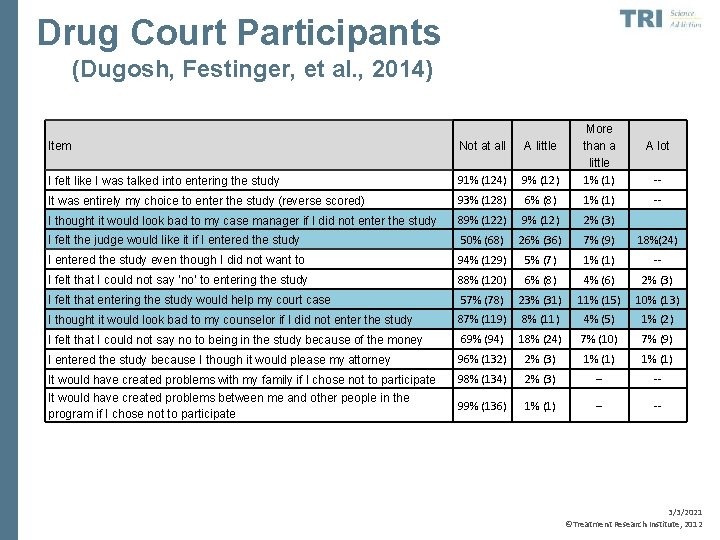
Drug Court Participants (Dugosh, Festinger, et al. , 2014) Item Not at all A little I felt like I was talked into entering the study 91% (124) 9% (12) More than a little 1% (1) It was entirely my choice to enter the study (reverse scored) 93% (128) 6% (8) 1% (1) -- I thought it would look bad to my case manager if I did not enter the study 89% (122) 9% (12) 2% (3) I felt the judge would like it if I entered the study 50% (68) 26% (36) 7% (9) 18%(24) I entered the study even though I did not want to 94% (129) 5% (7) 1% (1) -- I felt that I could not say ‘no’ to entering the study 88% (120) 6% (8) 4% (6) 2% (3) I felt that entering the study would help my court case 57% (78) 23% (31) 11% (15) 10% (13) I thought it would look bad to my counselor if I did not enter the study 87% (119) 8% (11) 4% (5) 1% (2) I felt that I could not say no to being in the study because of the money 69% (94) 18% (24) 7% (10) 7% (9) I entered the study because I though it would please my attorney 96% (132) 2% (3) 1% (1) It would have created problems with my family if I chose not to participate 98% (134) 2% (3) -- -- It would have created problems between me and other people in the program if I chose not to participate 99% (136) 1% (1) -- -- A lot -- 3/3/2021 ©Treatment Research Institute, 2012
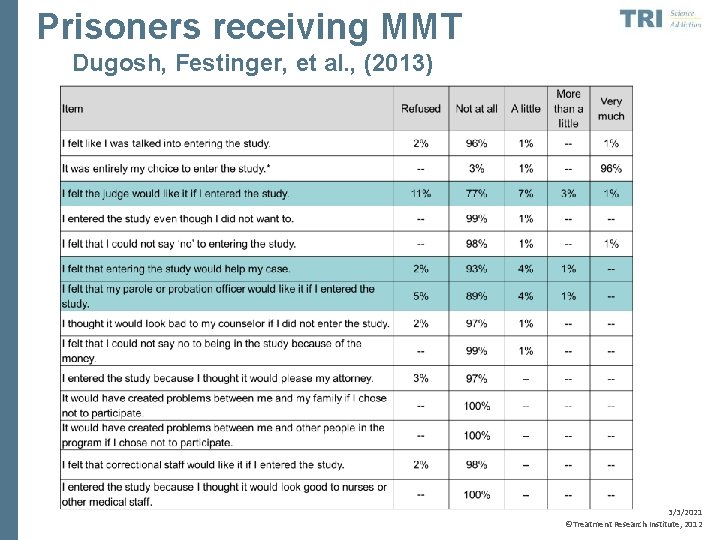
Prisoners receiving MMT Dugosh, Festinger, et al. , (2013) 3/3/2021 ©Treatment Research Institute, 2012

Voluntariness and payment • Widely held belief that providing monetary incentives to substance abusers is an undue influence • Lower SES, lower educational attainment • Address this by providing gift card payments, nonmonetary goods and services 3/3/2021 ©Treatment Research Institute, 2012

Conclusions • Substance abusers present unique challenges related to informed consent to research • Problems may be magnified in genetics research • Fortunately, there are empirically-validated techniques and tools available to help improve informed consent among substance using participants 3/3/2021 ©Treatment Research Institute, 2012
- Slides: 26