ISOTOPES NUCLIDES A protons neutrons nucleons alpha beta

ISOTOPES, NUCLIDES A protons, neutrons, nucleons, alpha, beta, positron, gamma, Z E n+ p n protons and neutrons

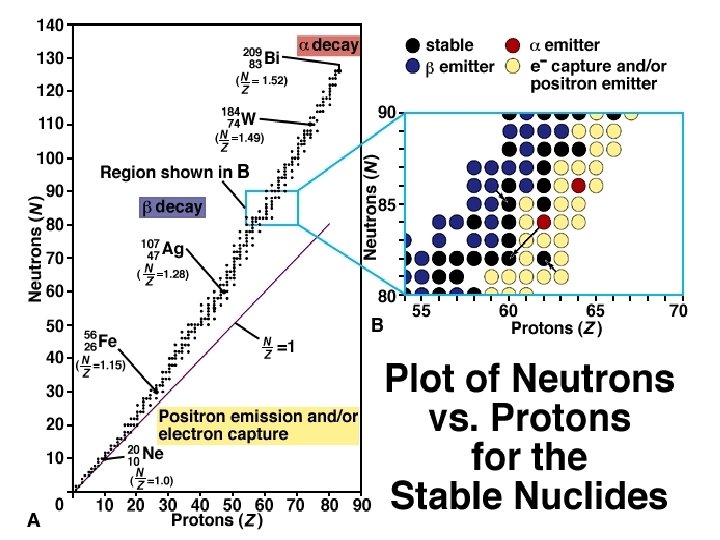
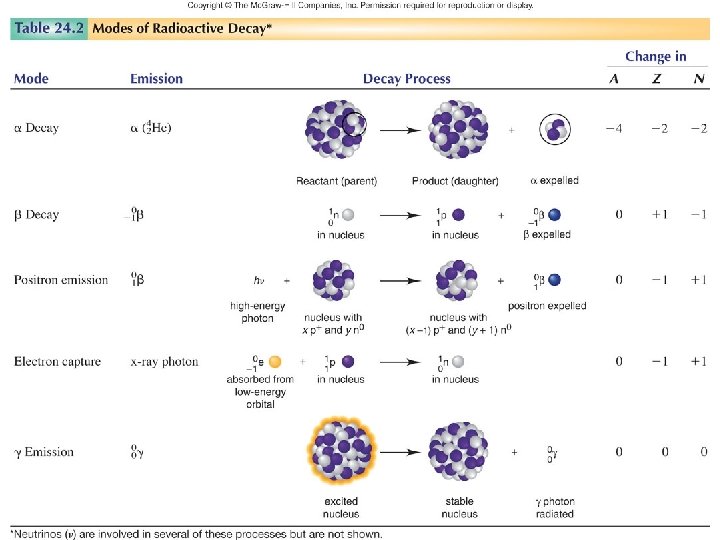
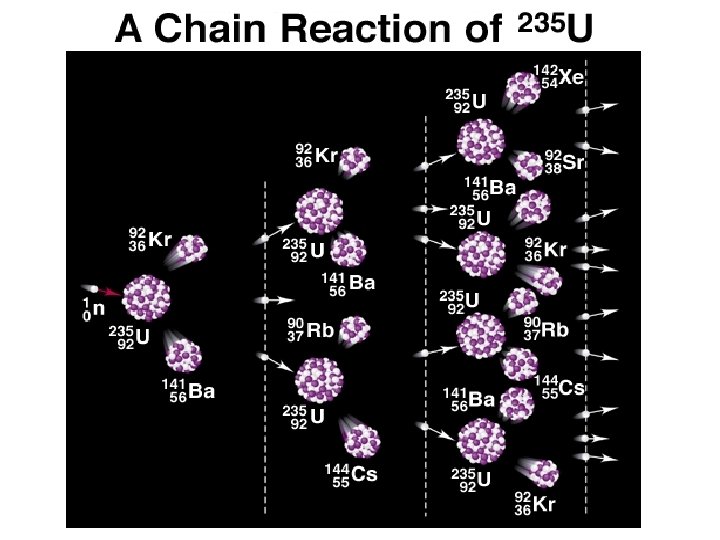
NUCLEAR STABILITY Modes of Radioactive Decay Alpha decay–heavy isotopes: 42 He or Beta decay–neutron rich isotopes: e- or Positron emission–proton rich isotopes: Electron capture–proton rich isotopes: x-rays Gamma-ray emission( – Decay of nuclear excited states • Spontaneous fission– very heavy isotopes • • •

Natural Radioactive Decay Processes Reason for Nuclear Instability Excess Mass N/Z too high N/Z too low Energetically Radioactive Emitted Process Radiation decay 4 Nuclear Change Loss of 2 protons and 2 neutrons occurs 0 - decay -1 A neutron is converted into a proton and an electron. 0 + decay +1 a proton is converted into a neutron and a positron. Electron Neutrino A proton combines with capture an inner-shell electron to become a neutron. emission Gamma ray Loss of excess nuclear energy occurs. 2 Change in N/Z Ratio Slight increase Decrease Increase None

Natural Decay Series for Uranium-238 238 U 234 Th 234 Pa 234 U 230 Th 226 Ra 222 Rn 218 Po 218 At 214 Bi 214 Po = decay = decay 238 U: 214 Pb 210 Tl 210 Pb 210 Bi 210 Po 206 Pb 8 decays and 6 decays leaves you with 206 Pb 206 Hg 206 Tl
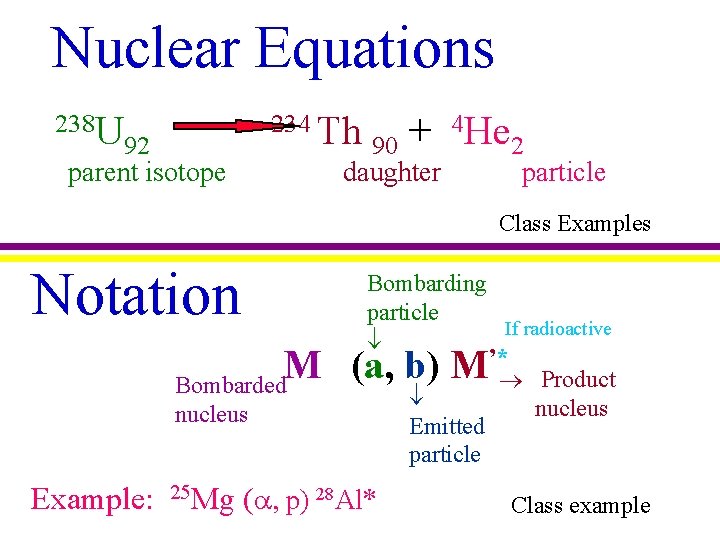
Nuclear Equations 238 U 234 Th 92 parent isotope 4 He + 90 2 daughter particle Class Examples Notation Bombarding particle If radioactive ’* M (a, b) M Bombarded nucleus Example: 25 Mg ( , p) 28 Al* Emitted particle Product nucleus Class example
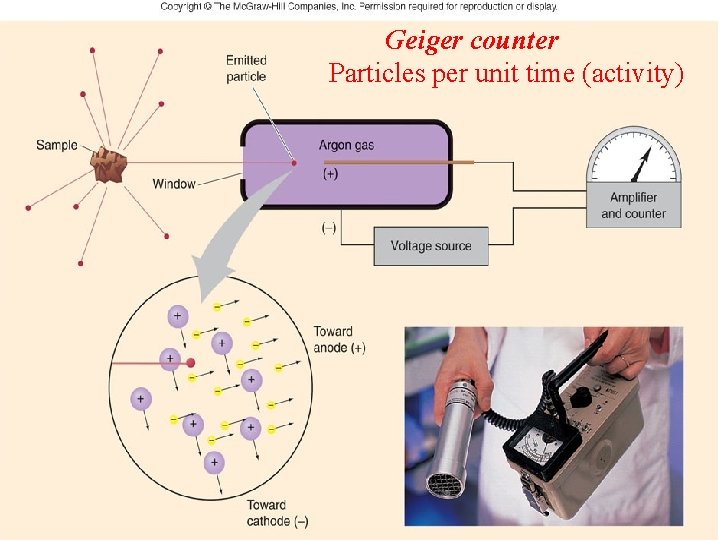
Geiger counter Particles per unit time (activity)

Rate of Radioactive Decay Rate independent of temperature implies Ea = 0 EXPLAIN? Draw diagram First Order Reactions: A B rate law = ? Conc. - time relationship? Half- life ?
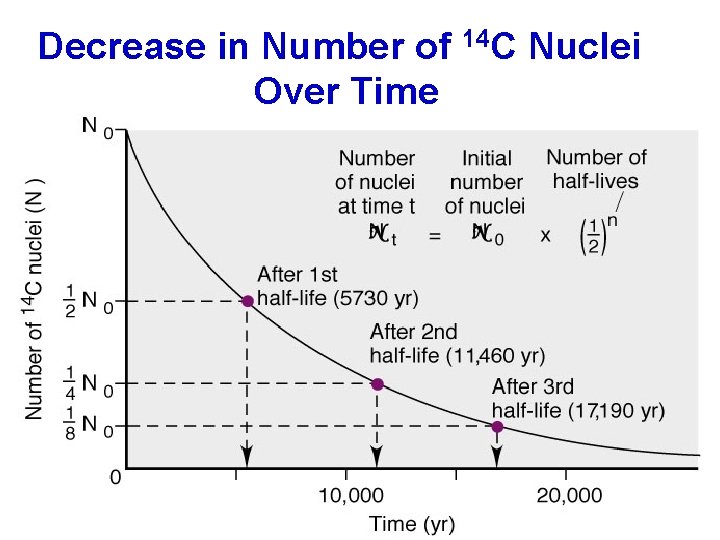
Decrease in Number of 14 C Nuclei Over Time
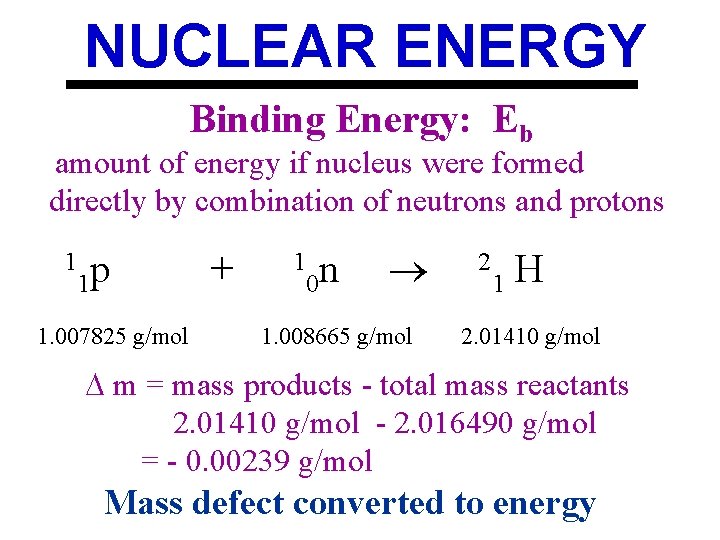
NUCLEAR ENERGY Binding Energy: Eb amount of energy if nucleus were formed directly by combination of neutrons and protons 1 p 1 1. 007825 g/mol + 1 0 n 1. 008665 g/mol 2 1 H 2. 01410 g/mol m = mass products - total mass reactants 2. 01410 g/mol - 2. 016490 g/mol = - 0. 00239 g/mol Mass defect converted to energy

Mass Energy EINSTEIN’S EQUATION FOR THE CONVERSION OF MASS INTO ENERGY E = mc 2 m = mass (kg) c = Speed of light 8 = 2. 998 x 10 m/s E = (-2. 39 x 10 -6 Kg) (2. 998 x 108 m/s)2 = - 2. 15 x 1011 J = - 2. 15 x 108 k. J Class problem

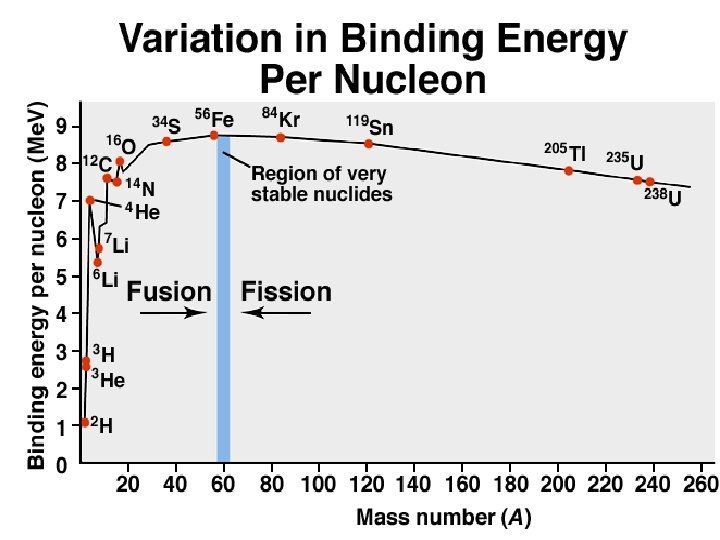
Sample Problem 24. 6 Calculating the Binding Energy per Nucleon PROBLEM: Iron-56 is an extremely stable nuclide. Compute the binding energy per nucleon for 56 Fe and compare it with that for 12 C (mass of 56 Fe atom = 55. 934939 amu; mass of 1 H atom = 1. 007825 amu; mass of neutron = 1. 008665 amu). PLAN: Find the mass defect, m; multiply that by the Me. V equivalent and divide by the number of nucleons. SOLUTION: Mass Defect = [(26 x 1. 007825 amu) + (30 x 1. 008665 amu)] - 55. 934939 m = 0. 52846 amu Binding energy = 12 C (0. 52846 amu)(931. 5 Me. V/amu) 56 nucleons = 8. 790 Mev/nucleon has a binding energy of 7. 680 Me. V/nucleon, so 56 Fe is more stable.

Units of Radiation Dose rad = Radiation-absorbed dose The quantity of energy absorbed per kilogram of tissue: 1 rad = 1 x 10 -2 J/kg rem = Roentgen equivalent for man The unit of radiation dose for a human: 1 rem = 1 rad x RBE = 10 for RBE = 1 for x-rays, and ’s
- Slides: 16