Isotopes Ions and Bohr Model Pick up the

Isotopes, Ions, and Bohr Model Pick up the following materials: Warm-up Notes Build an atom activity Homework Take out: Notecards if finished Important dates: 9/14 – quiz on big bang, subatomic particles, bohr model, isotopes, ions, equipment activity 9/18 – half cards due 9/21 – all cards due 9/21 – quiz on elements Warm up: How many protons, neutrons, and electrons does Fluorine have with a mass of 21? P+ = 9 e- = 9 NO = 12 (21 -9 = 12)

ISOTOPES l. Atoms of the same element that differ in mass. l(They have the same # of + p , but different # of N°)
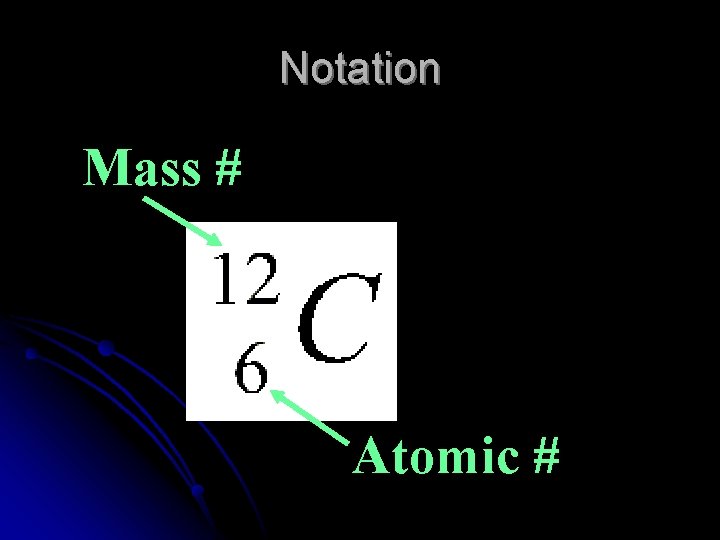
Notation Mass # Atomic #

Notation l. Can also be written as ELEMENT – MASS # l. Example: Carbon - 12

IONS l Charged particles that are created by a loss or gain of electrons. l For example: contains: 20 protons 20 neutrons 18 electrons

Using the Periodic Table l The charges of many common ions can be determined by their placement on the 0 periodic table. +1 +2 +3 ± 4 -3 -2 -1

Try This How many no, p+ and e- are in a nitrogen 15 ion? l How many no, p+ and e- in ? l 7 p+; 8 no; 10 e 16 p+; 18 no; 18 e-
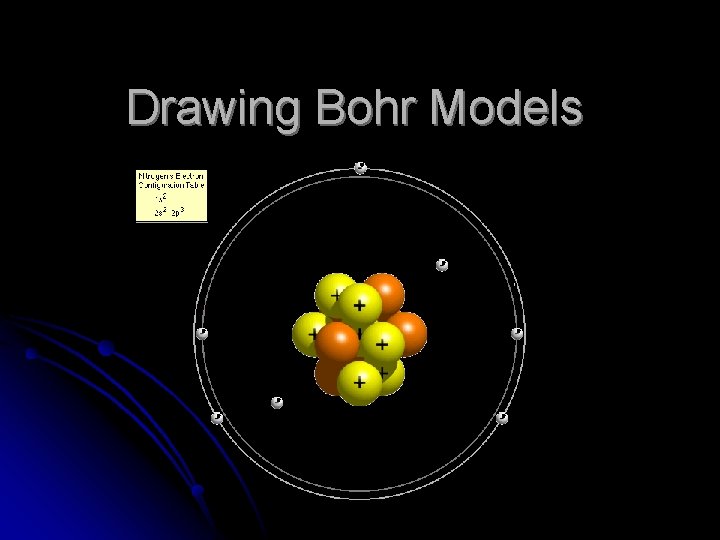
Drawing Bohr Models

Bohr Models 1. 2. 3. Bohr models are used to predict reactivity in elements. Reactivity refers to how likely an element is to form a compound with another element. When looking at Bohr models, we look at its valence electrons (the electrons on the last energy level) to determine reactivity.

Drawing Bohr Models 1. 2. 3. 4. 5. Draw the nucleus. Write the number of neutrons and the number of protons in the nucleus. Draw the first energy level. Draw the electrons in the energy levels according to the rules below. Make sure you draw the electrons in pairs. Keep track of how many electrons are put in each level and the number of electrons left to use.

Rules for Energy Levels 1. 2. 3. 4. Level 1 (closest to the nucleus) can hold a maximum of 2 e. Level 2 can hold a max of 8 e. Level 3 can hold a max of 18 e. Level 4 can hold a max of 32 e. You must fill one level before going on to draw the next level!


Guided Practice In order to draw Bohr models of these elements, you must first determine the number of protons, neutrons, and electrons. Once you have found this information, follow the directions to draw your model. 6 C Carbon 12. 011 6 6 6 Protons: _____ Neutrons: _____ Electrons: ______ 2 How many energy shells will this have? ____ 4 How many valence (outer) electrons does this element have? ____ Bohr Model:

Guided Practice 16 S Sulfur 32. 066 16 16 16 Protons: _____ Neutrons: _____ Electrons: ______ 3 How many energy shells will this have? ____ 6 How many valence (outer) electrons does this element have? ____ Bohr Model:

Guided Practice 3 Li Lithium 6. 941 3 4 3 Protons: _____ Neutrons: _____ Electrons: ______ 2 How many energy shells will this have? ____ 1 How many valence (outer) electrons does this element have? ____ Bohr Model:

Guided Practice 10 Ne Neon 20. 180 10 10 10 Protons: _____ Neutrons: _____ Electrons: ______ 2 How many energy shells will this have? ____ 8 How many valence (outer) electrons does this element have? ____ Bohr Model:

Guided Practice 15 P Phosphorus 30. 974 15 16 15 Protons: _____ Neutrons: _____ Electrons: ______ 3 How many energy shells will this have? ____ 5 How many valence (outer) electrons does this element have? ____ Bohr Model:

Guided Practice 11 Na Sodium 22. 990 11 12 11 Protons: _____ Neutrons: _____ Electrons: ______ 3 How many energy shells will this have? ____ 1 How many valence (outer) electrons does this element have? ____ Bohr Model:
- Slides: 18