Isotopes and Relative Atomic Mass Key thing to







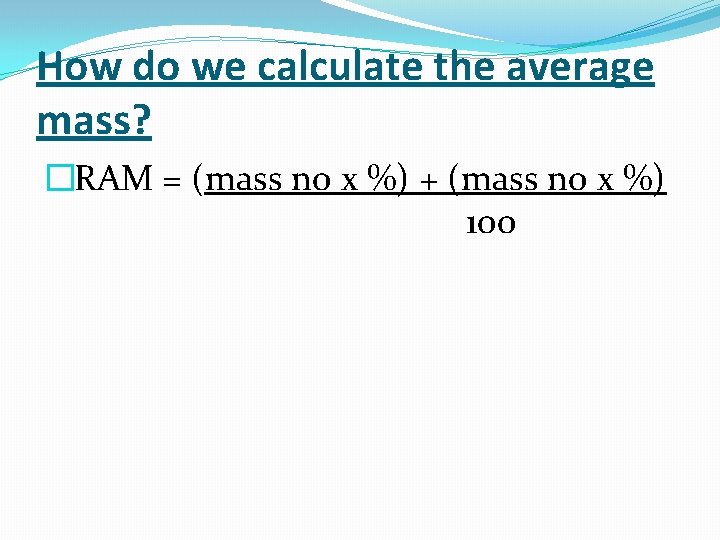







- Slides: 20

Isotopes and Relative Atomic Mass Key thing to remember: The proton number is what defines an element, if that changes it becomes a different element!

Relative Atomic Mass �Mass of atoms is incredibly small! So we have to use relative atomic mass. �The symbol for relative atomic mass is Ar �Carbon- 12 atom contains 6 protons, 6 neutrons and 6 elections. Its relative atomic mass is defined as exactly 12. �Every other atom is compared to this. �E. g Helium is one 3 rd of a carbon atom. So its relative atomic mass is 4.

Defining Relative atomic mass � The mean mass of an atom relative to the mass of an atom of carbon-12, which is assigned a mass of 12.

Isotopes �Isotopes are atoms of the same element with different numbers of neutrons. �That is; � Same Atomic Number � Same no. of protons � Different Mass Number � Different no. of neutrons

But if the neutron number has changed, is it a different element? NO!! It is the proton number that defines an element, if the neutron number has changed it is the same atom, just a different version (isotope).

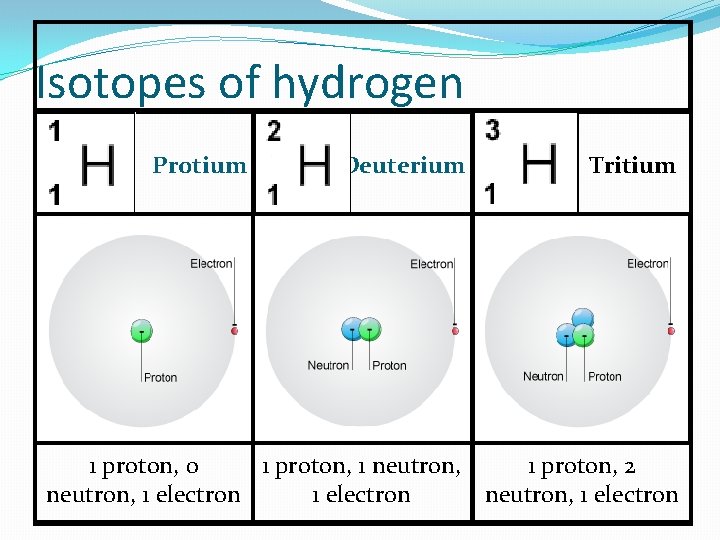
Isotopes of hydrogen Isotope Protium Deuterium Tritium Symbol P N E Uses 1 0 1 Used as fuel, for hardening margarine. 1 Used in heavy or deuterated water, solvent 1 Radioactive, Used in H-bombs, glow in the dark paints 1 1 1 2

Isotopes of hydrogen Protium Deuterium Tritium 1 proton, 0 1 proton, 1 neutron, 1 proton, 2 neutron, 1 electron

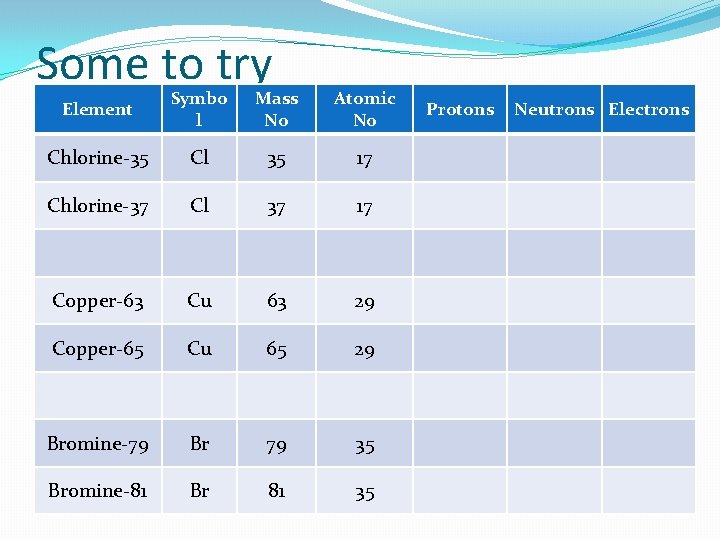
Some to try Element Symbo l Mass No Atomic No Chlorine-35 Cl 35 17 Chlorine-37 Cl 37 17 Copper-63 Cu 63 29 Copper-65 Cu 65 29 Bromine-79 Br 79 35 Bromine-81 Br 81 35 Protons Neutrons Electrons

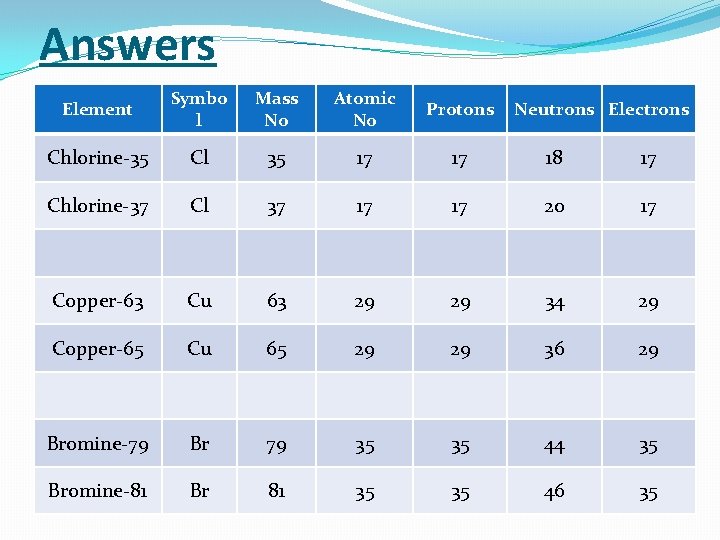
Answers Element Symbo l Mass No Atomic No Protons Chlorine-35 Cl 35 17 17 18 17 Chlorine-37 Cl 37 17 17 20 17 Copper-63 Cu 63 29 29 34 29 Copper-65 Cu 65 29 29 36 29 Bromine-79 Br 79 35 35 44 35 Bromine-81 Br 81 35 35 46 35 Neutrons Electrons

So if there’s different isotopes has atoms, then what is the actual mass of an atom? We have to calculate an average To do this we need a few pieces of information. �The atomic mass of the isotope �Relative abundance of the atom (this means how much there is of each isotope compared to the total in the world)
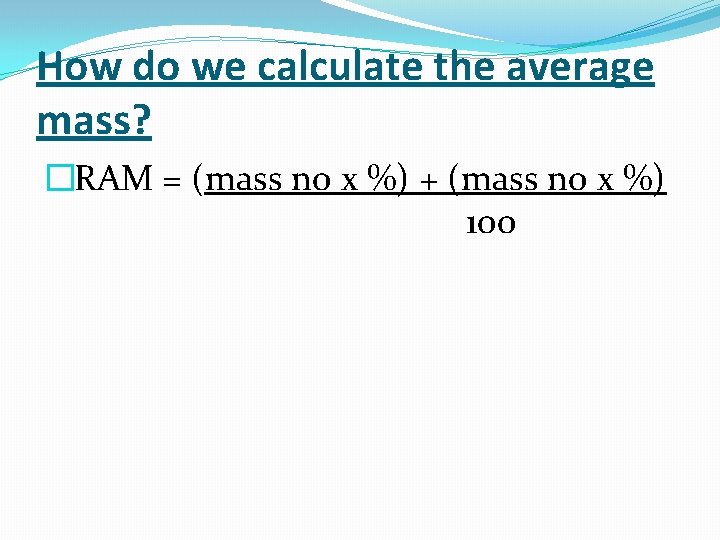
How do we calculate the average mass? �RAM = (mass no x %) + (mass no x %) 100

Lets try Chlorine and Copper �Chlorine has two isotopes �Copper has two main isotopes Chlorine 35 75% 37 25% Copper 63 69% 65 31%

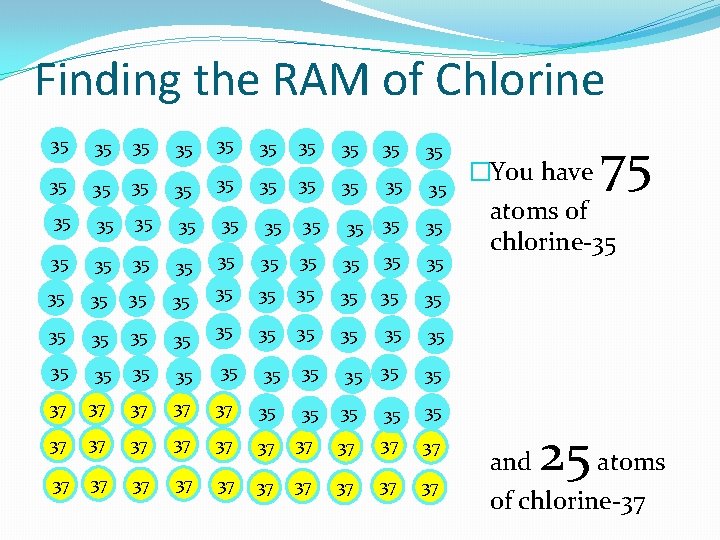
Finding the RAM of Chlorine 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 37 37 37 37 37 37 37 �If you assume you have 100 atoms then the average is easier to find.

Finding the RAM of Chlorine 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 37 37 37 37 37 37 37 �You have 75 atoms of chlorine-35 25 and atoms of chlorine-37

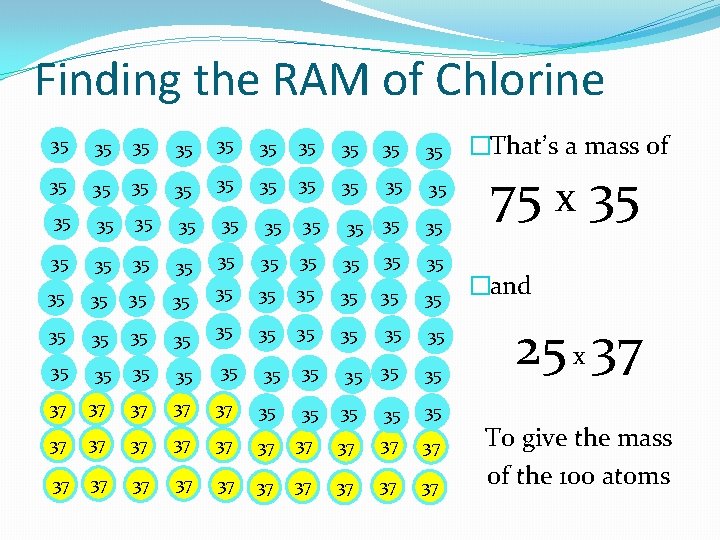
Finding the RAM of Chlorine 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 37 37 37 37 37 37 37 �That’s a mass of 75 x 35 �and 25 x 37 To give the mass of the 100 atoms

Using Weighted Averages 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 37 37 37 37 37 37 37 �So the average mass of a chlorine atom isn’t (35+37)/2 �Its � (35 x 75) + (37 x 25) � 100

From Weighted Averages to RAM �Weighted = (mass x number) + (mass x number) ave total number �So RAM = (35 x 75) + (37 x 25) 100 � RAM = (2625 + 925)/100 � RAM = 35. 5

Weighted Averages �Use �RAM = (mass no x %) + (mass no x %) 100 �To work out the RAM of Copper

Now you try copper �Chlorine has two isotopes �Copper has two main isotopes Chlorine 35 75% 37 25% Copper 63 69% 65 31%

RAM of copper � RAM = (mass no x %) + (mass no x %) 100 �So RAM = (63 x 69) + (65 x 31) 100 � RAM = (4347 + 2015)/100 � RAM = 63. 6