Isotonic solutions if the osmotic concentrations of both
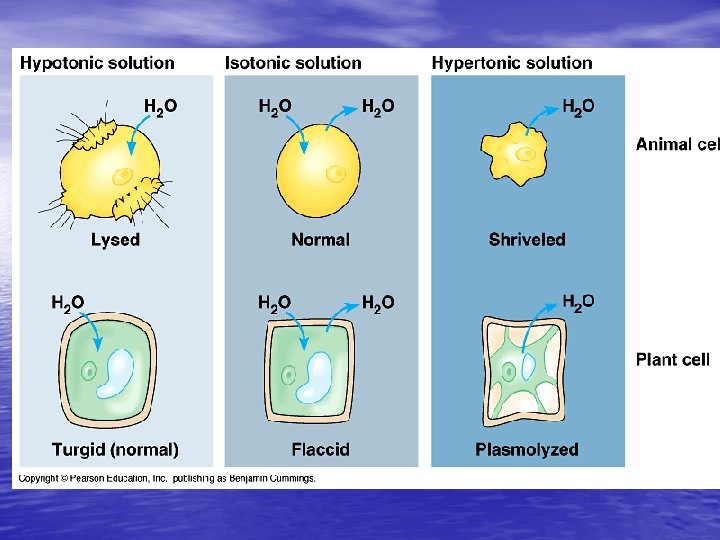
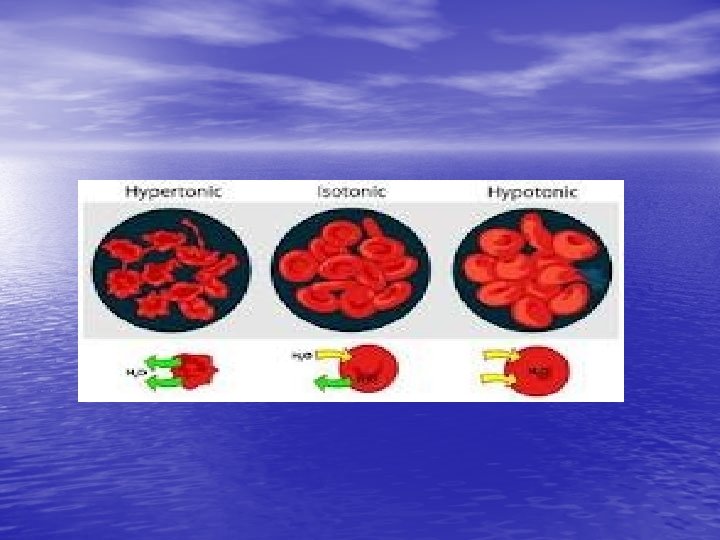

Isotonic solutions: • if the osmotic concentrations of both solutions are equal than no net movement of water occurs • Examples include most interstitial fluid and most intravenous solutions

• Hypertonic solution: the solution has more dissolved particles, salts, sugar, etc. , than is in the cells, so cells will shrivel up or shrink as water moves out of the cell

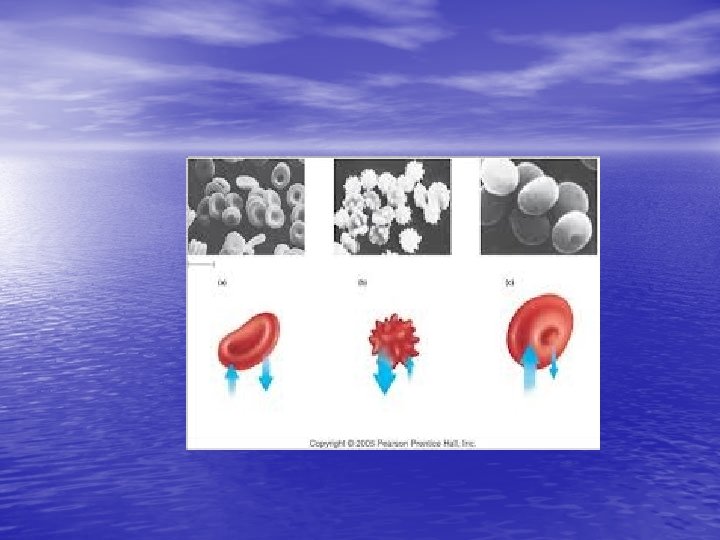
• If red blood cells are exposed to a hypertonic solution- a solution that contains more solutes, or dissolved substances, than there are inside cells- the cells will shrink, or crenate as water moves outside the cell (this is because water is in higher concentration inside the cell than outside, so it follows its concentration gradient and leaves the cell)
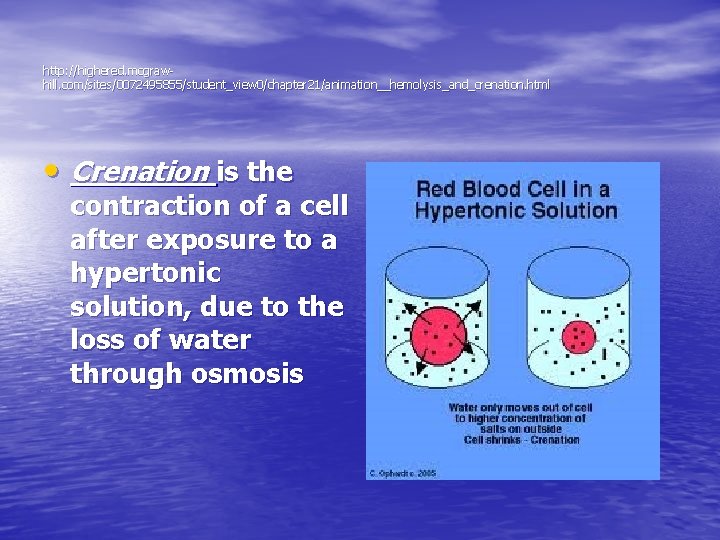
http: //highered. mcgrawhill. com/sites/0072495855/student_view 0/chapter 21/animation__hemolysis_and_crenation. html • Crenation is the contraction of a cell after exposure to a hypertonic solution, due to the loss of water through osmosis
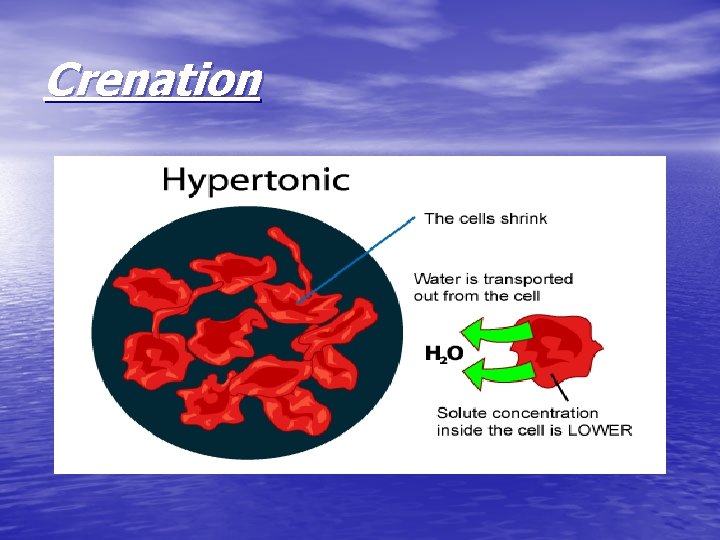
Crenation
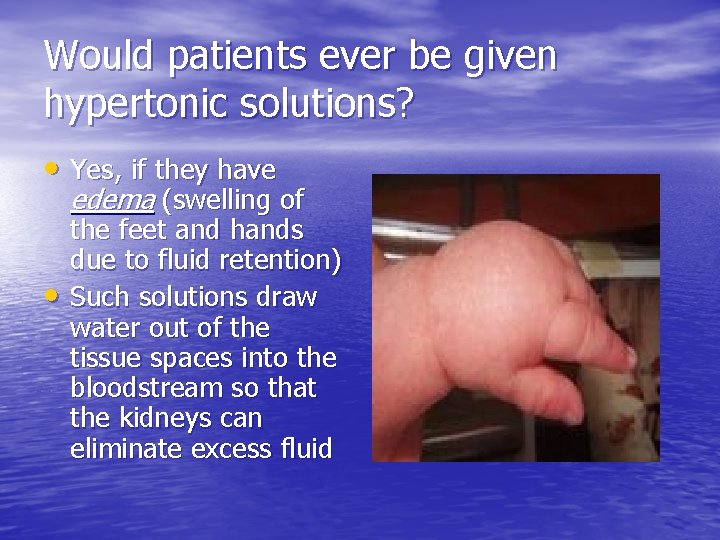
Would patients ever be given hypertonic solutions? • Yes, if they have edema (swelling of • the feet and hands due to fluid retention) Such solutions draw water out of the tissue spaces into the bloodstream so that the kidneys can eliminate excess fluid
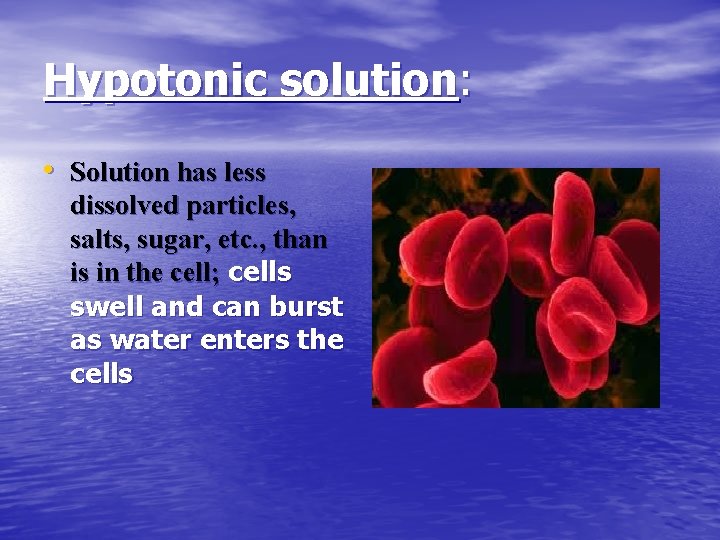
Hypotonic solution: • Solution has less dissolved particles, salts, sugar, etc. , than is in the cell; cells swell and can burst as water enters the cells

Hypotonic solution: • Distilled water is an • example of this type of solution (there are no solutes). Water will enter cells until they finally burst, or lyse. Given intravenously to rehydrate tissues of extremely dehydrated patients
- Slides: 11