Isotonic Solutions 342021 BAFPUJD Osmosis If a pure

Isotonic Solutions 3/4/2021 BA-FP-UJ-D
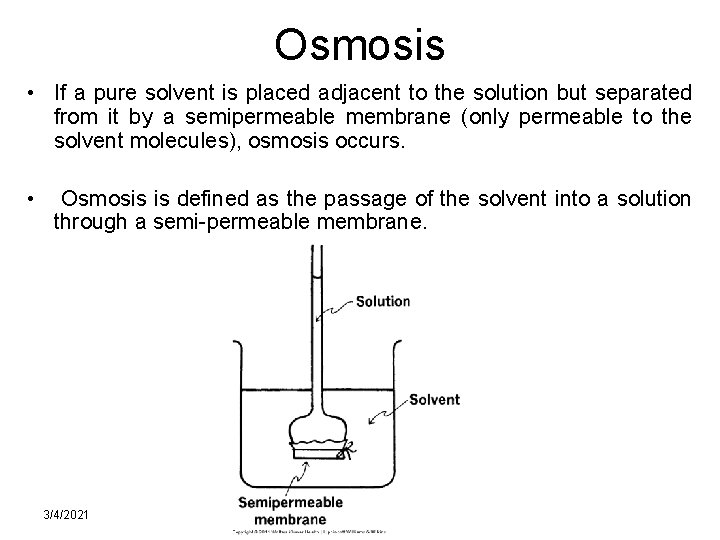
Osmosis • If a pure solvent is placed adjacent to the solution but separated from it by a semipermeable membrane (only permeable to the solvent molecules), osmosis occurs. • Osmosis is defined as the passage of the solvent into a solution through a semi-permeable membrane. 3/4/2021 BA-FP-UJ-D

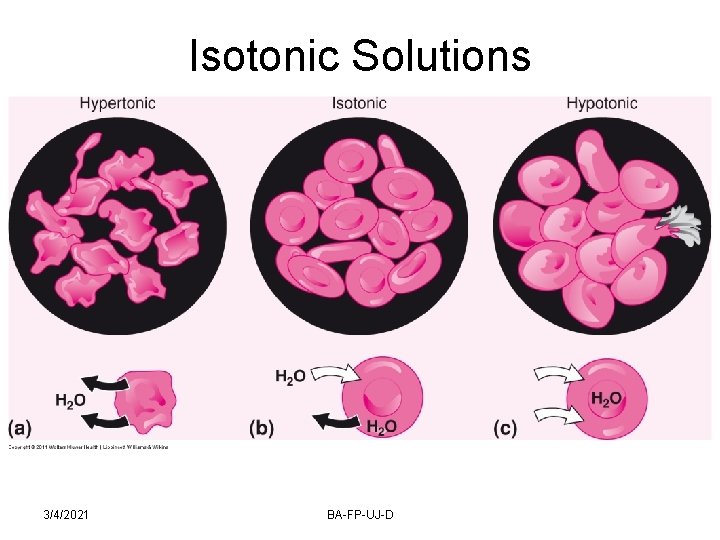
Isotonic Solutions ISOTONIC solutions cause no swelling or contraction of the tissues with which they come in contact, and produce no discomfort when instilled in the eye, nasal tract, blood, or other body tissues. Outward passage of water causes the cell to shrink and become WRINKLED or CRENATED. The salt solution is said to be HYPERTONIC. When water enters the blood cells, it causes them to swell and finally burst, and hemoglobin liberated, this is called HEMOLYSIS, solution is HYPOTONIC. 3/4/2021 BA-FP-UJ-D
Isotonic Solutions 3/4/2021 BA-FP-UJ-D

Measurement of Tonicity: • It is now well established that -0. 52 is the freezing point of both human blood and lacrimal fluid. • This temperature corresponds to the freezing point of a 0. 9% Na. Cl (0. 154 M) solution, which is therefore considered to be isotonic with both blood and lacrimal fluid. 3/4/2021 BA-FP-UJ-D

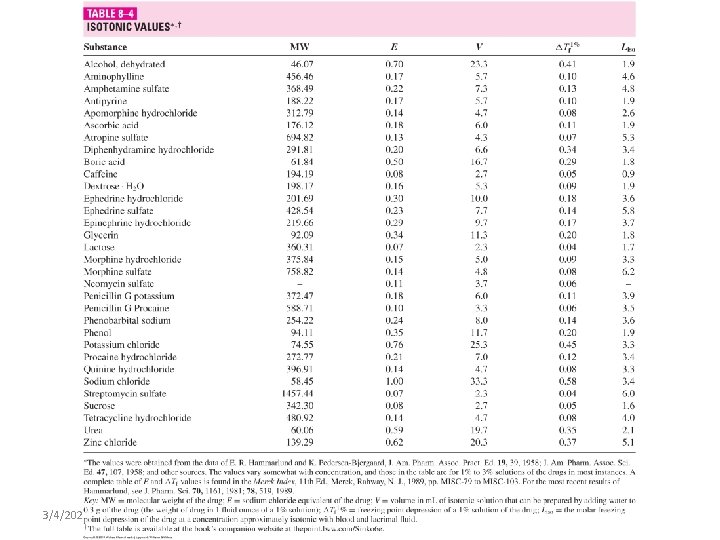
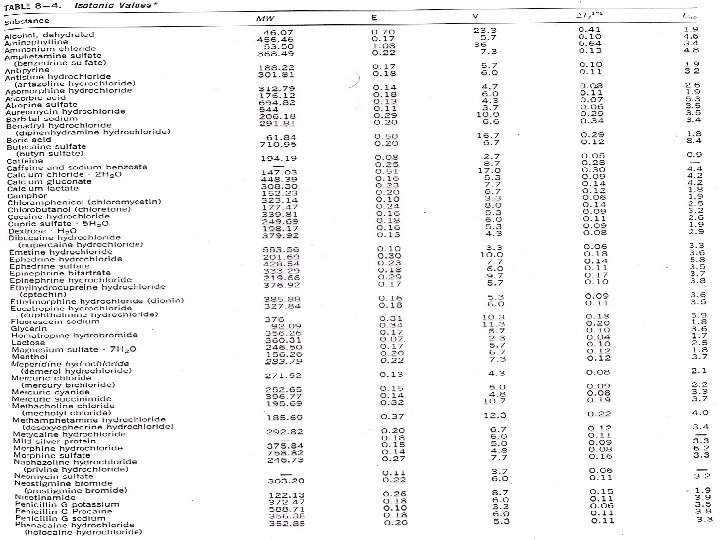
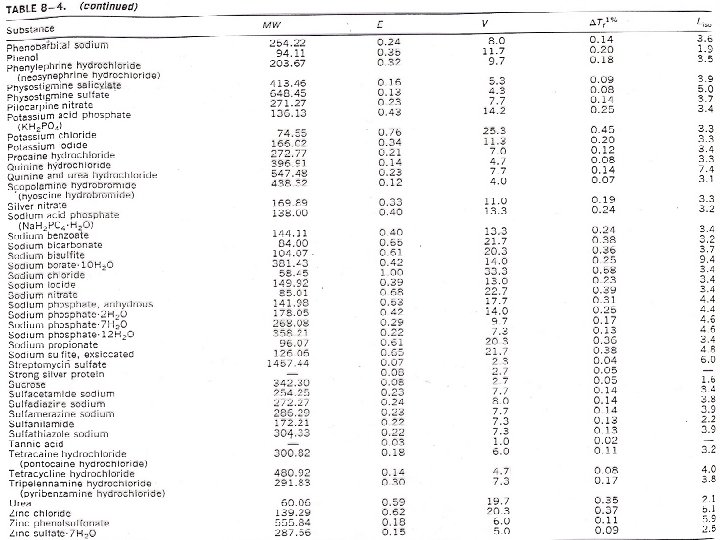
Calculating Tonicity Using Liso Values: A new factor is introduced for the freezing point depression of electrolytes which is ( L = i Kf ) Tf = i Kf m Tf = Lc where: L = i Kf and c = molar concentration Specific Liso value is obtained from the freezing point lowering of solutions at representative compounds of a given ionic type at a concentration (c) that is isotonic with body fluids. Liso: L value for the concentration which is isotonic with body fluids.

Calculating Tonicity Using Liso Values: e. g. : Calculate the Liso value for a 0. 9% (0. 154 M) solution of Na. Cl which has a freezing point depression of 0. 52. Liso value = Tf/c = 0. 52/0. 154 = 3. 4 The interionic interaction in solutions that are not too concentrated is roughly the same for all uni-univalent electrolytes (same Liso value) regardless of the chemical nature. Similarly, Liso values can be considered similar for different electrolytes under the same class (types of electrolytes) as shown in the following figure and table. 3/4/2021 BA-FP-UJ-D
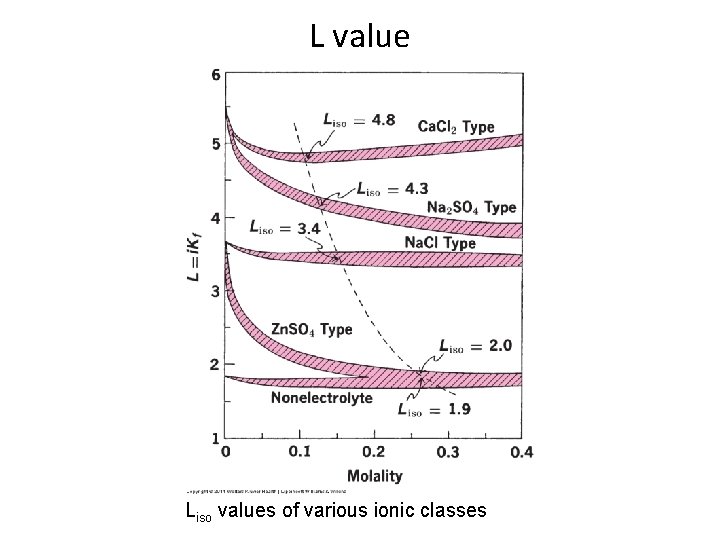
L value Liso values of various ionic classes
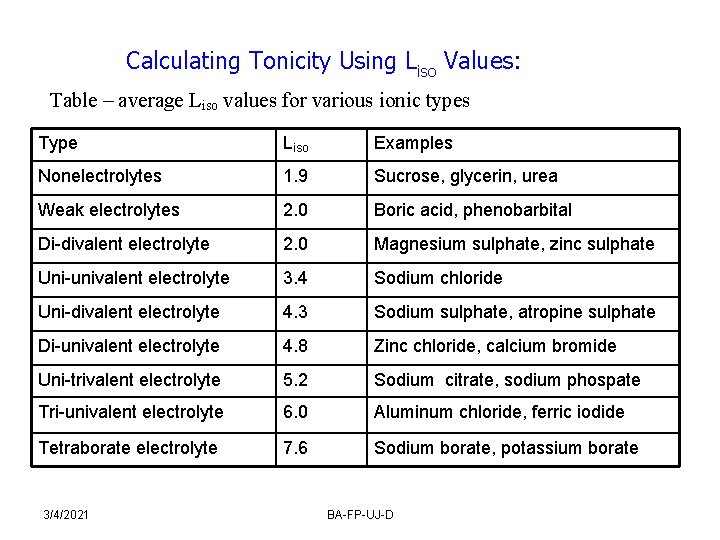
Calculating Tonicity Using Liso Values: Table – average Liso values for various ionic types Type Liso Examples Nonelectrolytes 1. 9 Sucrose, glycerin, urea Weak electrolytes 2. 0 Boric acid, phenobarbital Di-divalent electrolyte 2. 0 Magnesium sulphate, zinc sulphate Uni-univalent electrolyte 3. 4 Sodium chloride Uni-divalent electrolyte 4. 3 Sodium sulphate, atropine sulphate Di-univalent electrolyte 4. 8 Zinc chloride, calcium bromide Uni-trivalent electrolyte 5. 2 Sodium citrate, sodium phospate Tri-univalent electrolyte 6. 0 Aluminum chloride, ferric iodide Tetraborate electrolyte 7. 6 Sodium borate, potassium borate 3/4/2021 BA-FP-UJ-D

Example: What is the freezing point lowering at a 1% solution of Na Propionate (Mw= 96)? Na Propionate is uni- uinvalent; its Liso value is 3. 4. First we should calculate the molar concentration of the drug: c = number of moles/ volume of sol (L) c = (wt/Mw)/volume of sol (L) c = (1/96)/0. 1 L= 0. 104 M Tf = Liso x c= 3. 4 x 0. 104= 0. 35 °C 3/4/2021 BA-FP-UJ-D

Methods for adjusting tonicity: Method 1: Cryoscopic Method 2: Na. Cl Equivalent Method 3: White-Vincent Method (water is added to the drug in sufficient amount to form an isotonic solution). 3/4/2021 BA-FP-UJ-D

Cryoscopic Method: - Based on the calculation of Tf of the solution and how much Na. Cl should be added to make: Tf (solution) = Tf (blood) 3/4/2021 BA-FP-UJ-D

Example: How much Na. Cl should be added to 1% apomorphine. HCl (Mw= 312. 79) solution to make it isotonic with blood? Liso value of this drug is 2. 6. First we should calculate the molar concentration of the drug: c = number of moles/ volume of sol (L) c = (wt/Mw)/volume of sol (L) = (1/312. 79)/0. 1 L= 0. 032 M Tf = Liso x c= 2. 6 x 0. 032= 0. 08 °C Tf for the drug should be = 0. 52 C 0. 52 - 0. 08 = 0. 44 C (to be lowered) If 0. 9 gm Na. Cl in 100 ml have 0. 52 Tf , thus for 0. 44 0. 9 0. 44 0. 52 = 0. 76 gm Na. Cl the 100 ml solution. 3/4/2021 BA-FP-UJ-D that should be added to

Example: How much Na. Cl should be added to 2% apomorphine. HCl (Mw= 312. 79) solution to make it isotonic with blood? Liso value of this drug is 2. 6. First we should calculate the molar concentration of the drug: c = number of moles/ volume of sol (L) c = (wt/Mw)/volume of sol (L)= (2/312. 79)/0. 1 L= 0. 064 M Tf = Liso x c= 2. 6 x 0. 064= 0. 16 °C Tf for the drug should be = 0. 52 C 0. 52 - 0. 16 = 0. 36 C (to be lowered) If 0. 9 gm Na. Cl in 100 ml have 0. 52 Tf , thus for 0. 36 0. 9 0. 36 0. 52 = 0. 62 gm Na. Cl the 100 ml solution. 3/4/2021 BA-FP-UJ-D that should be added to

Sodium Chloride Equivalent Method: Na. Cl Equivalent (E): the amount of Na. Cl that is equivalent to (ie. Has the same osmotic effect as) 1 gm, or other weight unit, of the drug. Tf for (E) g of Na. Cl = Tf for (1)g of drug= 0. 52°C Liso x c = Liso x c Liso x (w/Mw)/1 L= Liso x (w/Mw)/1 L 3. 4 x (E/58. 4) = Liso x (1/Mw) Liso E= 17 x M. Wt 3/4/2021 BA-FP-UJ-D

Sodium Chloride Equivalent Method: E 17 Liso M. Wt Example: calculate the appropriate E value for a new amphetamine hydrochloride derivative (M. Wt = 187). The drug is uni-univalent salt, it has an Liso value of 3. 4. Its E value calculated for equation to be: E = 17 3. 4 = 0. 31 187 This answer means that 0. 31 g of Na. Cl is equivalent to 1 g of the drug amphetamine HCl 3/4/2021 BA-FP-UJ-D

Example: How much Na. Cl should be added to 1% apomorphine. HCl (Mw= 312. 79) solution to make it isotonic with blood? Liso value of this drug is 2. 6. E= 17 x (Liso/Mw)= 17 x (2. 6/ 312. 79)= 0. 14 g So 1 g of the drug is equivalent to 0. 14 g Na. Cl If 0. 9 gm Na. Cl in 100 ml have 0. 52 Tf , thus we still need: 0. 9 – 0. 14= 0. 76 gm Na. Cl the 100 ml solution. 3/4/2021 BA-FP-UJ-D that should be added to

Example: How much Na. Cl should be added to the following formula to make it isotonic with blood? Phenacaine Hydrochloride (Liso= 3. 3; Mw= 352. 85). . . 0. 06 gm Boric Acid (Liso= 1. 8; Mw= 61. 84). . . . 0. 30 gm Sterilized Distilled Water, enough to make. . . ………… 100. 0 ml Solution: For Phenacine: E= 17(Liso/Mw)= 17(3. 3/352. 85)= 0. 16 So 1 g of Phenancine is equivalent to 0. 16 g Na. Cl Thus: 0. 06 g of Phenacine is equivalent to 0. 0096 g Na. Cl For Boric acid: E= 17(Liso/Mw)= 17(1. 8/61. 84)= 0. 49 So 1 g of Boric acid is equivalent to 0. 49 g Na. Cl Thus: 0. 3 g of Boric acid is equivalent to 0. 147 g Na. Cl Overall, both drugs are equivalent to 0. 0096 + 0. 147= 0. 157 g As 0. 9 g Na. Cl in 100 m. L solution is isotonic, so 0. 9 -0. 157= 0. 743 g Na. Cl should be added to the mixture

White-Vincent Method: It involves the addition of certain volume of water to a specific weight of a drug (w) to make an isotonic solution, the final volume of this isotonic solution is (V). After adding the appropriate volume of water to prepare isotonic solution of drug, it is followed by the addition of an isotonic or isotonic-buffered diluting vehicle to bring the solution to the final volume. In other words, it is used when certain volume of isotonic solution is to be prepared then diluted with already available isotonic solution. V = w E 111. 1 Make your calculations to show this equation was derived!! V: volume of isotonic solution (m. L) prepared by addition of water to the drug. w: the weight of the drug in grams. E: Na. Cl equivalent. 3/4/2021 BA-FP-UJ-D

Example Make the following solution isotonic with respect to an ideal membrane. Phenacaine Hydrochloride (Liso= 3. 3; Mw= 352. 85). . . 0. 06 gm Boric Acid (Liso= 1. 8; Mw= 61. 84). . . . 0. 30 gm Sterilized Distilled Water, enough to make. . . ………… 100. 0 ml Solution: For Phenacine: E= 17(Liso/Mw)= 17(3. 3/352. 85)= 0. 16 For Boric acid: E= 17(Liso/Mw)= 17(1. 8/61. 84)= 0. 49 V = [w E 111. 1] = [(0. 06 0. 16) + (0. 3 0. 49)] 111. 1 = 17. 4 ml H 2 O dissolve the two drugs in water to prepare a final solution of 17. 4 isotonic solution, then add isotonic solution to make the volume 100 ml (82. 6 ml isotonic solution). 3/4/2021 BA-FP-UJ-D
3/4/2021 BA-FP-UJ-D
- Slides: 23