ISOTHERMAL HOMOGENEOUS REACTORS 2 PFR Tubular Reactor PFR

ISOTHERMAL HOMOGENEOUS REACTORS 2. PFR

Tubular Reactor PFR Assumptions: Ideal Tubular Reactor 1. Fluid moves like a solid plug or piston(PFR). 2. m. f. r. , temp. , pressure & composition are uniform across any section ┴ fluid motion. [no mixing in axial direc. & complete mixing in radial direc. ]
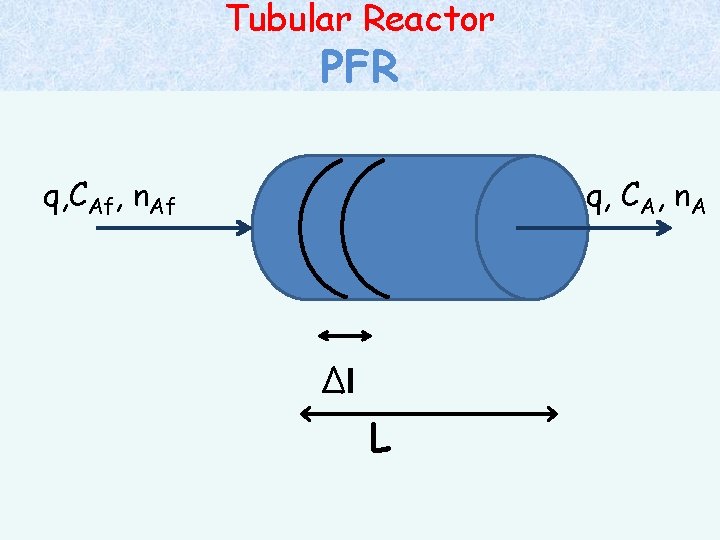
Tubular Reactor PFR q, CAf, n. Af q, CA, n. A ∆l L
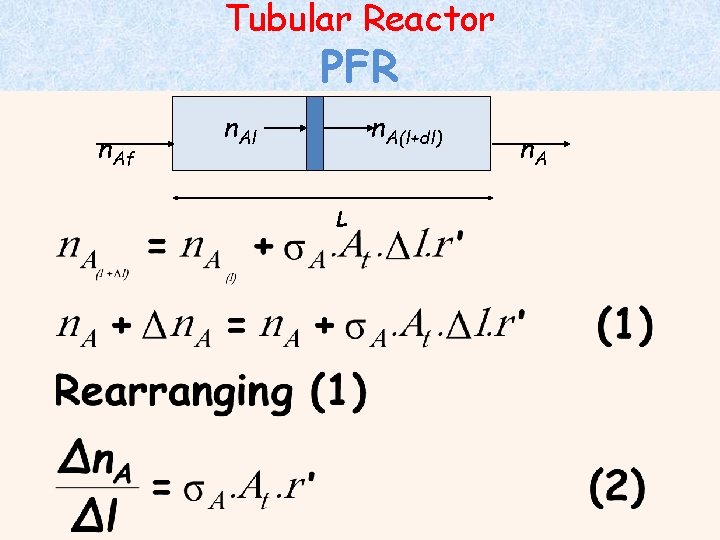
Tubular Reactor PFR n. Af n. Al n. A(l+dl) n. A L n. A

Tubular Reactor PFR Integrating (5) using initial conditions CA = CAf at VR= 0 The length of the reactor, L, can be calculated or VR; VR = At. L

Tubular Reactor PFR Or, in general using the definition of conversion,

Tubular Reactor PFR Let us consider the 1 st order liquid phase isothermal reaction A B to be operated in a PFR Separating variables,

Example: Assume isomerization is first order with k = 0. 23 min-1 What volume PFR and CSTR are required for 90% conversion and 10 L/min?

Tubular Reactor Example: PFR The elementary, liquid-phase, irreversible reaction is to be carried out in a flow reactor. Two reactors are available, an 800 dm 3 PFR that can only be operated at 300 K and a 200 dm 3 CSTR that can be operated at 350 K. The two feed streams to the reactor mix to form a single feed stream that is equal molar in A and B, with a total volumetric flowrate of 10 dm 3/min. Which of the two reactors will give us the highest conversion? Additional Information: at 300 K, k = 0. 07 dm 3/mol-min / E = 85000 J/mol-K CA 0 B = CB 0 B = 2 mol/dm 3 /v. A 0 = v. B 0 = 0. 5*v 0 = 5 dm 3/min; v = q
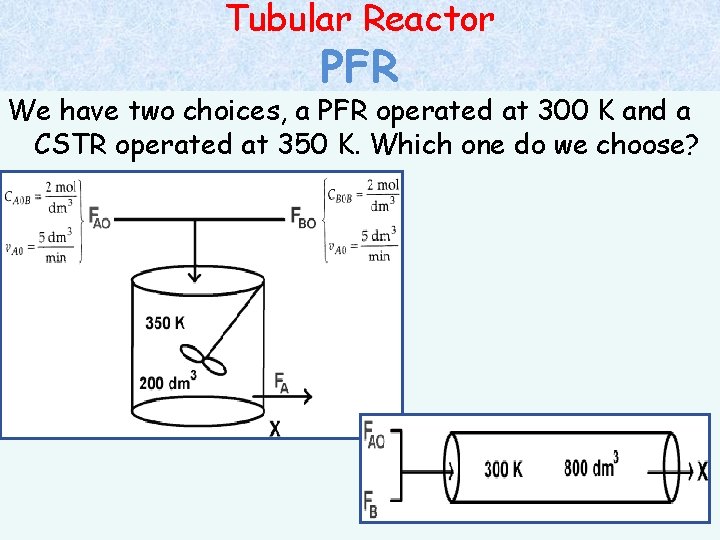
Tubular Reactor PFR We have two choices, a PFR operated at 300 K and a CSTR operated at 350 K. Which one do we choose?

Tubular Reactor PFR Given: k 0, E, V, q. Ao, CA 0 B, CB 0 B ; CAOB is feed conc. of A Before mixing with q. Bo. Required: x Assumptions: ü The CSTR is well mixed. ü There are no radial variations in the PFR. ü Isothermal, no pressure drop. Evaluations: ü At 350 K ü
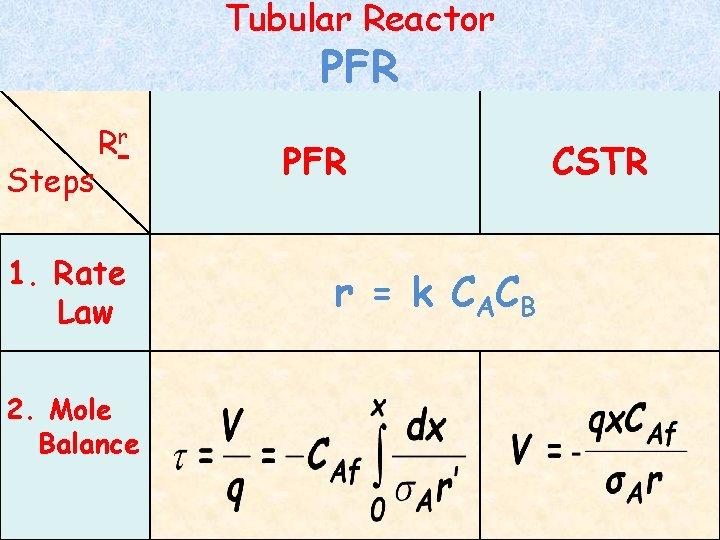
Tubular Reactor PFR Steps Rr 1. Rate Law 2. Mole Balance PFR r = k C AC B CSTR
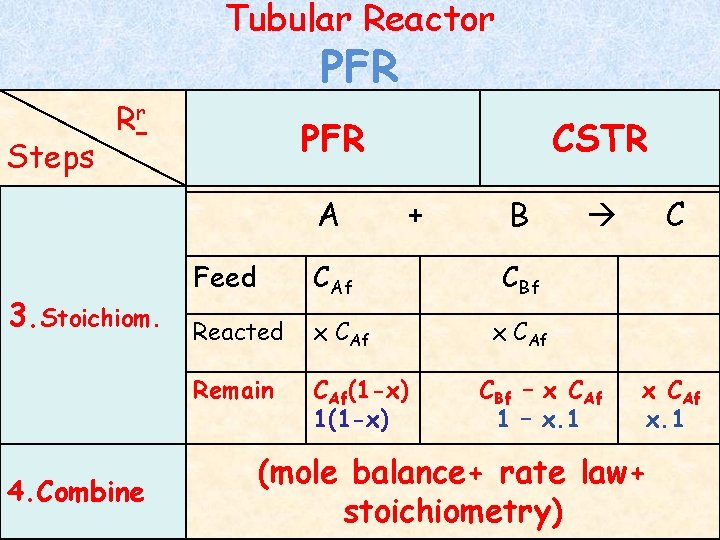
Tubular Reactor Steps PFR Rr PFR A 3. Stoichiom. 4. Combine CSTR + Feed CAf Reacted x CAf Remain CAf(1 -x) 1(1 -x) B C CBf x CAf CBf – x CAf 1 – x. 1 x CAf x. 1 (mole balance+ rate law+ stoichiometry)

Tubular Reactor PFR Example: The aqueous phase 1 st order decomposition of A is investigated in 2 PFRs in series. At st. with a feed conc. of 1 mole A/lit. and mean residence time of 96 seconds in the first reactor. The conc. in the first reactor is 0. 5 mole A/lit and in the second reactor is 0. 15 mole A/lit. Find the ratio of 2 nd reactor volume to 1 st one. (V 2/V 1)
- Slides: 14