Isomers Molecules with same molecular formula but their

Isomers Molecules with same molecular formula but their respective atoms are arranged differently in space 1

Structural Isomers ¢ Structural isomers have the same molecular formulas but they differ in their structural formulas. 2

Examples ¢ butane and methylpropane ¢ ethanol and methoxymethane

Stereoisomers ¢ ¢ Stereo isomers have the same structural formulas but they differ in their spatial arrangements. There are two types of stereoisomerism 1. 2. Geometrical isomerism Optical isomerism 4

Geometrical isomers ¢ ¢ Geometrical isomers occur in organic molecules where rotation around a bond is restricted This occurs most often around C=C Example: cis-but-2 -ene and trans-but-2 -ene 5
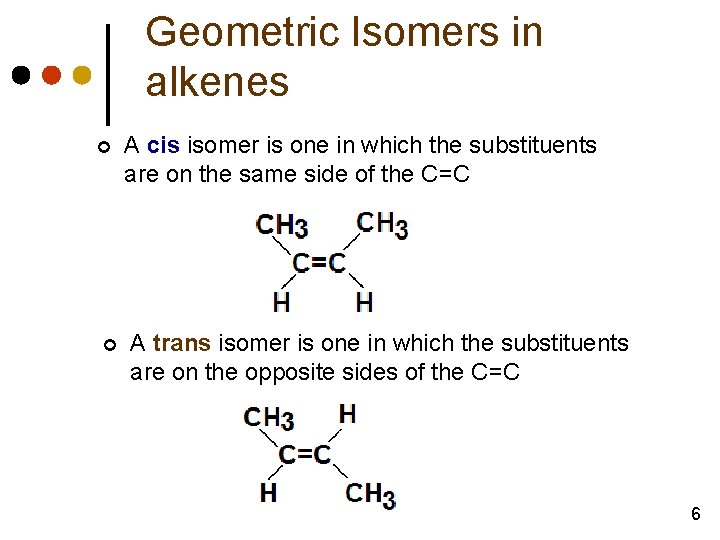
Geometric Isomers in alkenes ¢ ¢ A cis isomer is one in which the substituents are on the same side of the C=C A trans isomer is one in which the substituents are on the opposite sides of the C=C 6
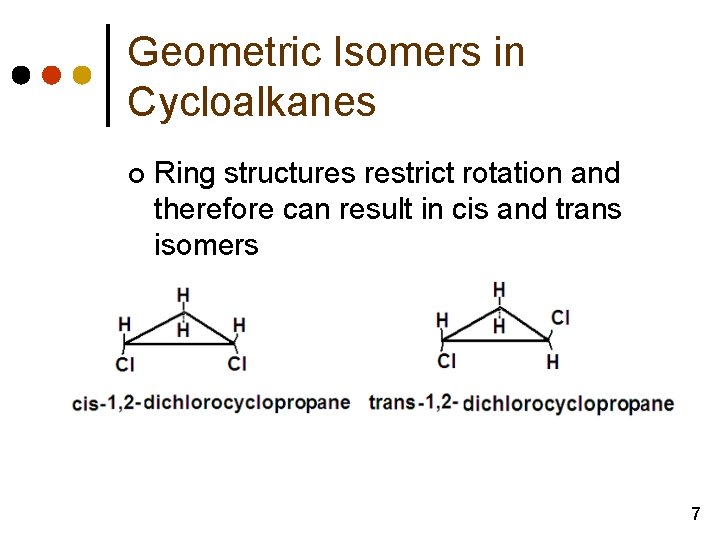
Geometric Isomers in Cycloalkanes ¢ Ring structures restrict rotation and therefore can result in cis and trans isomers 7

Properties of Geometrical Isomers The chemical properties of geometrical isomers tend to be similar but their physical properties are different 8
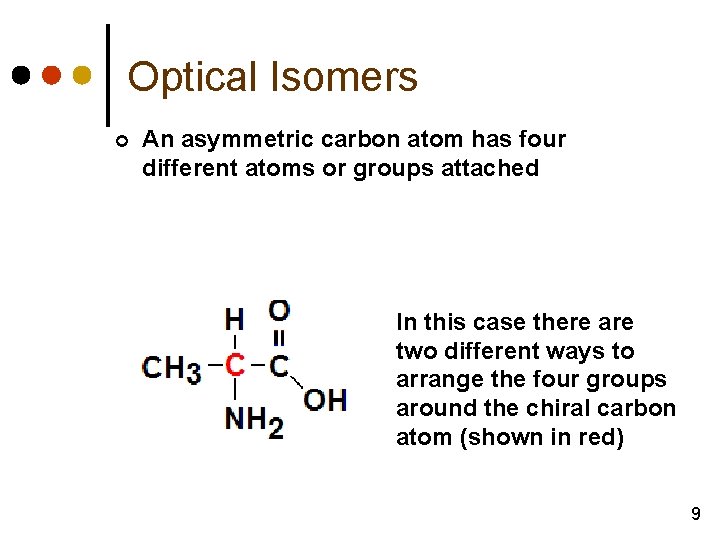
Optical Isomers ¢ An asymmetric carbon atom has four different atoms or groups attached In this case there are two different ways to arrange the four groups around the chiral carbon atom (shown in red) 9
Optical Isomers While these structures may look identical, in three dimensions they are mirror images of each other. Such molecules are called enantiomers. 10

Task ¢ Draw the structural isomers for C 4 H 10 O. How many do you get? ¢ https: //www. youtube. com/watch? v=9 X a. SI 2 XYGt 4
- Slides: 11