ISM Astrochemistry Lecture 6 Stellar Evolution Star Death












- Slides: 30

ISM & Astrochemistry Lecture 6

Stellar Evolution

Star Death

IRC+10216 (CW Leo) • Nearby (~130 pc) high mass-loss carbon star (AGB) • Brightest object in the sky at 2 microns – optically invisible • Carbon dust envelope detected out to 200’’ = 25, 000 AU ( ~ 1 lt yr) • Molecular shells at ~ 1000 - 4000 AU • >60 molecules detected: CO, C 2 H 2, HC 9 N. . . • Newly discovered anions C 8 H-, C 6 H-, C 4 H- , C 3 N - , C 5 N • Recent detections of H 2 O, OH and H 2 CO Figures from Leao et al. (2006) Lucas and Guelin et al. (1999)

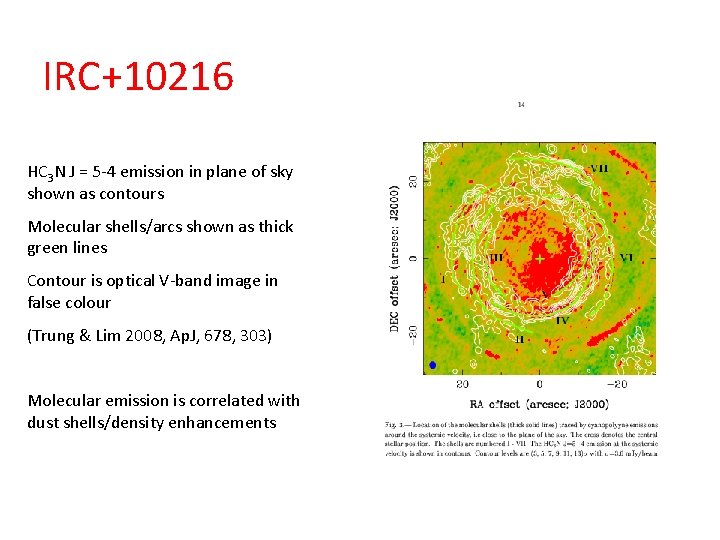
IRC+10216 HC 3 N J = 5 -4 emission in plane of sky shown as contours Molecular shells/arcs shown as thick green lines Contour is optical V-band image in false colour (Trung & Lim 2008, Ap. J, 678, 303) Molecular emission is correlated with dust shells/density enhancements

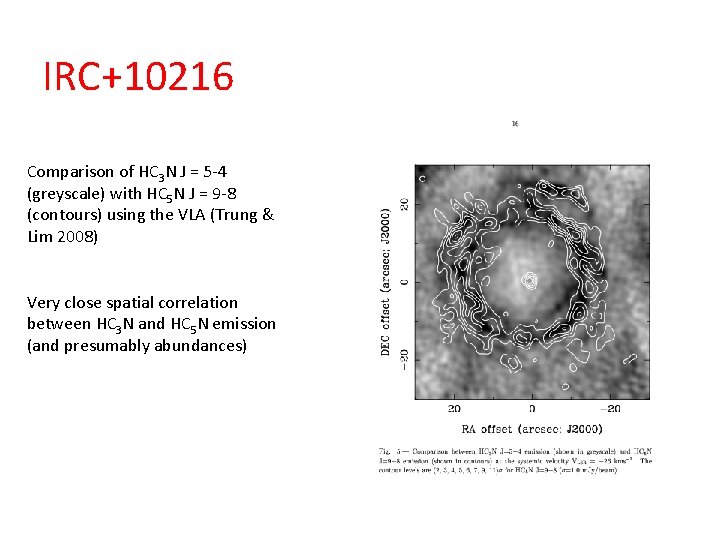
IRC+10216 Comparison of HC 3 N J = 5 -4 (greyscale) with HC 5 N J = 9 -8 (contours) using the VLA (Trung & Lim 2008) Very close spatial correlation between HC 3 N and HC 5 N emission (and presumably abundances)

Chemical Structure of AGB CSE

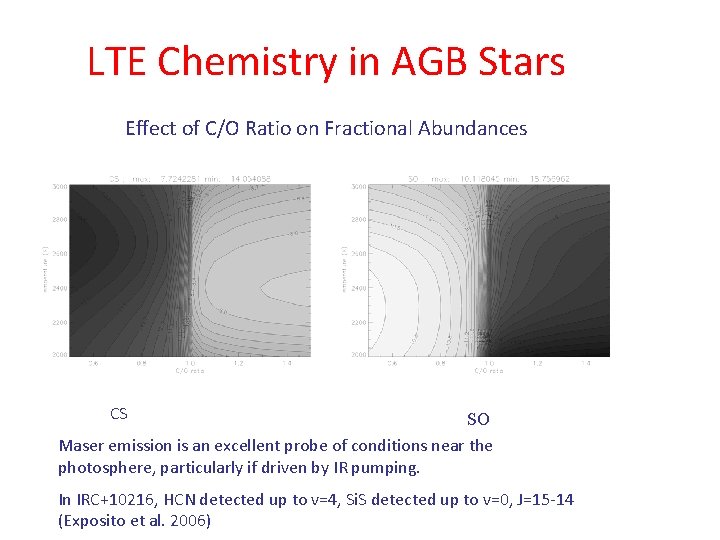
LTE Chemistry in AGB Stars Effect of C/O Ratio on Fractional Abundances CS SO Maser emission is an excellent probe of conditions near the photosphere, particularly if driven by IR pumping. In IRC+10216, HCN detected up to v=4, Si. S detected up to v=0, J=15 -14 (Exposito et al. 2006)

Dust Formation in AGB Stars Dust formation in AGB stars requires: (i) a cooling flow (ii) high collisional rates (iii) time Shocks caused by pulsations are critical – they give large density enhancements, strong post-shock cooling, and lift material slowly away from stellar surface Spatial scale is less than 5 stellar radii Probe with vibrationally excited/maser species (line-widths less than terminal velocity/IR pumped) – eg thermal v=1 Si. S emission (Exposito et al. 2006) SMA J = 5 -4 Si. O observations (Schoier et al. 2006) show Si. O much more abundant than LTE value in IRC+10216 at 3 -8 stellar radii but lower at larger radii – condensation on to dust grains?

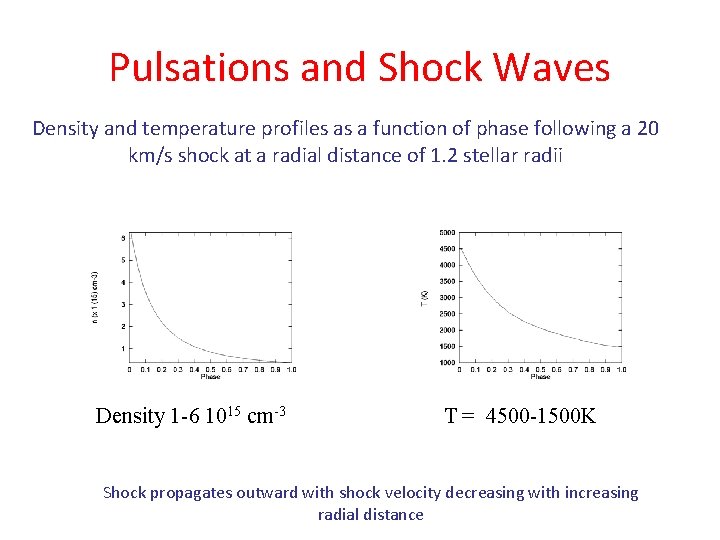
Pulsations and Shock Waves Density and temperature profiles as a function of phase following a 20 km/s shock at a radial distance of 1. 2 stellar radii Density 1 -6 1015 cm-3 T = 4500 -1500 K Shock propagates outward with shock velocity decreasing with increasing radial distance

Shock Chemistry Input – LTE abundances for IRC+10216 parameters Each cycle breaks down into three zones: (i) Immediate post-shock – H 2 and other LTE molecules destroyed (ii) Post-shock – H 2 and other molecules form (ii) Dynamical time less than chemical time – freeze -out of abundances (iv) No significant water formed in shock Abundances at the end of the pulsation zone can be very different from LTE Si. O enhanced, HCN, CS destroyed in C-stars (Willacy & Cherchneff 1998)

Shock Chemistry Formation of Molecules in O-rich and S-type Stars Form carbon-bearing molecules in O-rich stars – C/O < 1: (i) HCN increased by 6 orders of magnitude above LTE value (ii) CO 2 increased by 3 orders of magnitude (iii) CS increased by 3 orders of magnitude (Duari, Cherchneff & Willacy 1999) For S-type stars (Chi Cygni) – C/O ~1: HCN in inner envelope increased by 4 orders of magnitude (vibrationally excited HCN detected here – IR pumping within 33 stellar radii) (Duari & Hatchell 2000)

Photochemistry in CSEs Destruction of Parents by IS UV Radiation Field Self-Shielding H 2 – very reactive, daughter (H atoms) unreactive CO – very unreactive, daughters (C, C+) very reactive Cosmic ray ionisation f(H 3+) varies as r 2 N(H 3+) ~ 1012 cm-2 for CRI rate of 10 -17 s-1, an order of magnitude less than that detected in the interstellar medium Photodissociation and photoionisation Acetylene is the species which determines the complexity of the hydrocarbon chemistry

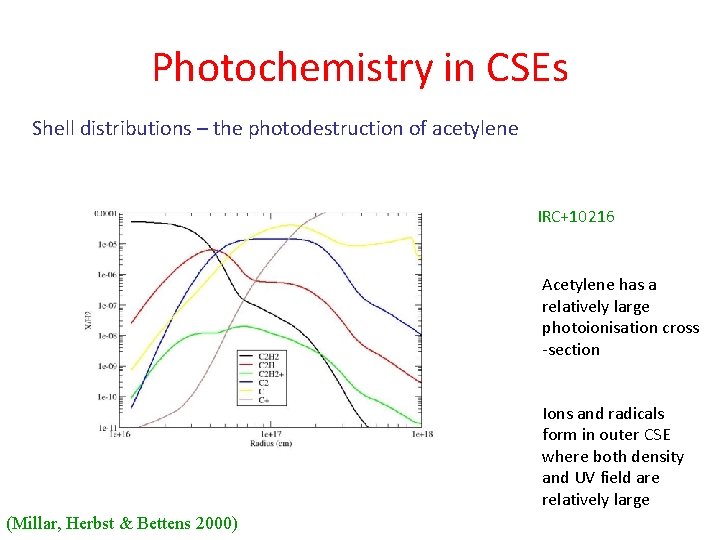
Photochemistry in CSEs Shell distributions – the photodestruction of acetylene IRC+10216 Acetylene has a relatively large photoionisation cross -section Ions and radicals form in outer CSE where both density and UV field are relatively large (Millar, Herbst & Bettens 2000)

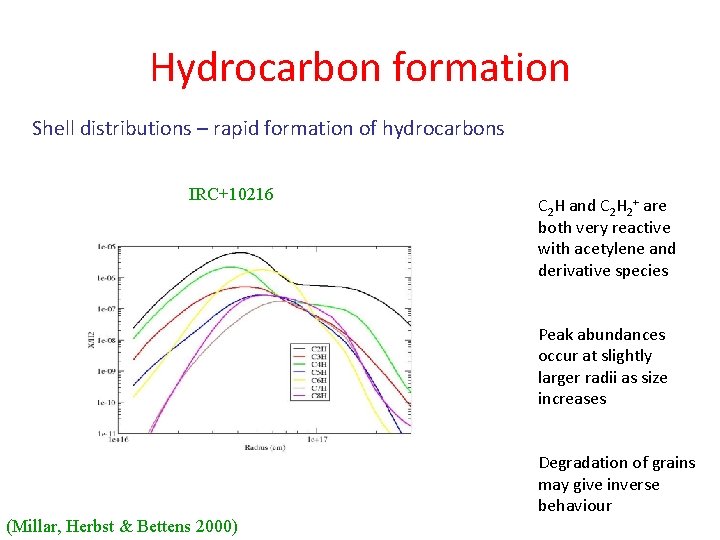
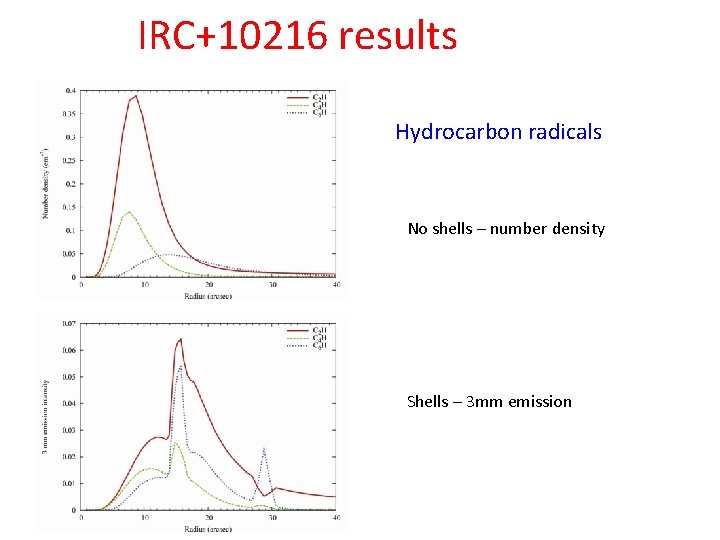
Hydrocarbon formation Shell distributions – rapid formation of hydrocarbons IRC+10216 C 2 H and C 2 H 2+ are both very reactive with acetylene and derivative species Peak abundances occur at slightly larger radii as size increases (Millar, Herbst & Bettens 2000) Degradation of grains may give inverse behaviour

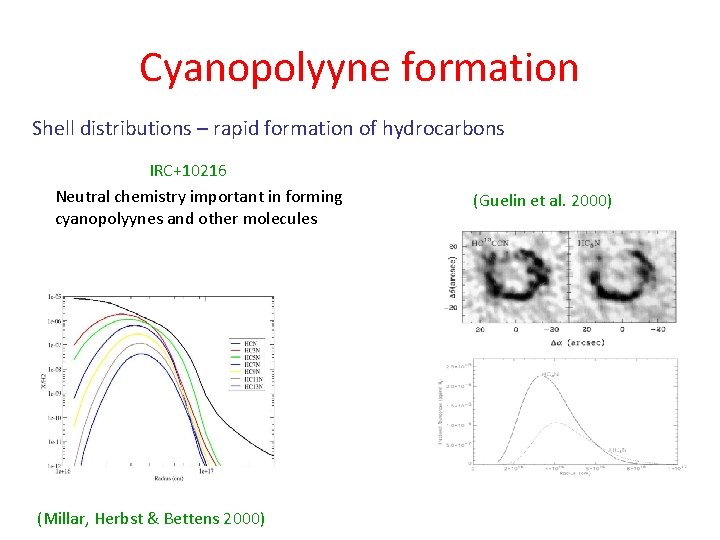
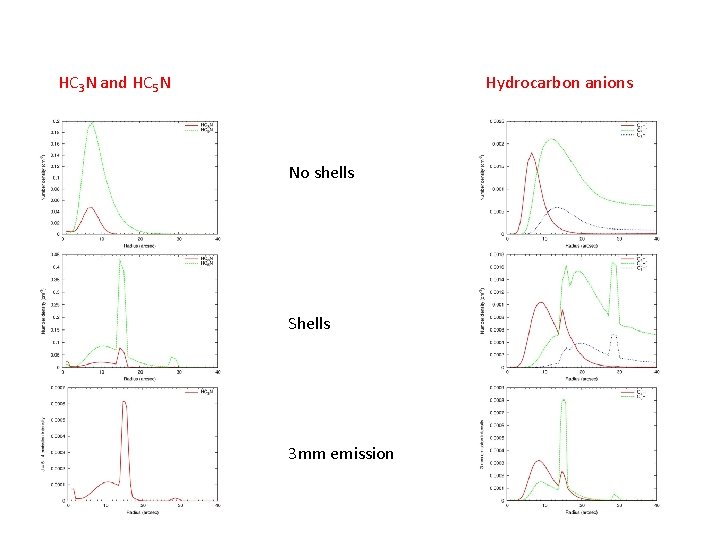
Cyanopolyyne formation Shell distributions – rapid formation of hydrocarbons IRC+10216 Neutral chemistry important in forming cyanopolyynes and other molecules (Millar, Herbst & Bettens 2000) (Guelin et al. 2000)

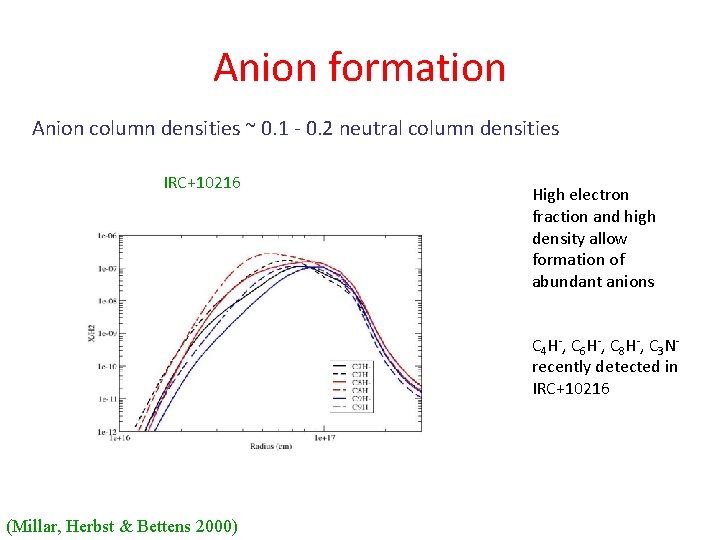
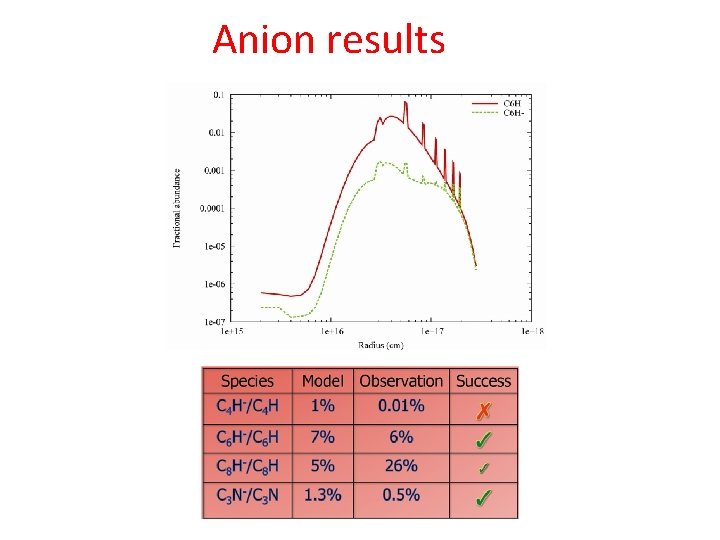
Anion formation Anion column densities ~ 0. 1 - 0. 2 neutral column densities IRC+10216 High electron fraction and high density allow formation of abundant anions C 4 H-, C 6 H-, C 8 H-, C 3 Nrecently detected in IRC+10216 (Millar, Herbst & Bettens 2000)

Modelling the circumstellar shells Leao et al (2006): Concentric rings

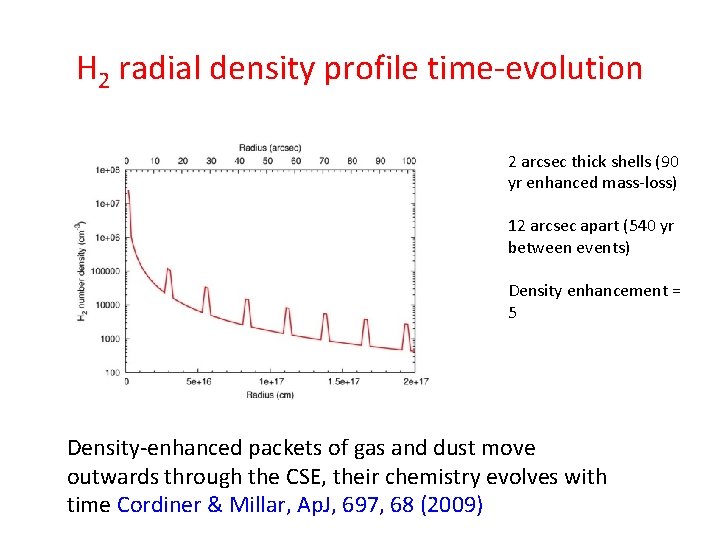
H 2 radial density profile time-evolution 2 arcsec thick shells (90 yr enhanced mass-loss) 12 arcsec apart (540 yr between events) Density enhancement = 5 Density-enhanced packets of gas and dust move outwards through the CSE, their chemistry evolves with time Cordiner & Millar, Ap. J, 697, 68 (2009)

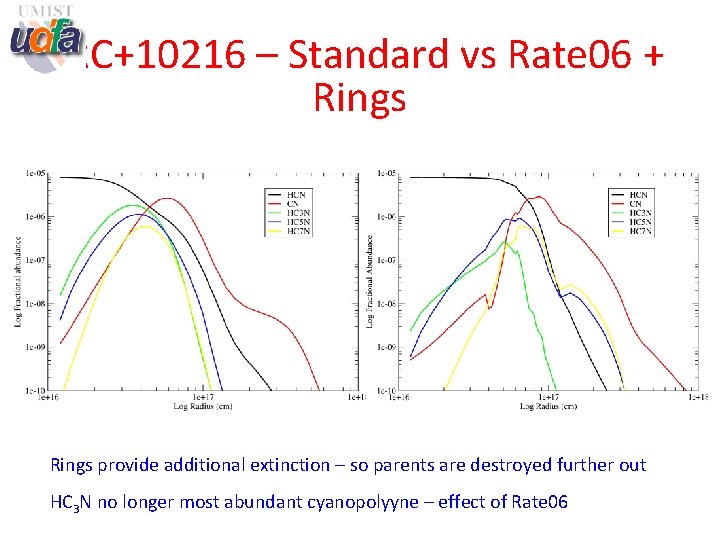
IRC+10216 – Standard vs Rate 06 + Rings provide additional extinction – so parents are destroyed further out HC 3 N no longer most abundant cyanopolyyne – effect of Rate 06

Anion results

IRC+10216 results Hydrocarbon radicals No shells – number density Shells – 3 mm emission

HC 3 N and HC 5 N Hydrocarbon anions No shells Shells 3 mm emission

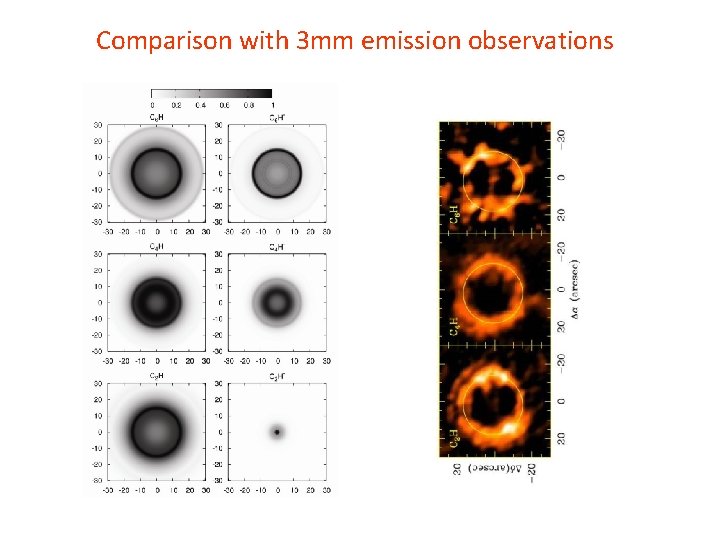
Comparison with 3 mm emission observations

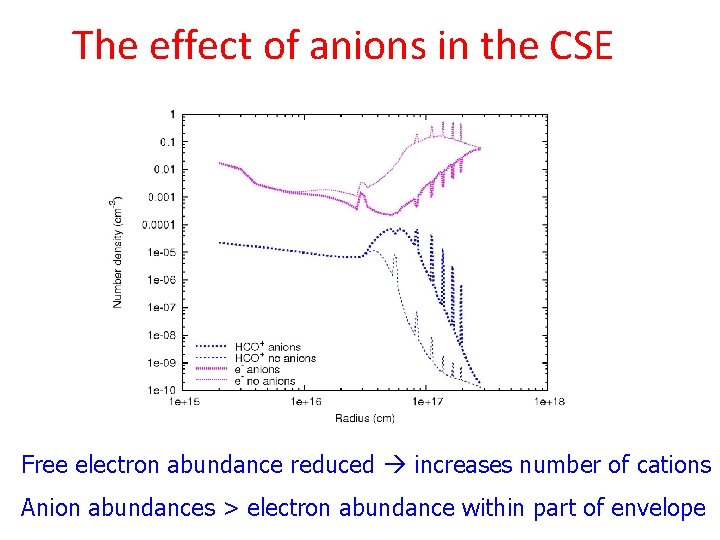
The effect of anions in the CSE Free electron abundance reduced increases number of cations Anion abundances > electron abundance within part of envelope

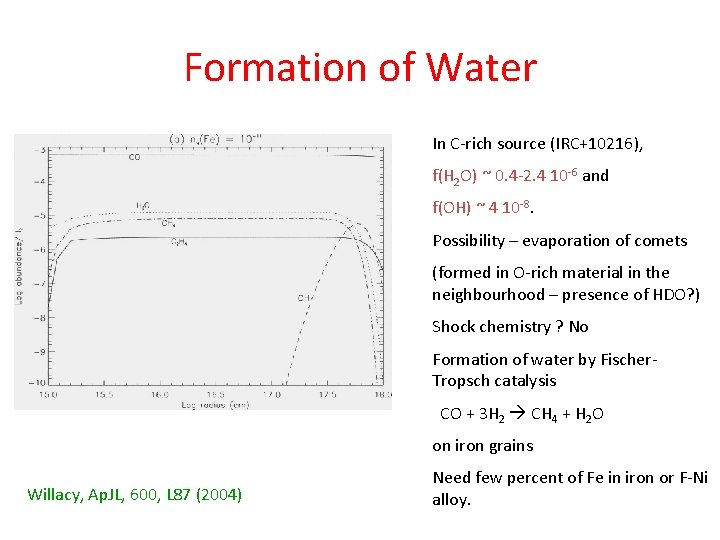
Formation of Water In C-rich source (IRC+10216), f(H 2 O) ~ 0. 4 -2. 4 10 -6 and f(OH) ~ 4 10 -8. Possibility – evaporation of comets (formed in O-rich material in the neighbourhood – presence of HDO? ) Shock chemistry ? No Formation of water by Fischer. Tropsch catalysis CO + 3 H 2 CH 4 + H 2 O on iron grains Willacy, Ap. JL, 600, L 87 (2004) Need few percent of Fe in iron or F-Ni alloy.

IRC+10216 summary • Successfully model C 6 H-, C 8 H-, C 3 N- abundances • C 6 H- and C 8 H- formed by radiative electron attachment, C 3 N- formed by Cn. H- + N • C 4 H- overproduced by ~ 100 times • CN- and C 2 H- predicted to be observable in the inner CSE even though the electron attachment rate is very low, due to reaction of H- with HCN and C 2 H 2 • Density-enhancements in the CSE may result in narrow emission rings / arcs of carbon-chain species and their anions

Photochemistry in O-rich CSEs Destruction of Parents by IS UV Radiation Field Self-Shielding H 2 – very reactive, daughter (H atoms) unreactive CO – very unreactive, daughters (C, C+) very reactive H 2 O – can self-shield but only weakly, daughter (OH) very reactive Photodissociation and photoionisation Water drives chemistry through production of OH Water photoionisation small so neutral-neutral chemistry dominates

Photochemistry in O-rich CSEs Radical reactions produce oxides OH reacts rapidly with other daughters O to produce O 2 N to produce NO S to produce SO SO to produce SO 2 Si to produce Si. O P to produce PO, etc

Problems C-rich AGB CSEs Detection of water, formaldehyde and OH in outer envelope of IRC+10216 Pulsational shocks – no, too little formed – and in inner region Photochemistry – no, not enough O freed from parents Fisher-Tropsch-Type chemistry (grains) – possibly Evaporation of comets - possibly O-rich AGB CSEs Formation of carbon-bearing molecules HCN, HNC, CN, CS, H 2 CO, OCS Not enough carbon released by photodissociation of CO Pulsational shocks – can help, but in inner region Grain chemistry – not yet studied