ISM Astrochemistry Lecture 2 Protoplanetary Nebula The evolutionary

ISM & Astrochemistry Lecture 2

Protoplanetary Nebula The evolutionary stage between evolved stars and planetary nebula CRL 618 – many organic molecules Including the only extra-solar system detection of benzene, C 6 H 6 Time scale of chemistry and evolution of this object is 600 -1000 years

Molecule formation in shocks Supersonic shock waves: Sound speed ~ 1 km s-1 Shocks compress and heat the gas Hydrodynamic (J-type) shocks: immediately post-shock, density jumps by 4 -6, gas temperature ~ 3000(VS/10 km s-1)2 Gas cools quickly (~ few tens, hundred years) and increases its density further as it cools – path lengths are small. MHD (C-type) shocks: shock front is preceded by a magnetic precursor, gas density and temperature change continuously, ions and neutrals move at different velocities – path lengths are large Importance for chemistry: Endothermic neutral-neutral
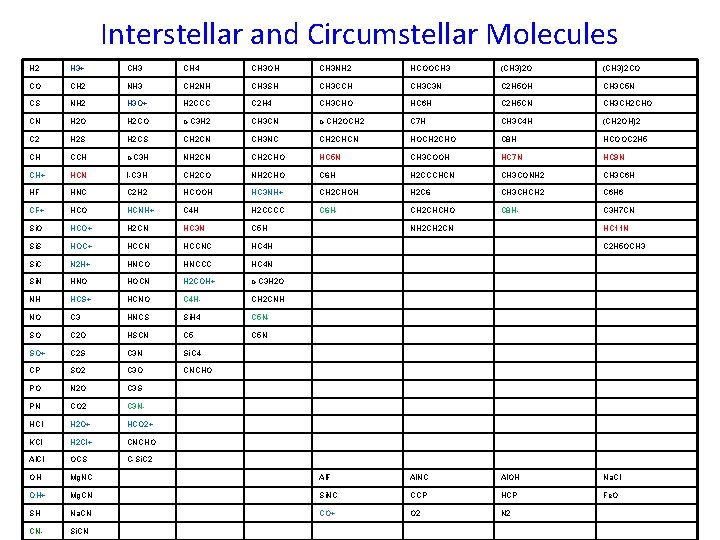
Interstellar and Circumstellar Molecules H 2 H 3+ CH 3 CH 4 CH 3 OH CH 3 NH 2 HCOOCH 3 (CH 3)2 O (CH 3)2 CO CO CH 2 NH 3 CH 2 NH CH 3 SH CH 3 CCH CH 3 C 3 N C 2 H 5 OH CH 3 C 5 N CS NH 2 H 3 O+ H 2 CCC C 2 H 4 CH 3 CHO HC 6 H C 2 H 5 CN CH 3 CH 2 CHO CN H 2 O H 2 CO c-C 3 H 2 CH 3 CN c-CH 2 OCH 2 C 7 H CH 3 C 4 H (CH 2 OH)2 C 2 H 2 S H 2 CS CH 2 CN CH 3 NC CH 2 CHCN HOCH 2 CHO C 8 H HCOOC 2 H 5 CH CCH c-C 3 H NH 2 CN CH 2 CHO HC 5 N CH 3 COOH HC 7 N HC 9 N CH+ HCN l-C 3 H CH 2 CO NH 2 CHO C 6 H H 2 CCCHCN CH 3 CONH 2 CH 3 C 6 H HF HNC C 2 H 2 HCOOH HC 3 NH+ CH 2 CHOH H 2 C 6 CH 3 CHCH 2 C 6 H 6 CF+ HCO HCNH+ C 4 H H 2 CCCC C 6 H- CH 2 CHCHO C 8 H- C 3 H 7 CN Si. O HCO+ H 2 CN HC 3 N C 5 H Si. S HOC+ HCCNC HC 4 H Si. C N 2 H+ HNCO HNCCC HC 4 N Si. N HNO HOCN H 2 COH+ c-C 3 H 2 O NH HCS+ HCNO C 4 H- CH 2 CNH NO C 3 HNCS Si. H 4 C 5 N- SO C 2 O HSCN C 5 N SO+ C 2 S C 3 N Si. C 4 CP SO 2 C 3 O CNCHO PO N 2 O C 3 S PN CO 2 C 3 N- HCl H 2 O+ HCO 2+ KCl H 2 Cl+ CNCHO Al. Cl OCS C-Si. C 2 OH Mg. NC Al. F Al. NC Al. OH Na. Cl OH+ Mg. CN Si. NC CCP HCP Fe. O SH Na. CN CO+ O 2 N 2 CN- Si. CN NH 2 CN HC 11 N C 2 H 5 OCH 3

One-body reactions Photodissociation/photoionisation: Unshielded photorates in ISM: β 0 = 10 -10 s-1 Within interstellar clouds, characterise extinction of UV photons by the visual extinction, AV, measured in magnitudes, so that: β = β 0 exp(-b. AV) where b is a constant (~ 1 - 3) and differs for different molecules

Cosmic Ray Ionisation H 2 + crp → H 2+ + e- H 2+ + H 2 → H 3+ + H He + crp → He+ + e- He+ + H 2 → products exothermic but unreactive H 3+: P. A. (H 2) very low Proton transfer reactions very efficient Key to synthesising molecules He+: I. P. (He) very large Breaks bonds in reaction Key to destruction of molecules IS Chemistry efficient because He+ does not react with H 2

Two-body reactions Ion-neutral reactions: Neutral-neutral reactions: Ion-electron dissociative recombination (molecular ions) Ion-electron radiative recombination (atomic ions) Radiative association Three-body reactions (only if density is very large, 1013 cm-3)
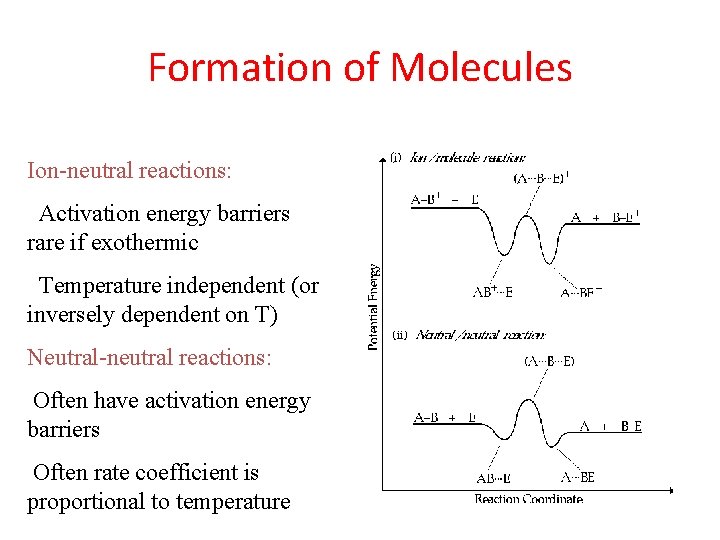
Formation of Molecules Ion-neutral reactions: Activation energy barriers rare if exothermic Temperature independent (or inversely dependent on T) Neutral-neutral reactions: Often have activation energy barriers Often rate coefficient is proportional to temperature

Formation of Molecules Ion-electron dissociative recombination reactions: Fast, multiple products, inverse T dependence Atomic ion-electron radiative recombination: Neutral complex stabilises by emission of a photon, about 1000 times slower than DR rate coefficients Radiative association: A+ + B → AB+ + hν Photon emission more efficient as size of complex grows, therefore can be important in synthesising large molecular ions CH 3+ + H 2 → CH 5+ + h ν k(T) = 1. 3 10 -13(T/300)-1 cm 3 s-1 CH 3+ + HCN → CH 3 CNH+ + h ν k(T) = 9. 0 10 -9(T/300)-0. 5 cm 3 s-1

Chemical Kinetics A+B→C+D k = <σv> cm 3 s-1 Loss of A (and B) per unit volume per second is: dn(A)/dt = - kn(A)n(B) cm-3 s-1 where n(A) = no. of molecules of A per unit volume Formation of C (and D) per unit volume per second is: dn(C)/dt = + kn(A)n(B) cm-3 s-1 - Second-order kinetics – rate of formation and loss proportional to the concentration of two reactants

First-order kinetics A + hν → C + D β (units s-1) Loss of A (and B) per unit volume per second is: dn(A)/dt = - βn(A) cm-3 s-1 where β = photodissociation rate of A (s-1) Aside: The number, more accurately, flux of UV photons or cosmic-ray particles, is contained within β or ς - First-order kinetics – rate of formation and loss proportional to the concentration of one reactant
![General case dn(Xj)/dt = Σ klm[Xl][Xm] + Σ βn[Xn] - [Xj]{Σ kjl[Xl] + Σ General case dn(Xj)/dt = Σ klm[Xl][Xm] + Σ βn[Xn] - [Xj]{Σ kjl[Xl] + Σ](http://slidetodoc.com/presentation_image_h2/bd6c1af65811e71c378e95ed3f4842f9/image-12.jpg)
General case dn(Xj)/dt = Σ klm[Xl][Xm] + Σ βn[Xn] - [Xj]{Σ kjl[Xl] + Σ βj} m-3 s-1 or d[X]/dt = FX – LX[X] Need to solve a system of first-order, non-linear ODEs - solve using GEAR techniques -Steady-state approximation – rate of formation = rate of loss FX = LX[X]ss so that [X]ss = FX/LX Need to solve a system of non-linear algebraic equations - solve using Newton-Raphson methods
![Time scales d[X]/dt = FX – LX[X] For simplicity, assume FX and LX are Time scales d[X]/dt = FX – LX[X] For simplicity, assume FX and LX are](http://slidetodoc.com/presentation_image_h2/bd6c1af65811e71c378e95ed3f4842f9/image-13.jpg)
Time scales d[X]/dt = FX – LX[X] For simplicity, assume FX and LX are constants and [X] = 0 at t =0 (initial condition) Solution is: [X, t] = (FX/LX){1 – e-Lxt} [X, t] = [X]ss{1 – e-t/tc} where tc = 1/LX Note: As t → ∞, [X] → [X]ss When t = tc, [X, tc] = 0. 63[X]ss, so most molecular evolution occurs within a few times tc
![Grain Surface Time-scales Collision time: tc = [v. H(πr 2 nd)]-1 ~ 109/n(cm-3) years Grain Surface Time-scales Collision time: tc = [v. H(πr 2 nd)]-1 ~ 109/n(cm-3) years](http://slidetodoc.com/presentation_image_h2/bd6c1af65811e71c378e95ed3f4842f9/image-14.jpg)
Grain Surface Time-scales Collision time: tc = [v. H(πr 2 nd)]-1 ~ 109/n(cm-3) years Thermal hopping time: th = ν 0 -1 exp(Eb/k. T) Tunnelling time: tt = v 0 -1 exp[(4πa/h)(2 m. Eb)1/2] Thermal desorption time: tev = ν 0 -1 exp(ED/k. T) Here Eb ~ 0. 3 ED, so hopping time < desorption time For H at 10 K, ED = 300 K, tt ~ 2 10 -11 s, th ~ 7 10 -9 s Tunnelling time < hopping time only for lightest species (H, D) For O, ED ~ 800 K, th ~ 0. 025 s. For S, ED ~ 1100 K, th ~ 250 s, tt ~ 2 weeks Heavy atoms are immobile compared to H atoms

Formation of H 2 Gas phase association of H atoms far too slow, k ~ 10 -30 cm 3 s-1 Gas and dust well-mixed In low-density gas, H atoms chemisorb and fill all binding sites (106) per grain Subsequently, H atoms physisorb Surface mobility of these H atoms is large, even at 10 K. H atoms scans surface until it finds another atom with which it combines to form H
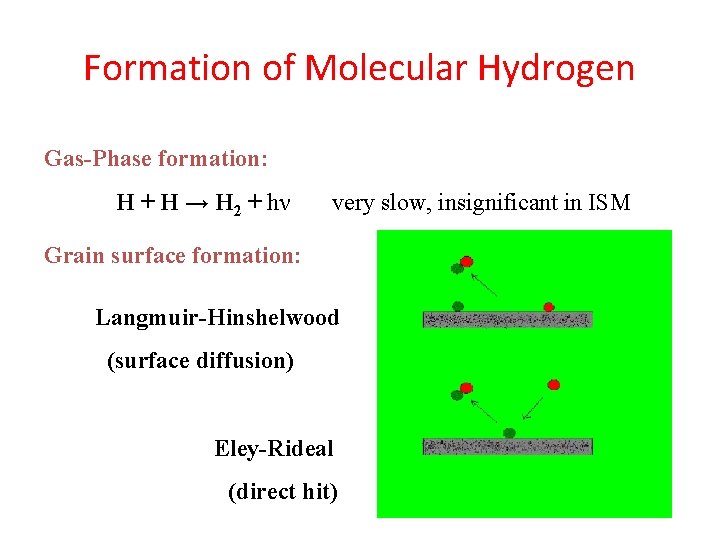
Formation of Molecular Hydrogen Gas-Phase formation: H + H → H 2 + hν very slow, insignificant in ISM Grain surface formation: Langmuir-Hinshelwood (surface diffusion) Eley-Rideal (direct hit)

Grain Surface Chemistry Zero-order approximation: Since H atoms are much more mobile than heavy atoms, hydrogenation dominates if n(H) > Σn(X), X = O, C, N Zero-order prediction: Ices should be dominated by the hydrogenation of the most abundant species which can accrete from the gas-phase Accretion time-scale: tac(X) = (SXv. Xσnd)-1, where SX is the sticking coefficient ~ 1 at 10 K tac (yrs) ~ 109/n(cm-3) ~ 104 – 105 yrs in a dark cloud
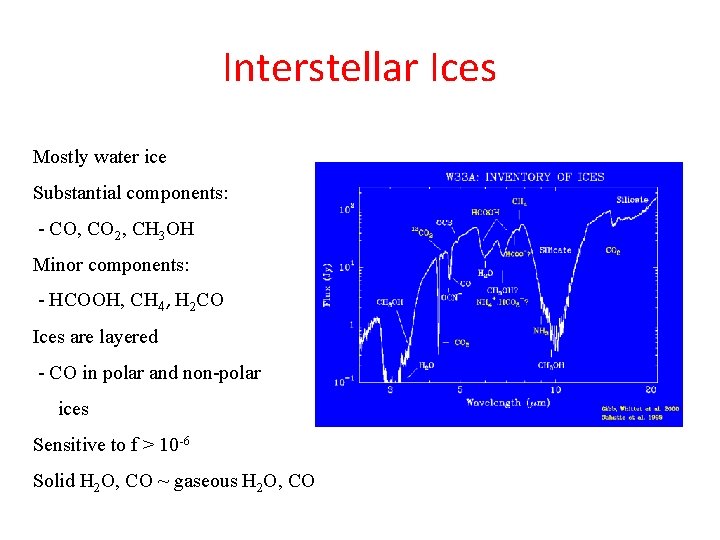
Interstellar Ices Mostly water ice Substantial components: - CO, CO 2, CH 3 OH Minor components: - HCOOH, CH 4, H 2 CO Ices are layered - CO in polar and non-polar ices Sensitive to f > 10 -6 Solid H 2 O, CO ~ gaseous H 2 O, CO
- Slides: 18