ISE Ion Selective Electrodes Prepared By Michigan Department

ISE Ion Selective Electrodes Prepared By Michigan Department of Environmental Quality Operator Training and Certification Unit
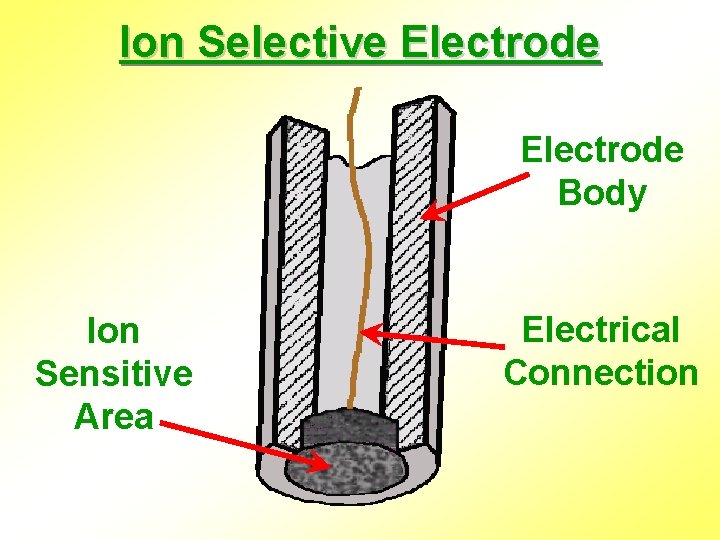
Ion Selective Electrode Body Ion Sensitive Area Electrical Connection
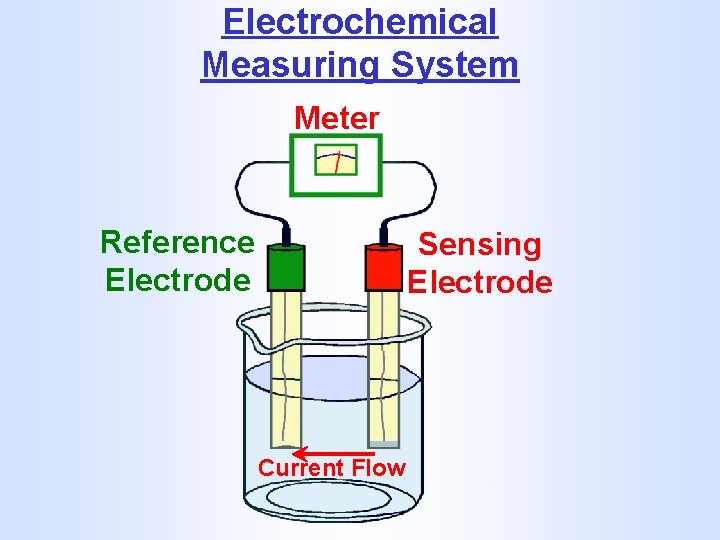
Electrochemical Measuring System Meter Reference Electrode Sensing Electrode Current Flow
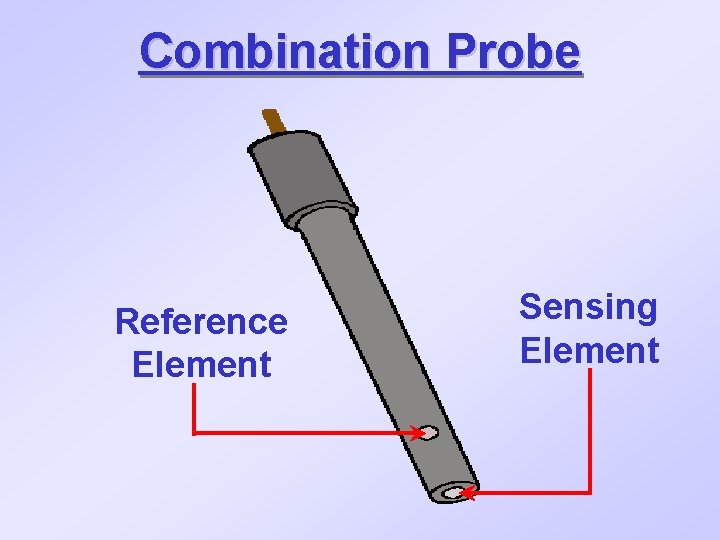
Combination Probe Reference Element Sensing Element
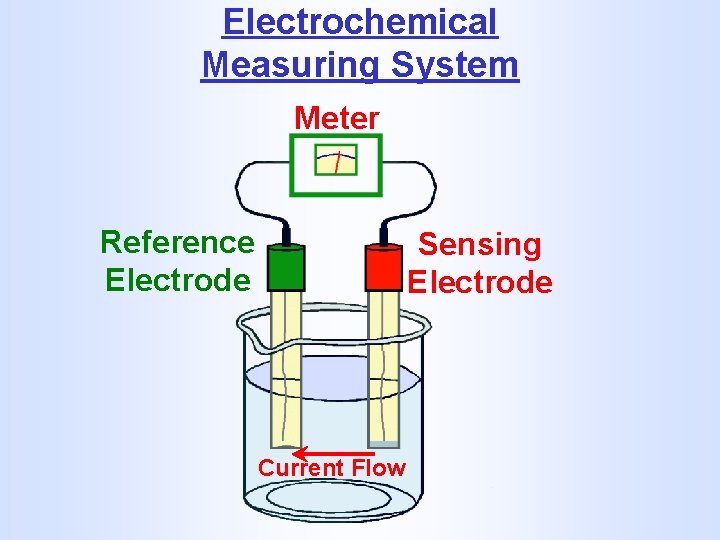
Electrochemical Measuring System Meter Reference Electrode Sensing Electrode Current Flow

NERNST EQUATION E= Eo T Log C + 2. 3 n E = Measured Voltage Eo = Reference Constant T = Temperature n = Charge on Ion = Ionic Strength C = Concentration

NERNST EQUATION E= Eo T Log C + 2. 3 n E = Measured Voltage Eo = Reference Constant T = Temperature n = Charge on Ion = Ionic Strength C = Concentration

NERNST EQUATION E= Eo T Log C + 2. 3 n E = Measured Voltage What the Meter Tells Us Eo = Reference Constant Meter Reading T = Temperature Also Affected By All This n = Charge on Ion = Ionic Strength C = Concentration What We Want To Know }

NERNST EQUATION E= Eo T Log C + 2. 3 n E = Measured Voltage What the Meter Tells Us Eo = Reference Constant Must Be T = Temperature Accounted For To Get True n = Charge on Ion Concentration = Ionic Strength C = Concentration What We Want To Know }

NERNST EQUATION E= Eo T Log C + 2. 3 n E = Measured Voltage What the Meter Tells Us Eo = Reference Constant Must Maintain T = Temperature Reference n = Charge on Ion Electrode = Ionic Strength C = Concentration What We Want To Know

NERNST EQUATION E= Eo T Log C + 2. 3 n E = Measured Voltage What the Meter Tells Us Will Be Eo = Reference Constant for T = Temperature Specific Ion, Whole Number, n = Charge on Ion 1, 2, 3, etc. + or = Ionic Strength C = Concentration What We Want To Know

NERNST EQUATION E= Eo T Log C + 2. 3 n E = Measured Voltage What the Meter Tells Us Eo = Reference Constant Must Be T = Temperature Controlled By Making It A Very n = Charge on Ion High Value = Ionic Strength C = Concentration What We Want To Know

NERNST EQUATION E= Eo T Log C + 2. 3 n E = Measured Voltage What the Meter Tells Us Eo = Reference Constant T = Temperature Add Ions ISAB n = Charge on Ion Na+ OH = Ionic Strength C = Concentration What We Want To Know

NERNST EQUATION E= Eo T Log C + 2. 3 n E = Measured Voltage What the Meter Tells Us Eo = Reference Constant Follow T = Temperature The Directions! n = Charge on Ion = Ionic Strength C = Concentration What We Want To Know

NERNST EQUATION E= Eo T Log C + 2. 3 n E = Measured Voltage What the Meter Tells Us Eo = Reference Constant Must Be T = Temperature Controlled n = Charge on Ion = Ionic Strength C = Concentration What We Want To Know

NERNST EQUATION E= Eo T Log C + 2. 3 n E = Measured Voltage Eo = Reference Constant T = Temperature n = Charge on Ion = Ionic Strength C = Concentration

NERNST EQUATION E= Eo T Log C + 2. 3 n y = mx + b T 2. 3 n = slope Slope is Direction of “Curve” When Plotting E vs. C
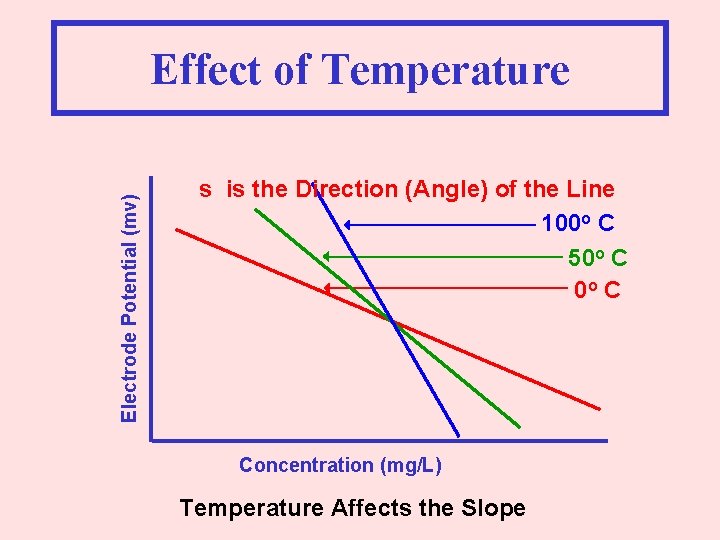
Electrode Potential (mv) Effect of Temperature s is the Direction (Angle) of the Line 100 o C 50 o C Concentration (mg/L) Temperature Affects the Slope
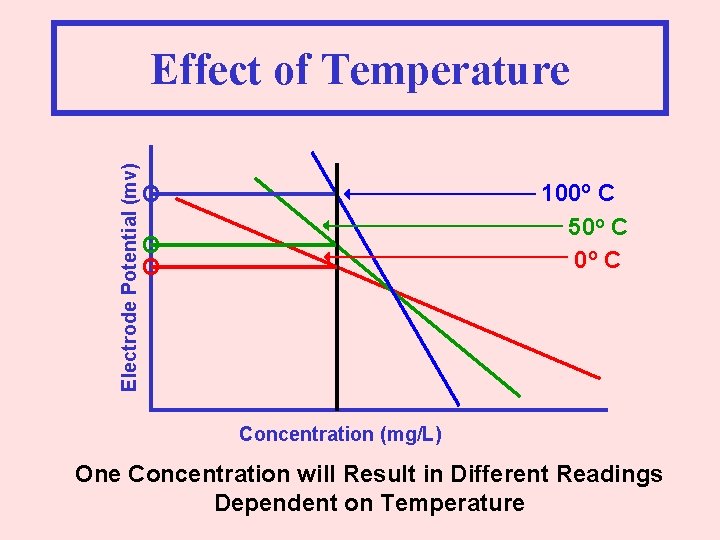
Electrode Potential (mv) Effect of Temperature 100 o C 50 o C O O O Concentration (mg/L) One Concentration will Result in Different Readings Dependent on Temperature
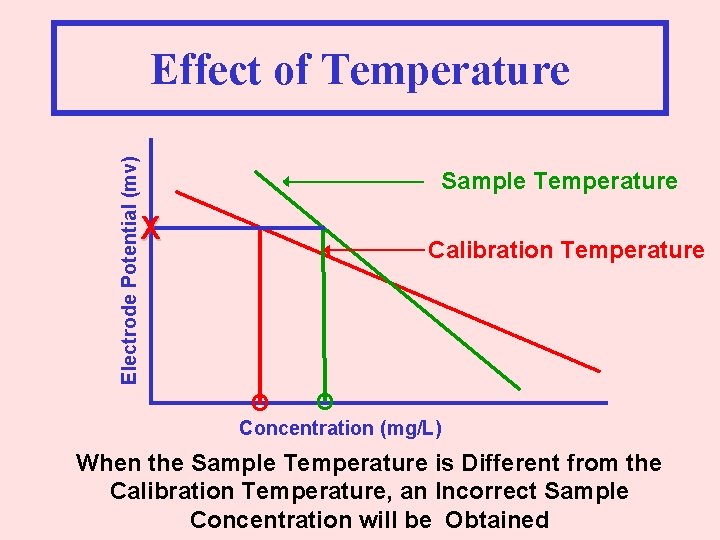
Electrode Potential (mv) Effect of Temperature Sample Temperature X Calibration Temperature O O Concentration (mg/L) When the Sample Temperature is Different from the Calibration Temperature, an Incorrect Sample Concentration will be Obtained
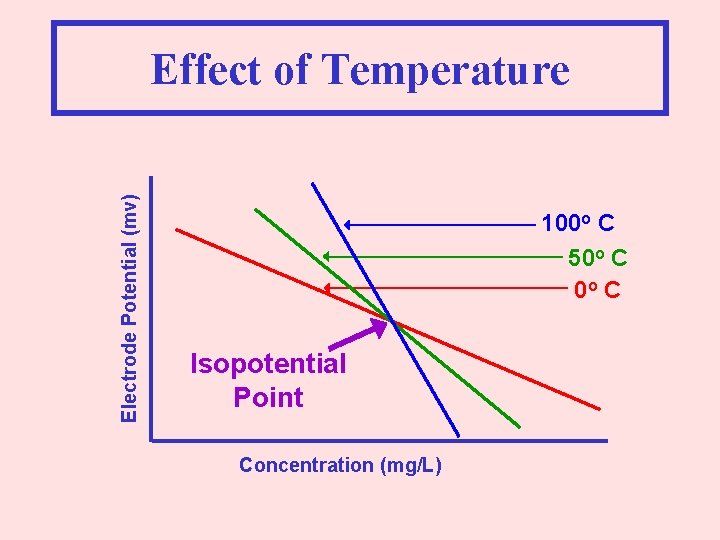
Electrode Potential (mv) Effect of Temperature 100 o C 50 o C Isopotential Point Concentration (mg/L)
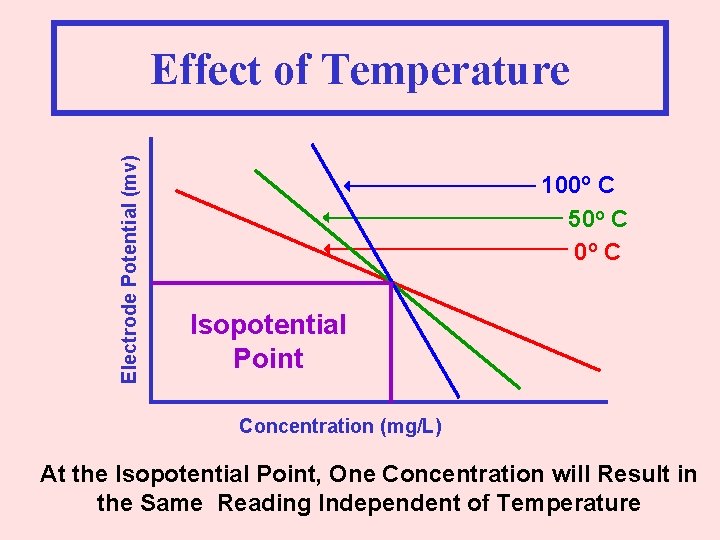
Electrode Potential (mv) Effect of Temperature 100 o C 50 o C Isopotential Point Concentration (mg/L) At the Isopotential Point, One Concentration will Result in the Same Reading Independent of Temperature
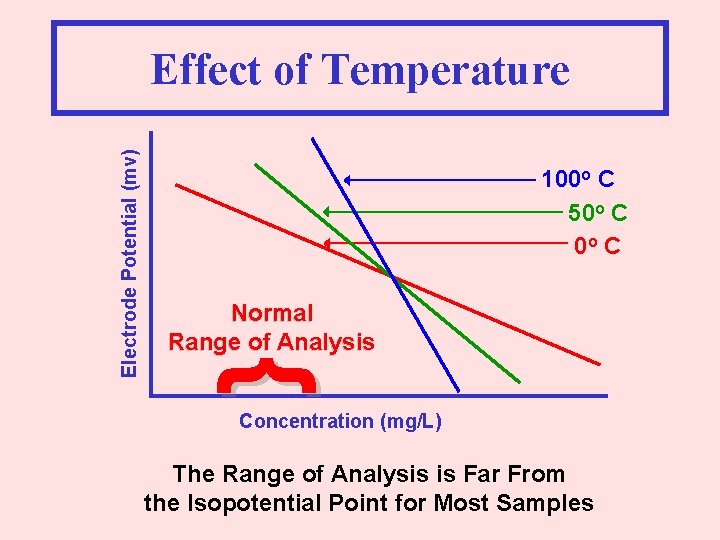
100 o C 50 o C Normal Range of Analysis } Electrode Potential (mv) Effect of Temperature Concentration (mg/L) The Range of Analysis is Far From the Isopotential Point for Most Samples
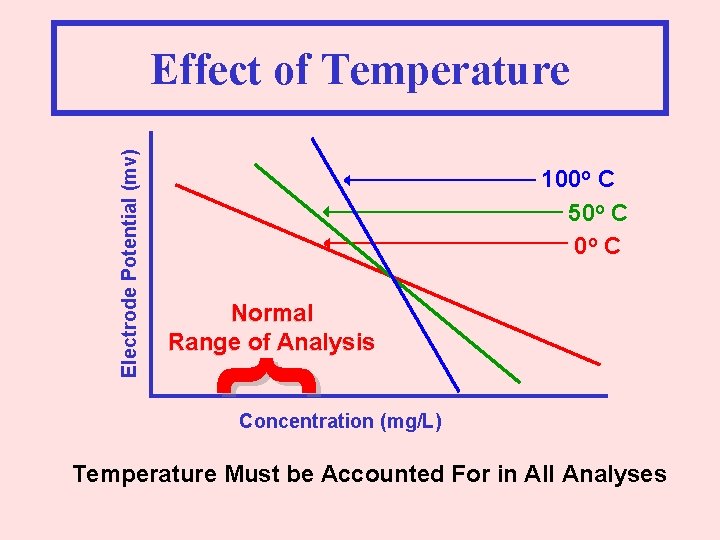
100 o C 50 o C Normal Range of Analysis } Electrode Potential (mv) Effect of Temperature Concentration (mg/L) Temperature Must be Accounted For in All Analyses

Automatic Temperature Compensation ATC Adjusts Slope in Relation to Temperature Sounds Good! Does Not Work!

Automatic Temperature Compensation ATC Adjusts Slope in Relation to Temperature Is Accurate Only When Analysis is Close to Isopotential Point Only For p. H

Automatic Temperature Compensation ATC (Except For p. H)
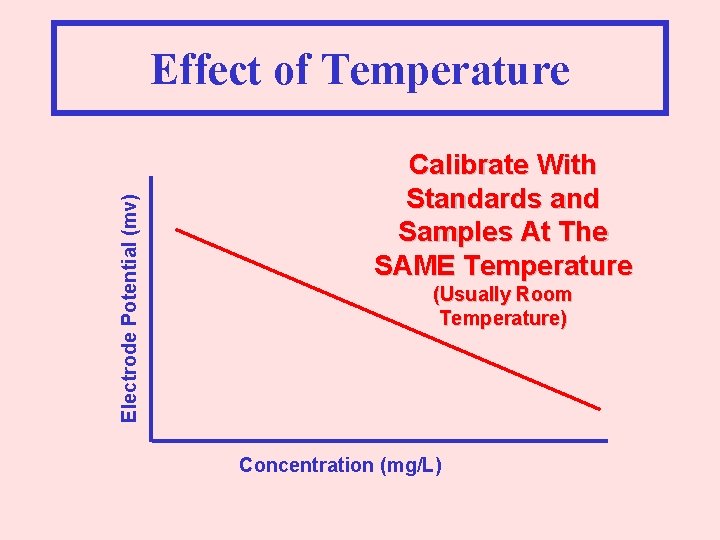
Electrode Potential (mv) Effect of Temperature Calibrate With Standards and Samples At The SAME Temperature (Usually Room Temperature) Concentration (mg/L)

Electrode Potential (mv) Calibration Control . Standard Solution Concentration (mg/L) The Calibration Control Adjusts the “Curve” to the Point of Standard Used
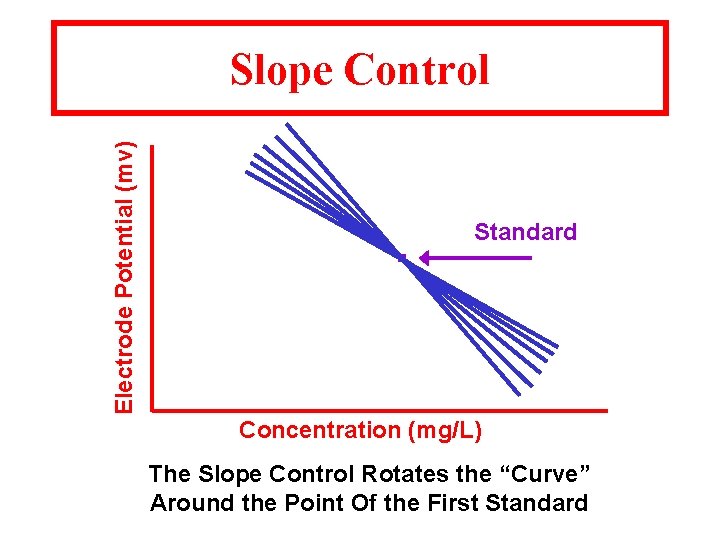
Electrode Potential (mv) Slope Control . Standard Concentration (mg/L) The Slope Control Rotates the “Curve” Around the Point Of the First Standard
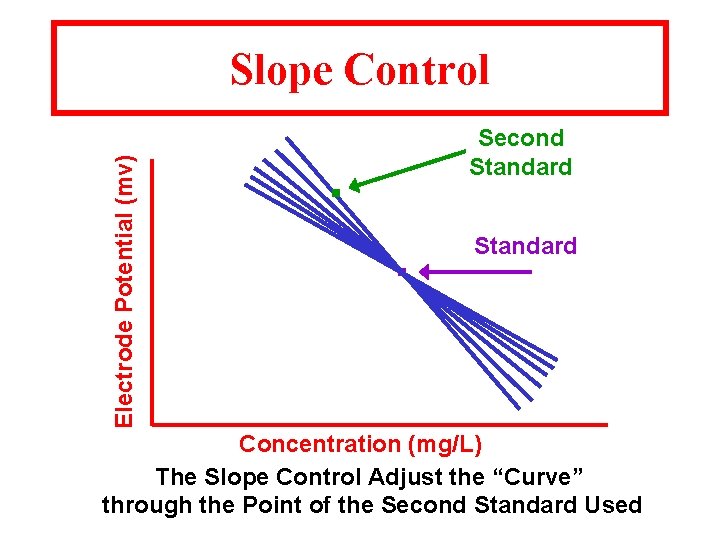
Electrode Potential (mv) Slope Control Second Standard . . Standard Concentration (mg/L) The Slope Control Adjust the “Curve” through the Point of the Second Standard Used

NERNST EQUATION o E 1 = E + s Log C 1 E 2 = Eo + s Log C 2 If C 2 = 10 x C 1 Then: E 2 = o E + s Log 10 C 1

NERNST EQUATION o E 1 = E + s Log C 1 E 2 = Eo + s Log C 2 If C 2 = 10 x C 1 E 2 - E 1 = s The Slope of an Electrode is the millivolt Change that is seen for a Ten Times Change in Concentration.

ISE Checking Electrode Slope Carefully Prepare Two Standards 10 X Concentration Difference Set Meter to Read in millivolts (or Relative mv) Record mv Reading for Each Standard Determine Difference = Slope
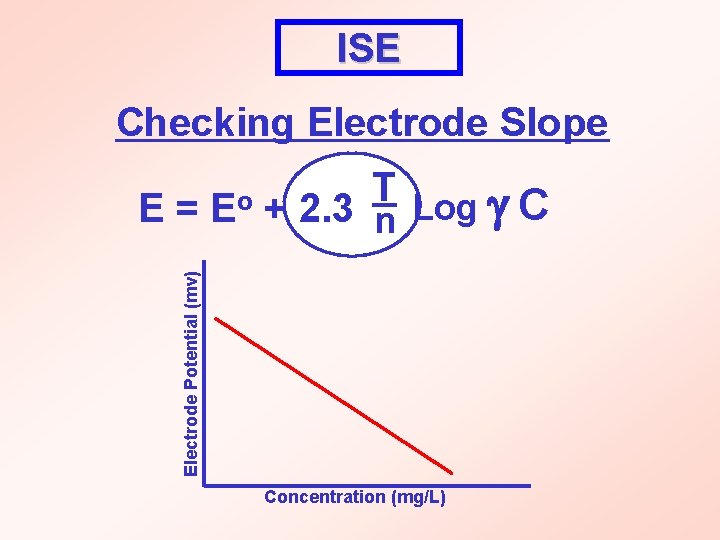
ISE Checking Electrode Slope T Log C + 2. 3 n Electrode Potential (mv) E= Eo Concentration (mg/L)

ISE Checking Electrode Slope E= Eo T Log C + 2. 3 n The Slope Value Determined is Affected By: Temperature (T) Charge on the Ion of Interest (n) Positive or Negative 1, 2, etc.

ISE Checking Electrode Slope E= Eo T Log C + 2. 3 n The Slope Value Determined is Affected By: Temperature (T) Charge on the Ion of Interest (n) Quality of the Standards Efficiency of the Electrode

ISE Checking Electrode Slope The Slope of the Electrode Will Change {Loss of Efficiency} The Slope Should be Checked Regularly to Assure Reliable Results Most Meters Give Slope When Calibrating ( In mv or %) Record Slope for QA/QC Daily (or at least every two weeks)

ISE Checking Electrode Slope Ideal Slope Depends on Ion of interest (n) Charge on the Ion of Interest (+ or -) Approximately 59 mv for n=1 or 29 mv for n=2 Check Manufacturer for Acceptable Slope Range Usually ± 10 % of Ideal

ISE Checking Electrode Slope If Determined Slope is Outside of Acceptable Range May Be Due To: Poor Quality of Standards Improper Probe Maintenance Faulty Electrode The Slope Should be Checked Regularly to Assure Reliable Results

ISE Ion Selective Electrodes Prepared By Michigan Department of Environmental Quality Operator Training and Certification Unit
- Slides: 41