ISARREACT 4 Discussion Deepak L Bhatt MD MPH

ISAR-REACT 4: Discussion Deepak L. Bhatt MD, MPH, FACC, FAHA Chief of Cardiology, VA Boston Healthcare System Director, Integrated Interventional Cardiovascular Program at Brigham and Women’s Hospital and the VA Boston Healthcare System Associate Professor of Medicine, Harvard Medical School Senior Investigator, TIMI Study Group

Disclosure for Dr. Bhatt Research grants from Amarin, Astra. Zeneca, Bristol-Myers Squibb, Eisai, Ethicon, Medtronic, Sanofi Aventis, The Medicines Company. This presentation discusses off-label and/or investigational uses of various drugs and devices.

Background • Majority of patients with NSTEMI undergo PCI, as the evidence supports • ISAR-REACT 2 showed that abciximab plus heparin was superior to heparin alone in PCI of NSTEMI patients • ACUITY showed that in NSTE-ACS, bivalirudin was noninferior to GPIIb/IIIa inhibitors plus heparin versus heparin alone, with significantly less major bleeding Bavry et al. AJM 2006. Kastrati et al. JAMA 2006. Stone et al. NEJM 2006.
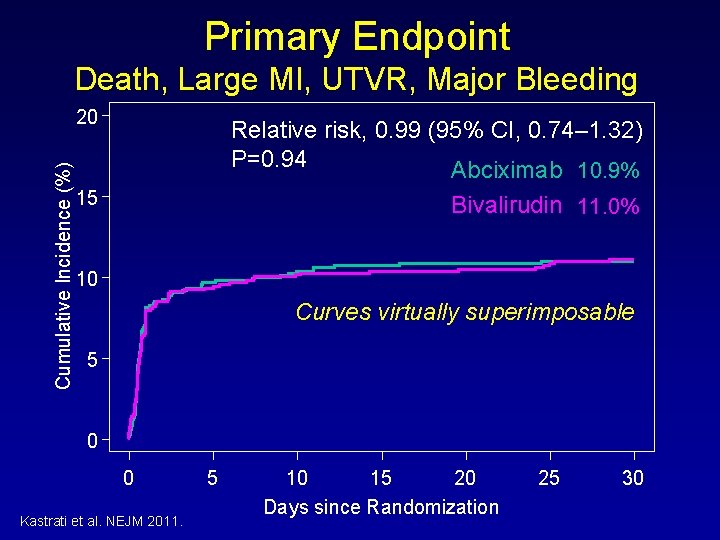
Primary Endpoint Death, Large MI, UTVR, Major Bleeding Cumulative Incidence (%) 20 Relative risk, 0. 99 (95% CI, 0. 74– 1. 32) P=0. 94 Abciximab 10. 9% 15 Bivalirudin 11. 0% 10 Curves virtually superimposable 5 0 0 Kastrati et al. NEJM 2011. 5 10 15 20 Days since Randomization 25 30
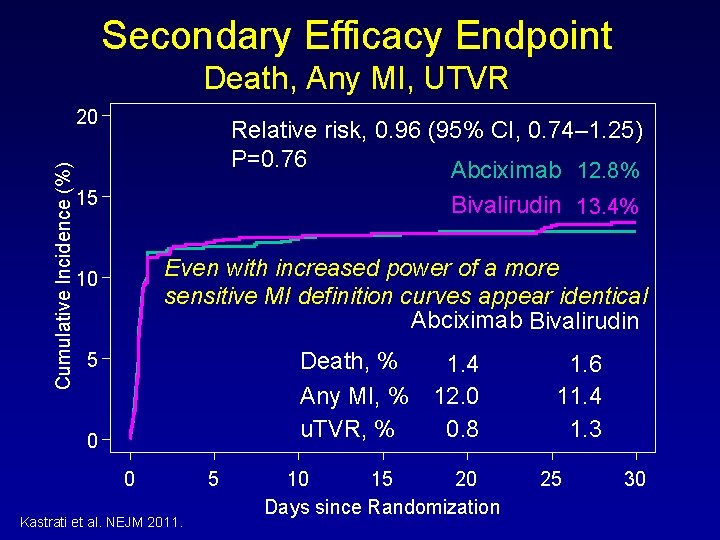
Secondary Efficacy Endpoint Death, Any MI, UTVR Cumulative Incidence (%) 20 Relative risk, 0. 96 (95% CI, 0. 74– 1. 25) P=0. 76 Abciximab 12. 8% 15 Bivalirudin 13. 4% Even with increased power of a more sensitive MI definition curves appear identical Abciximab Bivalirudin 10 Death, % 1. 4 Any MI, % 12. 0 u. TVR, % 0. 8 5 0 0 Kastrati et al. NEJM 2011. 5 10 15 20 Days since Randomization 1. 6 11. 4 1. 3 25 30
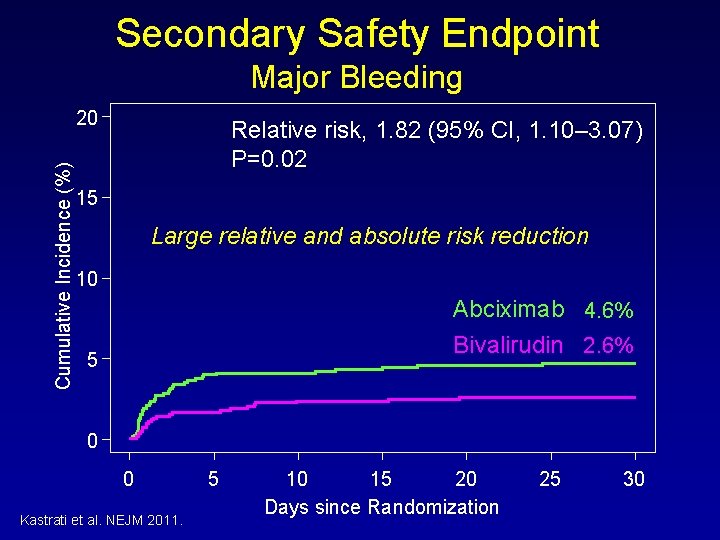
Secondary Safety Endpoint Major Bleeding Cumulative Incidence (%) 20 Relative risk, 1. 82 (95% CI, 1. 10– 3. 07) P=0. 02 15 Large relative and absolute risk reduction 10 Abciximab 4. 6% Bivalirudin 2. 6% 5 0 0 Kastrati et al. NEJM 2011. 5 10 15 20 Days since Randomization 25 30

ISAR-REACT 4: Strengths • Rigorous double-blind, double-dummy design (ACUITY was open-label, which - in theory - could affect ascertainment of non-fatal endpoints) • Comparison was against abciximab (which some still consider the gold standard GPIIb/IIIa inhibitor) • Strict definition of bleeding (intracranial, intraocular, or retroperitoneal; Hb decrease >4 g/d. L plus either overt bleeding or need for transfusion of 2 or more units) – difficult to argue with the importance of that

ISAR-REACT 4: Caveats • Patients pretreated with ASA + clopidogrel 600 mg – Results may not apply to ASA/clop untreated • Prasugrel or ticagrelor not used – Difficult to see how this would change the results • Bleeding advantage would have been attenuated if more radial cases were performed – But still numerically lower pericardial, GI, and GU bleeds, and no apparent loss of efficacy

Conclusions • In patients pretreated w/ ASA and clopidogrel 600 mg, – Bivalirudin results in less major bleeding than heparin + abciximab; also lower rate of severe thrombocytopenia – Similar efficacy – Shorter duration infusion – Likely lower cost • Coupled with HORIZONS-AMI, data from ISAR-REACT 4 support use of bivalirudin during PCI across the full spectrum of ACS
- Slides: 9