Is the Developing World the Answer Unethical clinical





























- Slides: 39

“Is the Developing World the Answer? ” Unethical clinical trials in the Third World Sammy Almashat, MD, MPH Sidney Wolfe, MD Public Citizen’s Health Research Group

Outline • History and structure of human experimentation • The commercialization of human experimentation: conflicts of interest abound • A story of self-regulation and the honor code • Globalization of clinical trials: “Is the developing world the answer? ” • Case studies of double-standards for the Third World and efforts to stop unethical trials

Clinical Trials • Thousands of trials of pharmaceuticals and medical devices conducted on tens of thousands of people every year • Human testing necessary to ensure safe and effective products prior to marketing • The question is: who’s doing the testing on whom, and who’s watching them?

The Birth of a Drug Test tube (in vitro) testing Animal testing Human testing Phase 1 • Few healthy patients, testing safety Phase 2 • First study with sick patients, testing safety and efficacy Phase 3 • Largest trials with sick patients, testing safety and efficacy

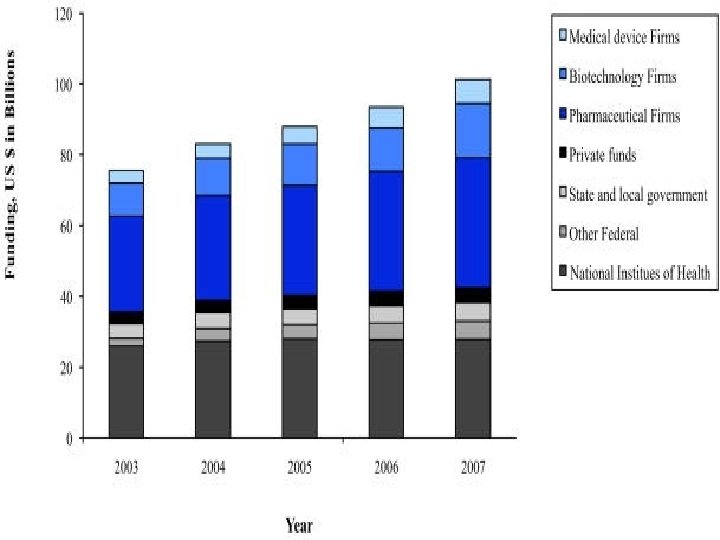
Time is Money • Drug development is risky, expensive, and time-consuming • Average drug reaches market 7 -10 years after first tests at a cost of anywhere from $55 million to $800 million per drug • Incentive is high to cut costs (and corners) and speed up the process

The Economics of Drug Development • Human Experimentation Corporations (HECs) – Started in the 1970 s as small companies specializing in biopharmaceutical (bench) testing – Grew dramatically in the past few decades and moved into clinical (human) research, growing to a $20 billion industry by 2008, with annual growth rates of 15 -17% • The IRB industry – Paralleled growth of HECs – Increasingly for-profit – “IRB-shopping”


Birth of Research Ethics • Atrocities of human experimentation in the 20 th century prompted the development of universal ethical principles to govern all such research in the future

Universal Principles in Human Subjects Research • • • What do we mean by “unethical”? Nuremburg Code Declaration of Helsinki Belmont Report and Common Rule (U. S. ) FDA (abandoned Helsinki in 2008)

Universal Principles in Human Subjects Research • • Informed consent Truly voluntary (not coerced) participation Beneficence Equity and justice

Clinical trial oversight X U. S. /Host Govt. X X IRBs Drug Company/HECs

2009 – GAO goes undercover • Fake IRB • Fake medical device company with fake device • Real medical device and IRB companies – and the Feds – all took the bait

“Globalization” of the Clinical Trial Industry

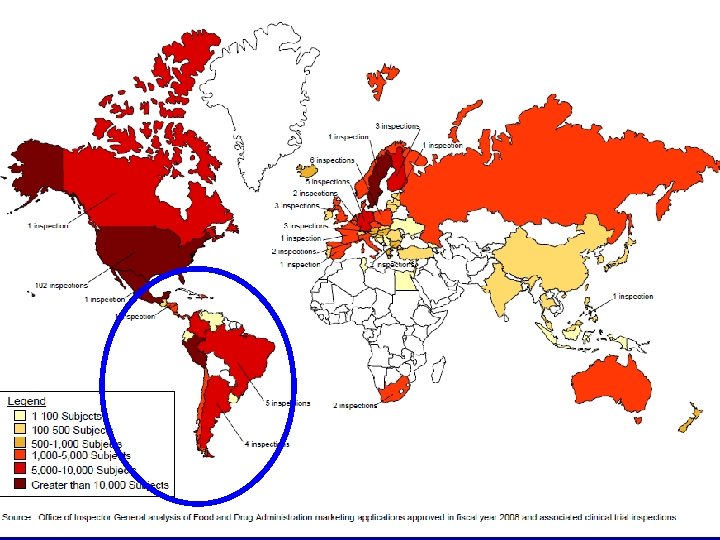
Clinical Trials in the Developing World



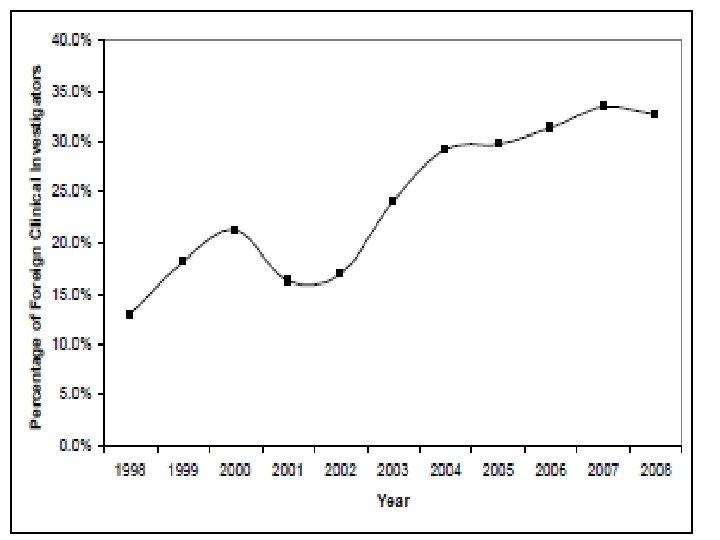
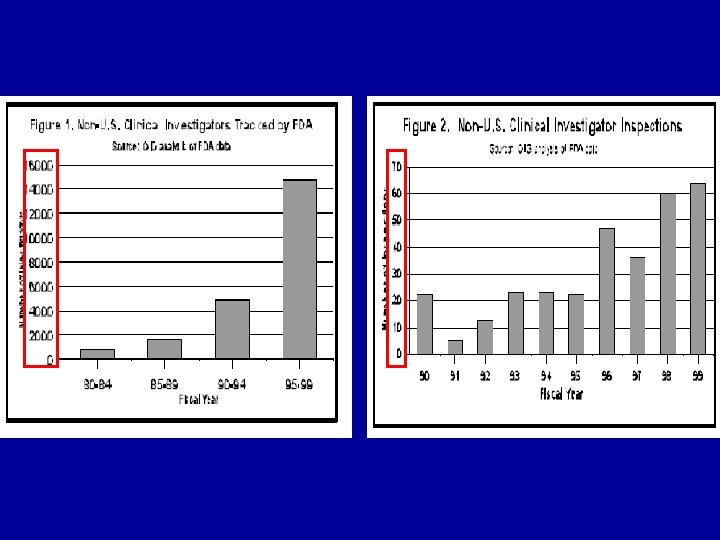
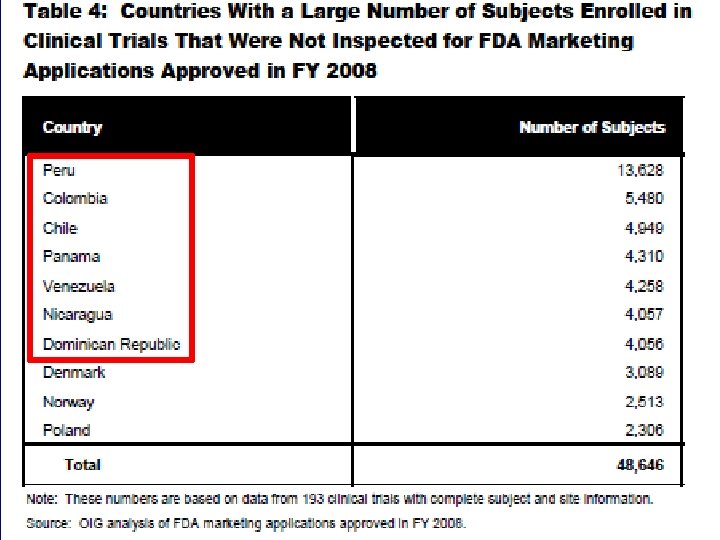
Globalization of Clinical Trials • 80% of drugs are now approved based on data from foreign clinical trials • Over half of clinical trial subjects and sites are located outside the United States • One-third of trial investigators are now from foreign sites

Why are companies moving abroad? • Cost • Speed – The “patient advantage” • Escaping regulatory “burdens”




Special considerations with vulnerable populations Exploitation is almost inherent in these trials - Lack of informed consent - Direct and indirect coercion - Lack of Equity - Lack of Recourse - Companies are not liable for intentional let alone unintentional harm caused by experimental drugs (certainly not liable for NOT giving effective treatment to a placebo group).

Case Studies: Double standards for the Third World

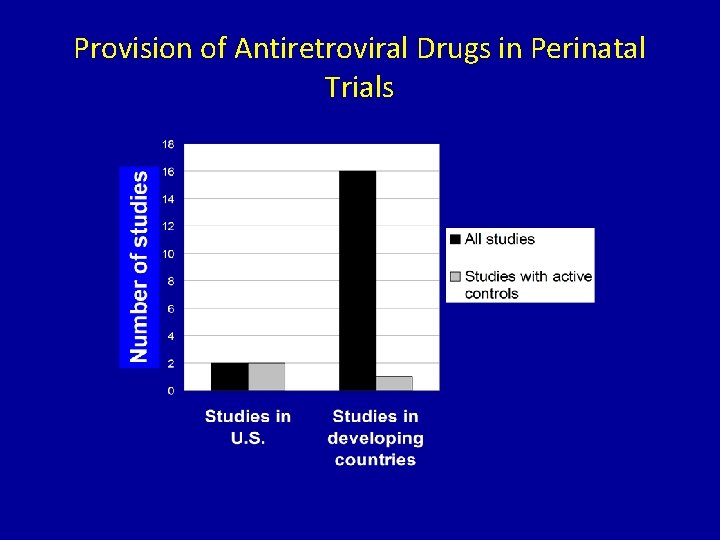
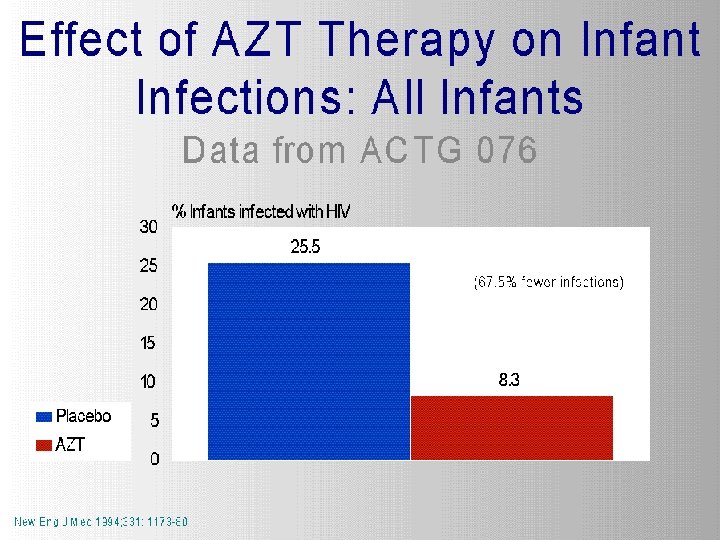
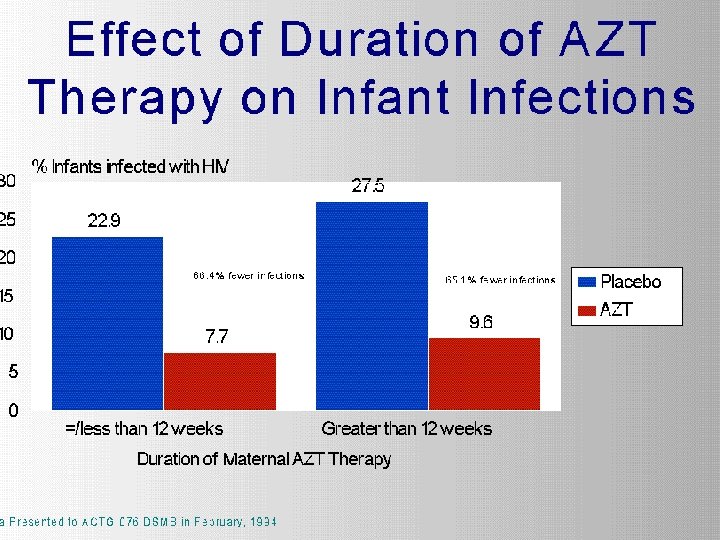
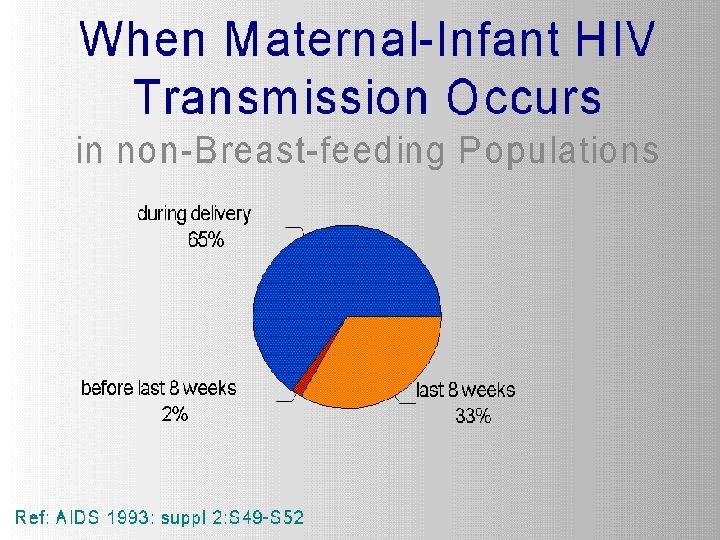
Case Study: Unethical HIV trials • 1997: Nine U. S. government-funded studies of pregnant women in Africa, Asia, and the Caribbean • Half the women in these trials received AZT, a therapy proven to help prevent HIV transmission to the fetus • The other half received ineffectual placebos


Provision of Antiretroviral Drugs in Perinatal Trials





Case Study: The Surfaxin trial • Title of internal FDA meeting: “Use of placebo-controls in life threatening diseases: is the developing world the answer? ” • Location: Mexico, Peru, Bolivia, Ecuador • Design: Surfaxin vs. placebo (vs. approved surfactant)

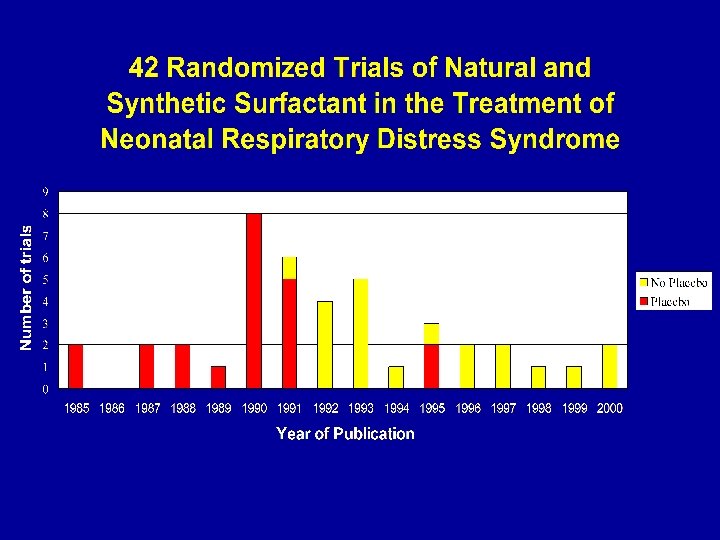
Case Study: The Surfaxin trial • Discovery Laboratories (Johnson & Johnson), Doylestown, PA • Synthetic surfactant (Surfaxin) • 4 surfactants on the market (1 st in 1990) • Associated with 34% relative reduction in neonatal mortality (Cochrane meta-analysis) “Without doubt the most thoroughly studied new therapy in neonatal care” (NEJM review)

Case Study: The Surfaxin trial • “Further placebo controlled trials of synthetic surfactant are no longer warranted. ” (Cochrane) • FDA: “Conduct of a placebo controlled surfactant trial for premature infants with RDS is considered unethical in the USA. ” • European trial: Surfaxin vs. approved surfactant


Case Study: The Surfaxin trial • February 2001: Public Citizen writes to HHS Secretary Tommy Thompson • March 2001: Bolivian health ministry says the study is “totally prohibited” for legal, ethical and social reasons • April 2001: Discovery announces study changed to compare to known effective surfactant

Advertisement by Human Experimentation Corporation (HEC) Quintiles to the Pharmaceutical Industry: January, 1998


What needs to be done • More oversight • Addressing conflicts of interest • Tying in with more high-profile Third World health justice causes (e. g. access to medicines)

www. citizen. org/hrgpublications