Is Hepatitis B Curable Marion G Peters MD

Is Hepatitis B Curable? Marion G. Peters, MD Professor of Medicine Chief of Hepatology Research University of California San Francisco, California

Financial Relationships With Commercial Entities Dr Peters has served as an advisor to Abbott. Her spouse is employed by Hoffman-La Roche (Updated 04/29/19) Slide 2 of 48

Learning Objectives After attending this presentation, learners will be able to: • List the limitations of current therapies for HBV • Describe types of HBV cure • Describe HBV latency • List mechanisms of action of putative new therapies for HBV Slide 3 of 48

ARS 1: HBV status in 2019: which is true? 1. Acute HBV in adults leads to loss of HBs. Ag usually 2. HBV infection can be cured 3. Currently available drugs lead to loss of HBs. Ag usually 4. HBV is not a latent virus Slide 4 of 48

ARS 2: New drugs in Phase I/II: which is false? 1. Target Hepatitis B core protein 2. Target HBV entry into hepatocyte 3. Target ccc. DNA in nucleus 4. Target HBV m. RNA 5. Target HBV secretion Slide 5 of 48
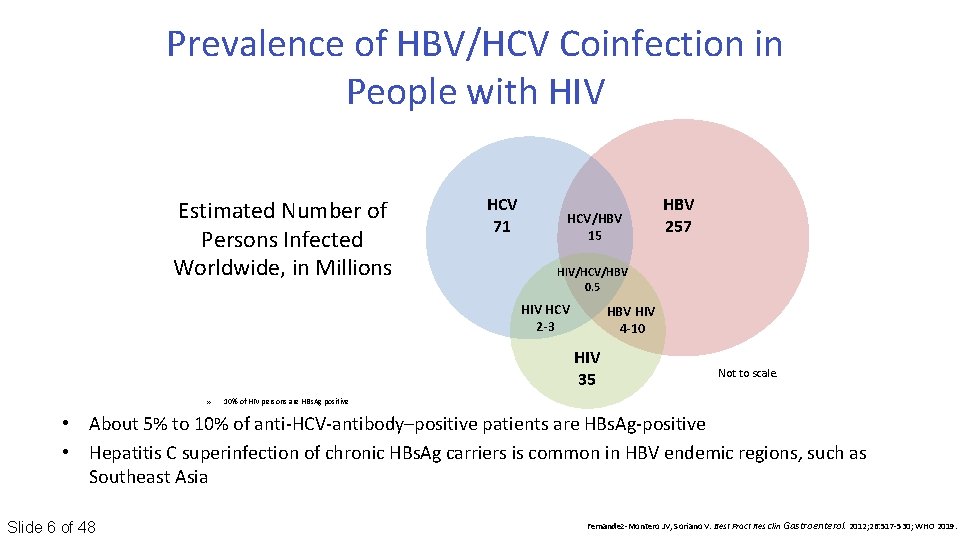
Prevalence of HBV/HCV Coinfection in People with HIV Estimated Number of Persons Infected Worldwide, in Millions HCV 71 HCV/HBV 15 HIV/HCV/HBV 0. 5 HIV HCV 2 -3 HBV HIV 4 -10 HIV 35 » HBV 257 Not to scale. 10% of HIV persons are HBs. Ag positive • About 5% to 10% of anti‐HCV‐antibody–positive patients are HBs. Ag‐positive • Hepatitis C superinfection of chronic HBs. Ag carriers is common in HBV endemic regions, such as Southeast Asia Slide 6 of 48 Fernandez‐Montero JV, Soriano V. Best Pract Res Clin Gastroenterol. 2012; 26: 517‐ 530; WHO 2019.

HBV is a life long, dynamic disease • Changes over time • Risk of end stage liver disease and cancer increases with ongoing inflammation and viremia in adults • Fibrosis can be reversible • Drugs can decrease fibrosis progression • HBV can be controlled but not cured • Reactivation can occur even in those who have lost HBs. Ag • HBV infection in neonates and young children leads to chronicity >90‐ 95% • HBV infection in adults (HIV) leads to chronicity <5% (~20%) Slide 7 of 48

HBV Control • Inflammatory: normalize serum ALT, biopsy • Virologic: decrease HBV DNA • Immune: seroconversion – HBe. Ag to anti‐HBe – HBs. Ag to anti‐HBs • HBV as of 2019 not “cured” but controlled Slide 8 of 48

Approved HBV treatments 2019 • • Slide 9 of 48 Interferon alfa‐ 2 b – 1991 Lamivudine – 1998 Adefovir – 2002 Entecavir – 2005 Peginterferon alfa‐ 2 a – 2005 Telbivudine – 2006 Tenofovir Disoproxil– 2008 Tenofovir alafenamide‐ 2017
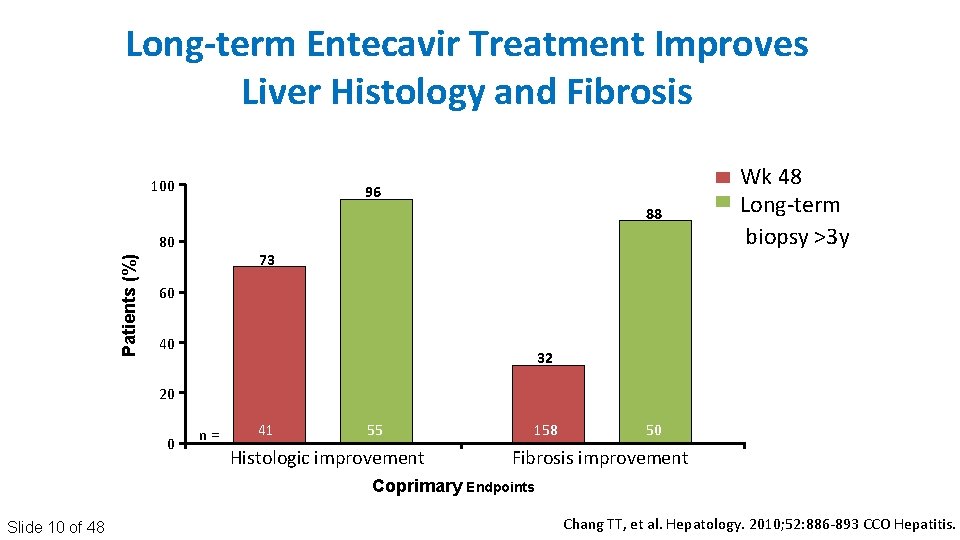
Long-term Entecavir Treatment Improves Liver Histology and Fibrosis 100 96 88 Patients (%) 80 73 Wk 48 Long‐term biopsy >3 y 60 40 32 20 0 n = 41 55 Histologic improvement 158 50 Fibrosis improvement Coprimary Endpoints Slide 10 of 48 Chang TT, et al. Hepatology. 2010; 52: 886‐ 893 CCO Hepatitis.
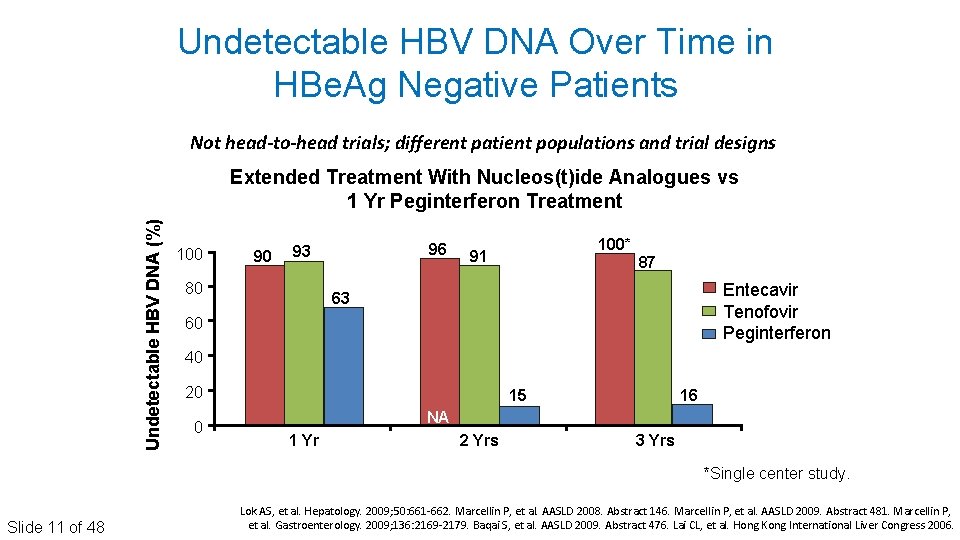
Undetectable HBV DNA Over Time in HBe. Ag Negative Patients Not head-to-head trials; different patient populations and trial designs Undetectable HBV DNA (%) Extended Treatment With Nucleos(t)ide Analogues vs 1 Yr Peginterferon Treatment 100 90 96 93 80 100* 91 87 Entecavir Tenofovir Peginterferon 63 60 40 20 0 15 16 NA 1 Yr 2 Yrs 3 Yrs *Single center study. Slide 11 of 48 Lok AS, et al. Hepatology. 2009; 50: 661‐ 662. Marcellin P, et al. AASLD 2008. Abstract 146. Marcellin P, et al. AASLD 2009. Abstract 481. Marcellin P, et al. Gastroenterology. 2009; 136: 2169‐ 2179. Baqai S, et al. AASLD 2009. Abstract 476. Lai CL, et al. Hong Kong International Liver Congress 2006.
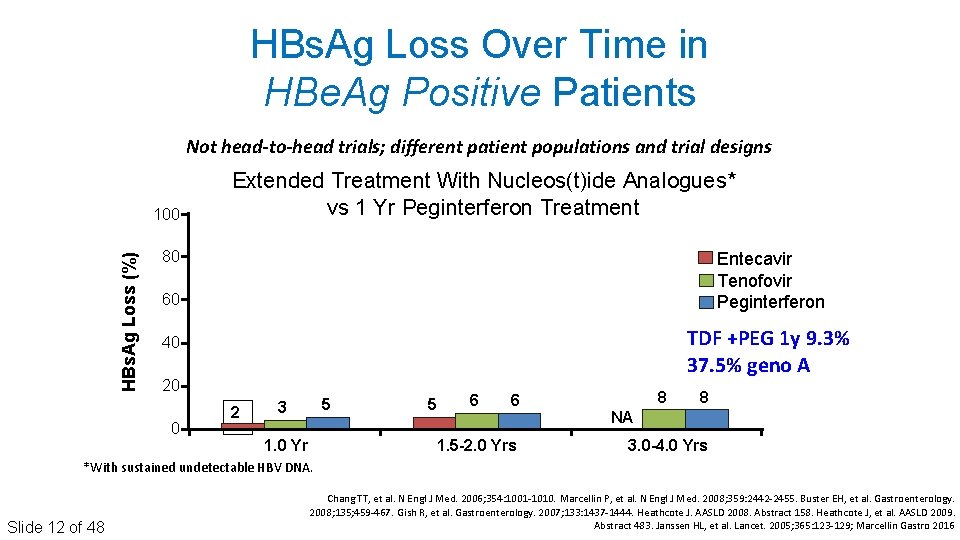
HBs. Ag Loss Over Time in HBe. Ag Positive Patients Not head-to-head trials; different patient populations and trial designs HBs. Ag Loss (%) 100 Extended Treatment With Nucleos(t)ide Analogues* vs 1 Yr Peginterferon Treatment 80 Entecavir Tenofovir Peginterferon 60 TDF +PEG 1 y 9. 3% 37. 5% geno A 40 20 0 2 5 3 1. 0 Yr 5 6 6 1. 5 -2. 0 Yrs 8 8 NA 3. 0 -4. 0 Yrs *With sustained undetectable HBV DNA. Slide 12 of 48 Chang TT, et al. N Engl J Med. 2006; 354: 1001‐ 1010. Marcellin P, et al. N Engl J Med. 2008; 359: 2442‐ 2455. Buster EH, et al. Gastroenterology. 2008; 135; 459‐ 467. Gish R, et al. Gastroenterology. 2007; 133: 1437‐ 1444. Heathcote J. AASLD 2008. Abstract 158. Heathcote J, et al. AASLD 2009. Abstract 483. Janssen HL, et al. Lancet. 2005; 365: 123‐ 129; Marcellin Gastro 2016
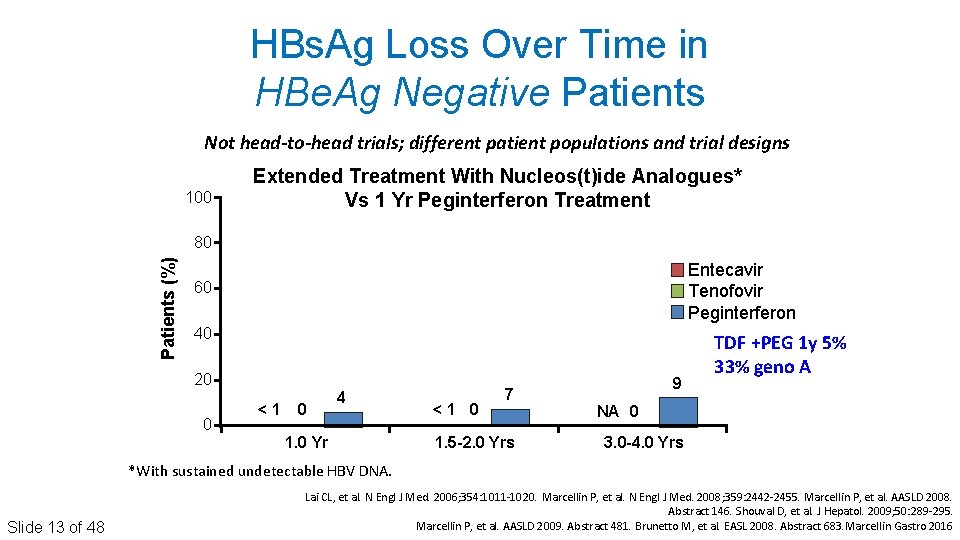
HBs. Ag Loss Over Time in HBe. Ag Negative Patients Not head-to-head trials; different patient populations and trial designs 100 Extended Treatment With Nucleos(t)ide Analogues* Vs 1 Yr Peginterferon Treatment Patients (%) 80 Entecavir Tenofovir Peginterferon 60 40 20 0 <1 0 4 1. 0 Yr <1 0 7 1. 5 -2. 0 Yrs 9 TDF +PEG 1 y 5% 33% geno A NA 0 3. 0 -4. 0 Yrs *With sustained undetectable HBV DNA. Slide 13 of 48 Lai CL, et al. N Engl J Med. 2006; 354: 1011‐ 1020. Marcellin P, et al. N Engl J Med. 2008; 359: 2442‐ 2455. Marcellin P, et al. AASLD 2008. Abstract 146. Shouval D, et al. J Hepatol. 2009; 50: 289‐ 295. Marcellin P, et al. AASLD 2009. Abstract 481. Brunetto M, et al. EASL 2008. Abstract 683. Marcellin Gastro 2016
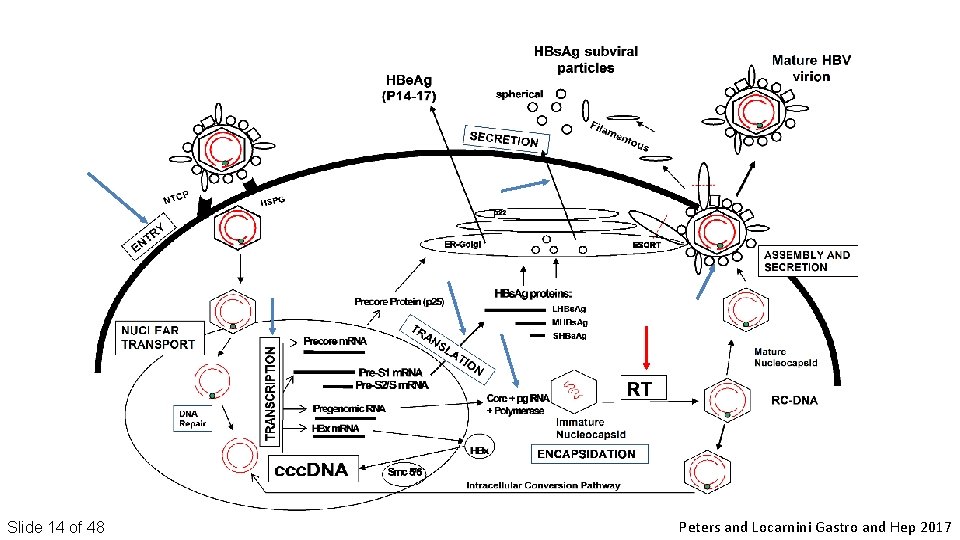
Slide 14 of 48 Peters and Locarnini Gastro and Hep 2017
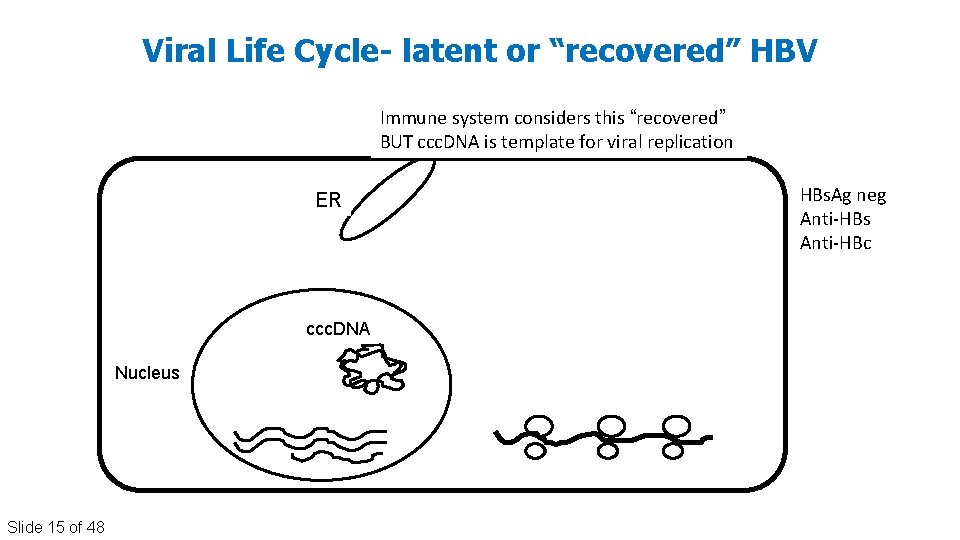
Viral Life Cycle- latent or “recovered” HBV Immune system considers this “recovered” BUT ccc. DNA is template for viral replication ER ccc. DNA Nucleus Slide 15 of 48 HBs. Ag neg Anti‐HBs Anti‐HBc

Types of HBV cure Functional Cure- clinical resolution Sustained, off drug: • No inflammation: ALT and liver biopsy • HBs. Ag loss • +/‐ anti‐HBs gain Complete cure- virological cure • All of above plus • Loss of ccc. DNA in liver Inactive state -an interim goal • No inflammation: ALT and liver biopsy • HBV DNA low or u/d • HBs. Ag positive Slide 16 of 48

Slide 17 of 48 Peters and Locarnini Gastro and Hep 2017

Strategies to Eradicate HBV Host immune approaches Virologic approaches • • Slide 18 of 48 Entry inhibitors Block ccc. DNA Transcription inhibitors RNA interference HBV capsid inhibitor Polymerase inhibitors Secretion inhibitors • • • Interferons RIG‐I agonists TLR‐ 7 PD‐ 1/ PDL‐ 1 IL‐ 7 Therapeutic vaccines – – – Immune complex vaccines Nasal HBV (NASVAC) vaccines DNA vaccines T cell vaccines Adenovirus based vaccines (TG 1050) Yeast based vaccines

Strategies to Eradicate HBV Host immune approaches Virologic approaches • • Slide 19 of 48 Entry inhibitors Block ccc. DNA Transcription inhibitors RNA interference HBV capsid inhibitor Polymerase inhibitors Secretion inhibitors • • • Interferons RIG‐I agonists TLR‐ 7 PD‐ 1/ PDL‐ 1 IL‐ 7 Therapeutic vaccines – – – Immune complex vaccines Nasal HBV (NASVAC) vaccines DNA vaccines T cell vaccines Adenovirus based vaccines (TG 1050) Yeast based vaccines
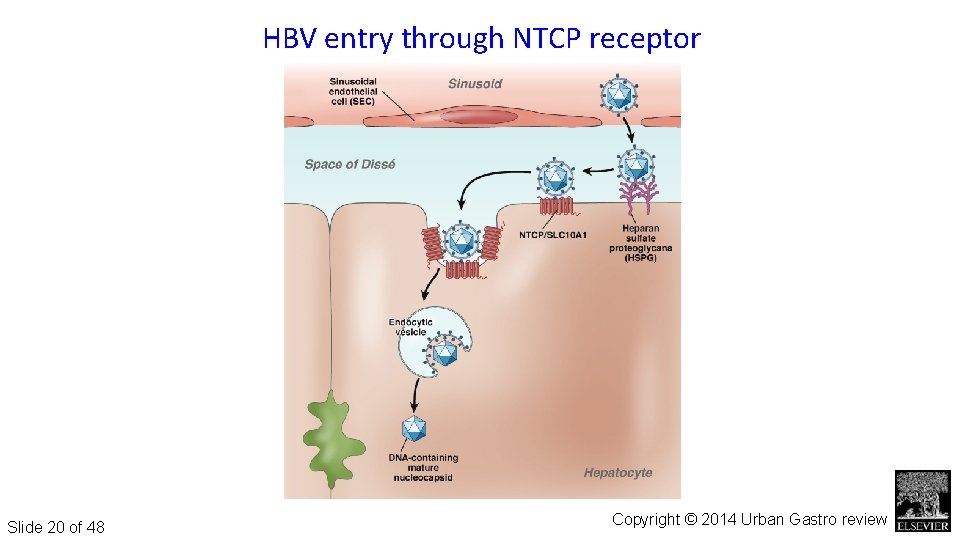
Figure 6 HBV entry through NTCP receptor Slide 20 of 48 Gastroenterology 2014 147, 48 -64 DOI: (10. 1053/j. gastro. 2014. 030) Copyright © 2014 Urban Gastro review
HBV Targeting cell entry Small molecule compounds binding to Sodium taurocholate cotransporting polypeptide (NTCP) • HBV pre-S 1 -derived lipopeptide Myrcludex-B competes with HBV/HDV for binding to NTCP – Prevents HBV/HDV entry – Blocks entry at p. M concentrations increased serum bile acids – Stops new infection of hepatocytes Slide 21 of 48 Urban AASLD 2016; Gastro 2014 Zeisel Gut 2015
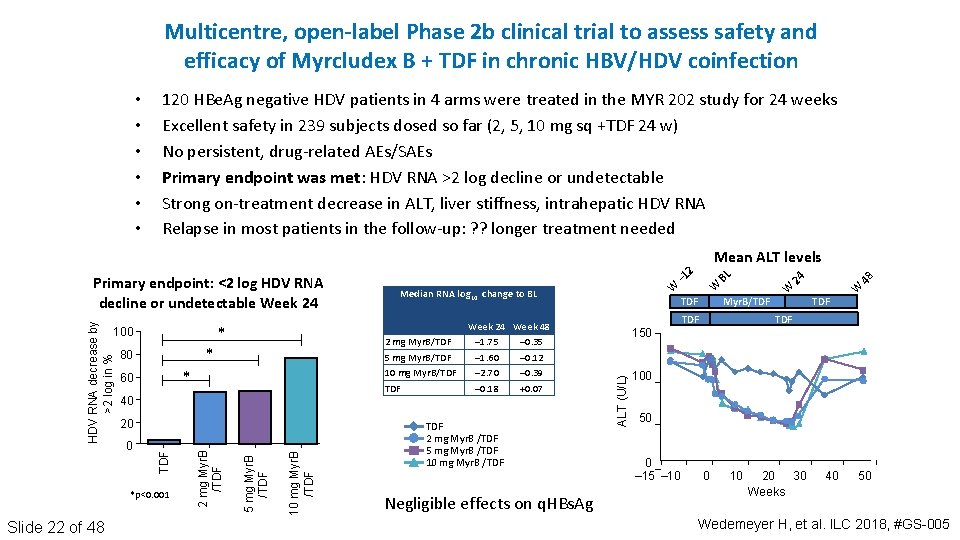
Multicentre, open-label Phase 2 b clinical trial to assess safety and efficacy of Myrcludex B + TDF in chronic HBV/HDV coinfection • • • 120 HBe. Ag negative HDV patients in 4 arms were treated in the MYR 202 study for 24 weeks Excellent safety in 239 subjects dosed so far (2, 5, 10 mg sq +TDF 24 w) No persistent, drug‐related AEs/SAEs Primary endpoint was met: HDV RNA >2 log decline or undetectable Strong on‐treatment decrease in ALT, liver stiffness, intrahepatic HDV RNA Relapse in most patients in the follow‐up: ? ? longer treatment needed * 80 40 10 mg Myr. B /TDF 2 mg Myr. B /TDF 0 TDF 20 Week 24 Week 48 – 1. 75 – 0. 35 – 1. 60 – 0. 12 – 2. 70 – 0. 39 – 0. 18 +0. 07 TDF 2 mg Myr. B /TDF 5 mg Myr. B /TDF 10 mg Myr. B /TDF Negligible effects on q. HBs. Ag Myr. B/TDF W 4 8 W 2 4 W B L W – 1 TDF 2 mg Myr. B/TDF 5 mg Myr. B/TDF 10 mg Myr. B/TDF * 60 *p<0. 001 Slide 22 of 48 * Median RNA log 10 change to BL TDF 150 ALT (U/L) 100 5 mg Myr. B /TDF HDV RNA decrease by >2 log in % Primary endpoint: <2 log HDV RNA decline or undetectable Week 24 2 Mean ALT levels 100 50 0 – 15 – 10 0 10 20 30 Weeks 40 50 Wedemeyer H, et al. ILC 2018, #GS-005
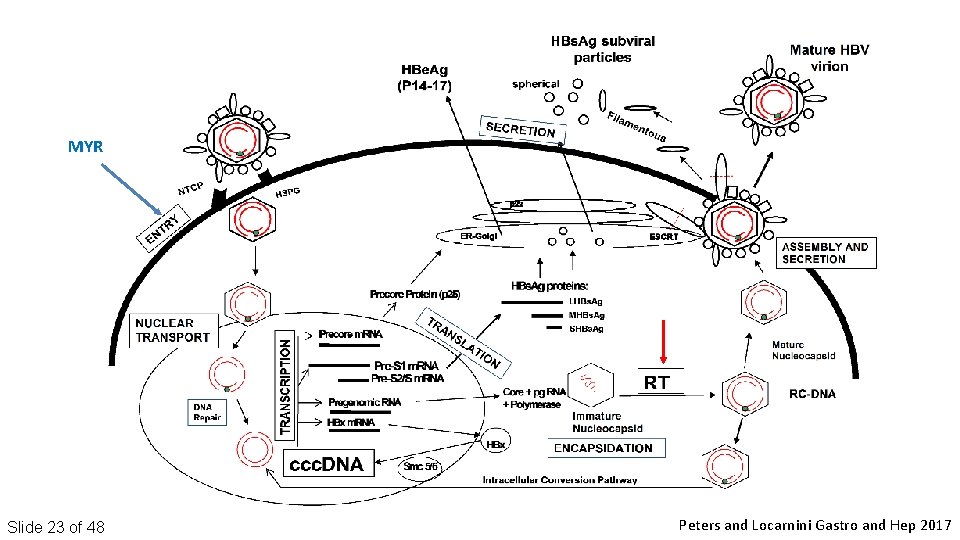
MYR Slide 23 of 48 Peters and Locarnini Gastro and Hep 2017

Strategies to Eradicate HBV Host immune approaches Virologic approaches • • Slide 24 of 48 Entry inhibitors Block ccc. DNA Transcription inhibitors RNA interference HBV capsid inhibitor Polymerase inhibitors Secretion inhibitors • • • Interferons RIG‐I agonists TLR‐ 7 PD‐ 1/ PDL‐ 1 IL‐ 7 Therapeutic vaccines – – – Immune complex vaccines Nasal HBV (NASVAC) vaccines DNA vaccines T cell vaccines Adenovirus based vaccines (TG 1050) Yeast based vaccines

ccc. DNA inhibitors Drug Class Antiviral agent Trials ccc. DNA Blocking Disubstituted sulphonamides Preclinical ccc. DNA Degradation RNA guided nucleases CRISPR/CAS 9) EBT 106 Anti‐HBV sg. RNA Preclinical Silencing histone acetyltransferase (HAT) inhibitors HBx targeting drugs Slide 25 of 48 Manufacturer Excision Biotherapeutics

MYR Preclin Slide 26 of 48 Peters and Locarnini Gastro and Hep 2017

Strategies to Eradicate HBV Host immune approaches Virologic approaches • • Slide 27 of 48 Entry inhibitors Block ccc. DNA Transcription inhibitors RNA interference HBV capsid inhibitor Polymerase inhibitors Secretion inhibitors • • • Interferons RIG‐I agonists TLR‐ 7 PD‐ 1/ PDL‐ 1 IL‐ 7 Therapeutic vaccines – – – Immune complex vaccines Nasal HBV (NASVAC) vaccines DNA vaccines T cell vaccines Adenovirus based vaccines (TG 1050) Yeast based vaccines
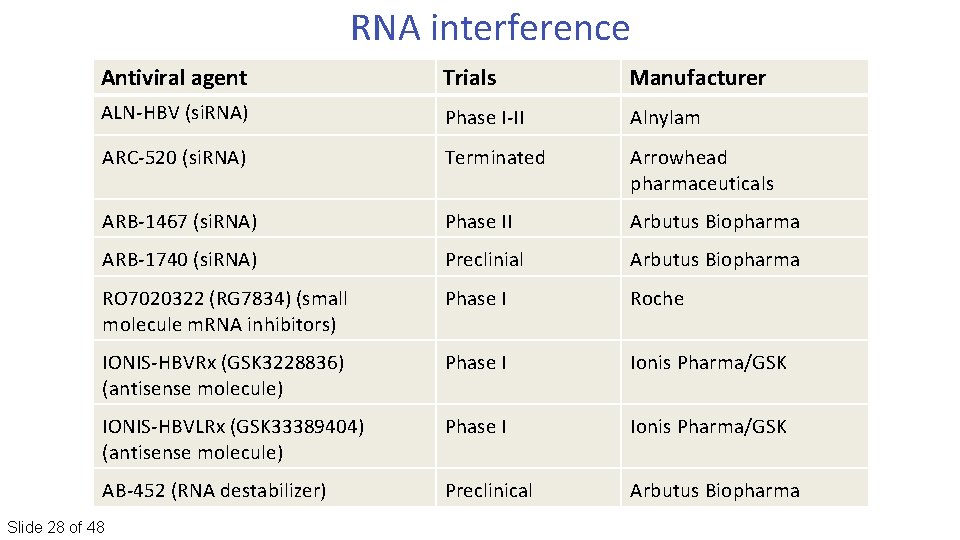
RNA interference Antiviral agent Trials Manufacturer ALN‐HBV (si. RNA) Phase I‐II Alnylam ARC‐ 520 (si. RNA) Terminated Arrowhead pharmaceuticals ARB‐ 1467 (si. RNA) Phase II Arbutus Biopharma ARB‐ 1740 (si. RNA) Preclinial Arbutus Biopharma RO 7020322 (RG 7834) (small molecule m. RNA inhibitors) Phase I Roche IONIS‐HBVRx (GSK 3228836) (antisense molecule) Phase I Ionis Pharma/GSK IONIS‐HBVLRx (GSK 33389404) (antisense molecule) Phase I Ionis Pharma/GSK AB‐ 452 (RNA destabilizer) Preclinical Arbutus Biopharma Slide 28 of 48

Silencing HBV gene expression using RNAi‐based therapy • ARC‐ 520 is a combination of si. RNAs directed against conserved HBV RNA sequences and efficiently knocks down HBV RNA, proteins and DNA levels. • 2 si. RNAs (cover 99. 6% of known HBV sequences) conjugated to cholesterol and hepatocyte‐targeted ligands • Taken up by endosomes in hepatocyte then released into cytoplasm after lysis of endosomal membrane – Given (Arrowhead Hepdart 2015) – Arbutus ARB‐ 1740 decreases HBs. Ag, HBe. Ag, HDV RNA (AASLD 2016) – ARO‐HBV EASL 2018 – 1 month human data Zeisel Gut 2015 Slide 29 of 48
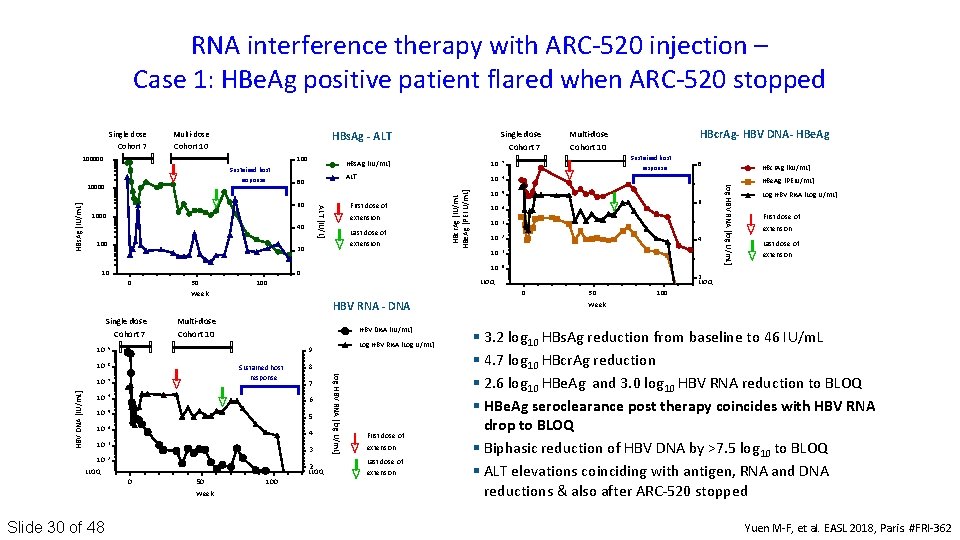
RNA interference therapy with ARC‐ 520 injection – Case 1: HBe. Ag positive patient flared when ARC‐ 520 stopped Multi‐dose Cohort 7 Cohort 10 HBs. Ag - ALT 10000 80 60 1000 40 10 7 ALT 10 6 10 5 10 4 10 3 10 2 10 1 10 0 First dose of extension Last dose of extension 20 10 HBs. Ag [IU/m. L] 0 0 50 HBV DNA [IU/m. L] H B V D N A [ I U/ m L ] 10 8 10 7 10 6 10 5 10 4 10 3 10 2 HBV DNA [IU/m. L] Sustained host response 8 7 6 5 4 3 2 LLOQ 0 50 Week Slide 30 of 48 Log HBV RNA [Log U/m. L] 9 100 Log HBV RNA [log U/m. L] V R N A [ L o g U/ m L ] H B 9 Multi‐dose Cohort 10 First dose of extension Last dose of extension 8 6 4 2 LLOQ 0 HBV RNA - DNA 10 Sustained host response LLOQ 100 Week Single dose Cohort 7 HBcr. Ag- HBV DNA- HBe. Ag Multi‐dose Cohort 10 50 H B V R N A [ L o g U/ m L ] L o g. Log HBV RNA [log U/m. L] Sustained host response A ALT [IU/L] L T [I U / L ] HBs. Ag [IU/m. L] H B s A g [I U/ m L ] 100000 Single dose Cohort 7 H B HBcr. Ag [IU/m. L c r A g [I U / m L ] H BHBe. Ag [PEI U/m. L] e A g [ P E I U/ m L ] Single dose HBcr. Ag [k. U/m. L] HBe. Ag [PEIU/m. L] Log HBV RNA [Log U/m. L] First dose of extension Last dose of extension 100 Week § 3. 2 log 10 HBs. Ag reduction from baseline to 46 IU/m. L § 4. 7 log 10 HBcr. Ag reduction § 2. 6 log 10 HBe. Ag and 3. 0 log 10 HBV RNA reduction to BLOQ § HBe. Ag seroclearance post therapy coincides with HBV RNA drop to BLOQ § Biphasic reduction of HBV DNA by >7. 5 log 10 to BLOQ § ALT elevations coinciding with antigen, RNA and DNA reductions & also after ARC‐ 520 stopped Yuen M‐F, et al. EASL 2018, Paris. #FRI‐ 362
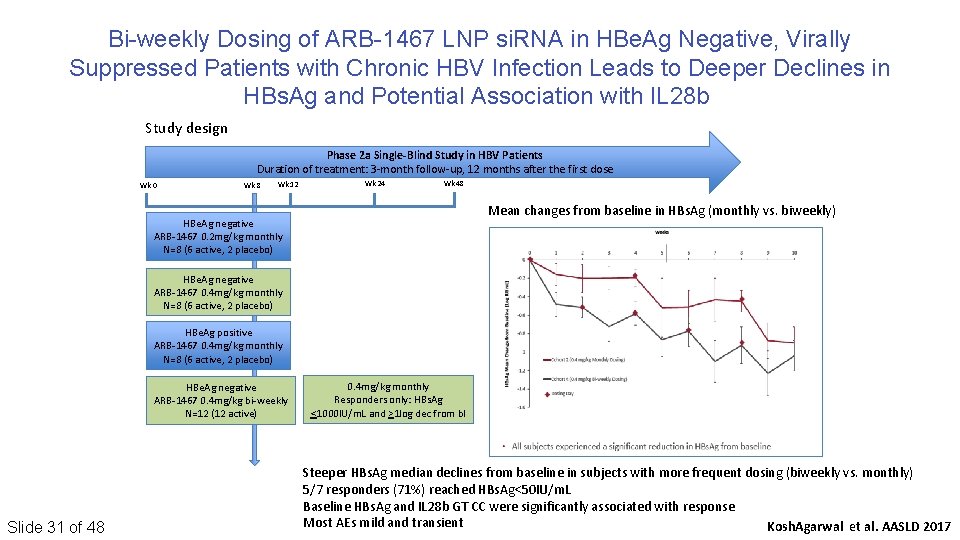
Bi-weekly Dosing of ARB-1467 LNP si. RNA in HBe. Ag Negative, Virally Suppressed Patients with Chronic HBV Infection Leads to Deeper Declines in HBs. Ag and Potential Association with IL 28 b Study design Phase 2 a Single-Blind Study in HBV Patients Duration of treatment: 3‐month follow‐up, 12 months after the first dose Wk 0 Wk 8 Wk 12 Wk 24 Wk 48 Mean changes from baseline in HBs. Ag (monthly vs. biweekly) HBe. Ag negative ARB‐ 1467 0. 2 mg/kg monthly N=8 (6 active, 2 placebo) HBe. Ag negative ARB‐ 1467 0. 4 mg/kg monthly N=8 (6 active, 2 placebo) HBe. Ag positive ARB‐ 1467 0. 4 mg/kg monthly N=8 (6 active, 2 placebo) HBe. Ag negative ARB‐ 1467 0. 4 mg/kg bi‐weekly N=12 (12 active) Slide 31 of 48 0. 4 mg/kg monthly Responders only: HBs. Ag <1000 IU/m. L and >1 log dec from bl Steeper HBs. Ag median declines from baseline in subjects with more frequent dosing (biweekly vs. monthly) 5/7 responders (71%) reached HBs. Ag<50 IU/m. L Baseline HBs. Ag and IL 28 b GT CC were significantly associated with response Most AEs mild and transient Kosh. Agarwal et al. AASLD 2017
MYR Preclin si. RNAs Ph 1 -2 Slide 32 of 48 Peters and Locarnini Gastro and Hep 2017

Strategies to Eradicate HBV Virologic approaches • • Slide 33 of 48 Entry inhibitors Block ccc. DNA Transcription inhibitors RNA interference HBV capsid inhibitor Polymerase inhibitors Secretion inhibitors Host immune approaches • • • Interferons RIG‐I agonists TLR‐ 7 PD‐ 1/ PDL‐ 1 IL‐ 7 Therapeutic vaccines – – – Immune complex vaccines Nasal HBV (NASVAC) vaccines DNA vaccines T cell vaccines Adenovirus based vaccines (TG 1050) Yeast based vaccines

Cp. AM: core protein allosteric modulator‐ Roche, ABI CAM: capsid assembly modulator ‐ J&J, Roche HBV core protein dimers Class II Cp. AM Phenylpropenamide and sulfamoylbenzamide derivatives pg. RNA-RT Class I Cp. AM Slide 34 of 48 e. g AT‐ 130, AB, JNJ, Functional nucleocapsids Heteroarylpyrimidine derivatives Core is essential for • • Empty capsids Aberrant core protein aggregates that are subsequently degraded e. g. GLS‐ 4, Bay‐ 41 HBV genome packaging Reverse transcription Intracellular trafficking Maintenance of chronic infection as encapsidated HBV genomes are imported into the nucleus.
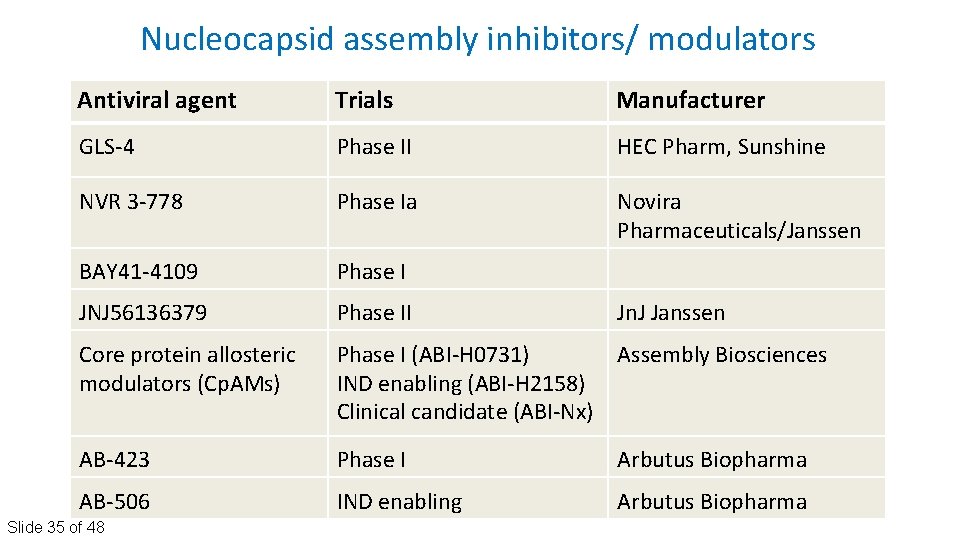
Nucleocapsid assembly inhibitors/ modulators Antiviral agent Trials Manufacturer GLS‐ 4 Phase II HEC Pharm, Sunshine NVR 3‐ 778 Phase Ia Novira Pharmaceuticals/Janssen BAY 41‐ 4109 Phase I JNJ 56136379 Phase II Core protein allosteric modulators (Cp. AMs) Phase I (ABI‐H 0731) Assembly Biosciences IND enabling (ABI‐H 2158) Clinical candidate (ABI‐Nx) AB‐ 423 Phase I Arbutus Biopharma AB‐ 506 IND enabling Arbutus Biopharma Slide 35 of 48 Jn. J Janssen

Antiviral Activity of JNJ-56136379, a novel HBV Nucleocapsid Inhibitor Session 8 JNJ‐ 379 100 mg or placebo (Day 1) 25 mg (D 2‐ 28) or placebo (D 2‐ 28), QD Sessions 8 and 9 n=12 per session (8 active/4 placebo) 28 days of treatment followed by 8 weeks of follow‐up Session 9 JNJ‐ 379 75 mg or placebo, QD Session 10 JNJ‐ 379 150 mg or placebo, QD Session 11 JNJ‐ 379 75 mg or placebo An oral dose regimen of 250 mg daily for 28 days is being evaluated Phase 2 a study is ongoing in treatment‐naïve and virologically suppressed CHB patients (NCT 03361956) Slide 36 of 48 Zoulim F et al. AASLD #LBO‐ 004, 2017 HBV DNA Baseline Day 29 Treatment Arm N Mean (SD), log 10 IU/m. L Mean (SD) change from baseline log 10 IU/m. L <LLOQ 25 mg QD 8 6. 90 (1. 91) ‐ 2. 16 (0. 49) 0 75 mg QD 8 5. 26 (1. 50) ‐ 2. 89 (0. 48) 3 Pooled placebo 8 5. 49 (1. 77) ‐ 0. 01 (0. 31) 0 56% with one AE, no SAEs, treatment d/c or deaths One patients with Grade III elevations in ALT and AST‐ TDF started
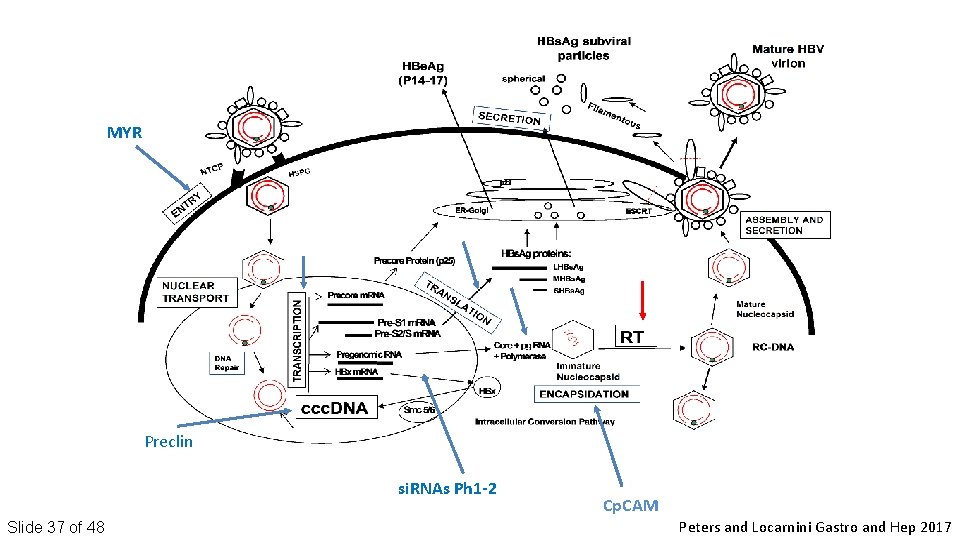
MYR Preclin si. RNAs Ph 1 -2 Slide 37 of 48 Cp. CAM Peters and Locarnini Gastro and Hep 2017

Strategies to Eradicate HBV Virologic approaches • • Slide 38 of 48 Entry inhibitors Block ccc. DNA Transcription inhibitors RNA interference HBV capsid inhibitor Polymerase inhibitors Secretion inhibitors Host immune approaches • • • Interferons RIG‐I agonists TLR‐ 7 PD‐ 1/ PDL‐ 1 IL‐ 7 Therapeutic vaccines – – – Immune complex vaccines Nasal HBV (NASVAC) vaccines DNA vaccines T cell vaccines Adenovirus based vaccines (TG 1050) Yeast based vaccines
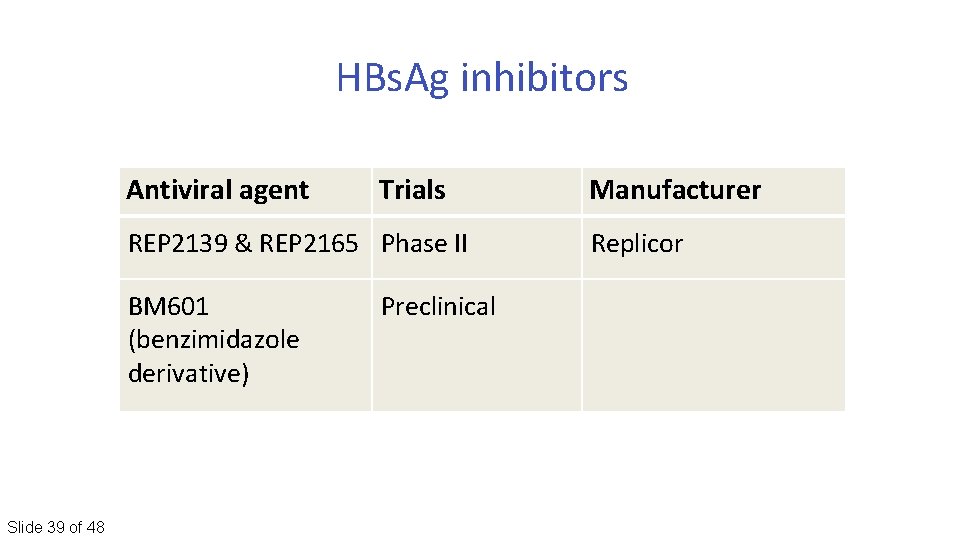
HBs. Ag inhibitors Antiviral agent Trials REP 2139 & REP 2165 Phase II BM 601 (benzimidazole derivative) Slide 39 of 48 Preclinical Manufacturer Replicor

REP 2139 -Mg and REP 2165 -Mg Combination Therapy in CHB (REP 401 - NCT 02565719) Global summary of follow‐up responses after removal of all therapy (2018) Participants entered into trial N=40 (20 with NAPs following 24 weeks of peg. IFN*) Participants currently completed treatment and ≥ 24 weeks of follow-up 34 Functional cure (HBs. Ag negative, HBV DNA TND) 14/34 (41%) Inactive chronic HBV state (HBV DNA < 2000 IU/m. L, normal ALT) 15/34 (44%) Slide 40 of 48 ACTG Personal communication, Vaillant A et al. 2018
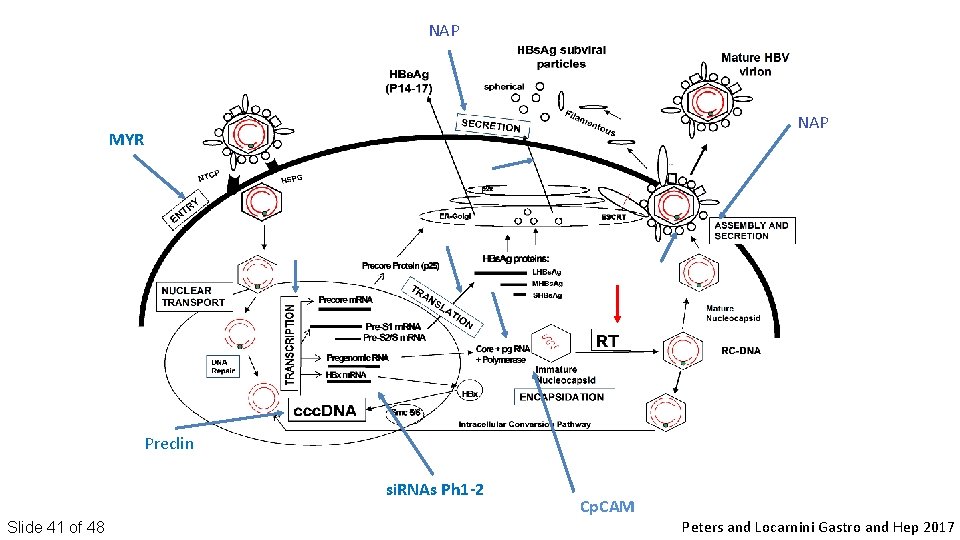
NAP MYR Preclin si. RNAs Ph 1 -2 Slide 41 of 48 Cp. CAM Peters and Locarnini Gastro and Hep 2017

Strategies to Eradicate HBV Virologic approaches • • Slide 42 of 48 Entry inhibitors Block ccc. DNA Transcription inhibitors RNA interference HBV capsid inhibitor Polymerase inhibitors Secretion inhibitors Host immune approaches • Interferons • Rig‐I agonist‐ modest • TLR‐ 7‐ no effect on HBs. Ag but increased IFN‐γ and IL‐ 2 • PD‐ 1/ PDL‐ 1 • IL‐ 7 • Birinapant selectively induces TNF mediated apoptosis (Pelligrini WEHI) • Therapeutic vaccines – – – Immune complex vaccines Nasal HBV (NASVAC) vaccines DNA vaccines T cell vaccines Adenovirus based vaccines (TG 1050) Yeast based vaccines

Strategies to Eradicate HBV Virologic approaches • • Slide 43 of 48 Entry inhibitors Block ccc. DNA Transcription inhibitors RNA interference HBV capsid inhibitor Polymerase inhibitors Secretion inhibitors Host immune approaches • Interferons • Rig‐I agonist‐ modest • TLR‐ 7‐ no effect on HBs. Ag but increased IFN‐γ and IL‐ 2 • PD‐ 1/ PDL‐ 1 • IL‐ 7 • Birinapant selectively induces TNF mediated apoptosis (Pelligrini WEHI) • Therapeutic vaccines – – – Immune complex vaccines Nasal HBV (NASVAC) vaccines DNA vaccines T cell vaccines Adenovirus based vaccines (TG 1050) Yeast based vaccines
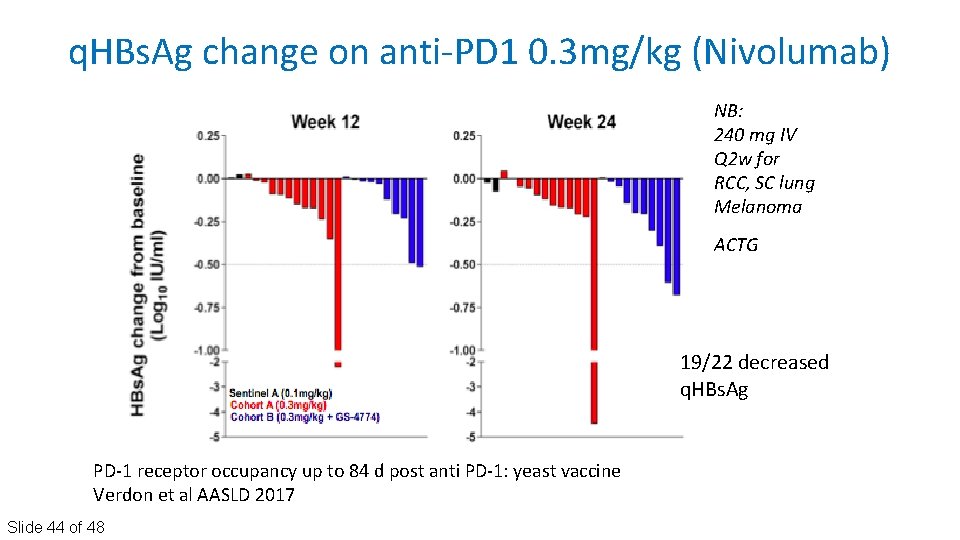
q. HBs. Ag change on anti‐PD 1 0. 3 mg/kg (Nivolumab) NB: 240 mg IV Q 2 w for RCC, SC lung Melanoma ACTG 19/22 decreased q. HBs. Ag PD‐ 1 receptor occupancy up to 84 d post anti PD‐ 1: yeast vaccine Verdon et al AASLD 2017 Slide 44 of 48

Emerging DAAs against HBV Many currently in the pipe‐line • • Novel polymerase inhibitors Capsid inhibitors ccc. DNA inhibition or eradication Core protein packaging inhibitors Small interfering RNA (si. RNA)-based strategies Secretion inhibitors Immune activators Combination therapy will likely be required for cure • Inhibitors of polymerase, entry, core, ccc. DNA etc • IFN, immune stimulant, TLR 7 • Checkpoint inhibitors PD-1/L 1 Slide 45 of 48 BUT Zeizel Gut 2015

Emerging DAAs against HBV Many currently in the pipe‐line • • • Novel polymerase inhibitors Capsid inhibitors ccc. DNA inhibition or eradication Core protein packaging inhibitors Small interfering RNA (si. RNA)-based strategies Immune activators Combination therapy will likely be required for cure • Inhibitors of polymerase, entry, core, ccc. DNA etc • IFN, immune stimulant, TLR 7 • Checkpoint inhibitors PD-1/L 1 Slide 46 of 48 BUT Selection of HBV patient will be critical Zeizel Gut 2015 Optimization of HBV endpoints needed

ARS 2: HBV status in 2019: which is true? 1. Acute HBV in adults leads to loss of HBs. Ag usually 2. HBV infection can be cured 3. Currently available drugs lead to loss of HBs. Ag usually 4. HBV is not a latent virus Slide 47 of 48

ARS 2: New drugs in Phase I/II: which is false? 1. Target Hepatitis B core protein 2. Target HBV entry into hepatocyte 3. Target ccc. DNA in nucleus 4. Target HBV m. RNA 5. Target HBV secretion Slide 48 of 48
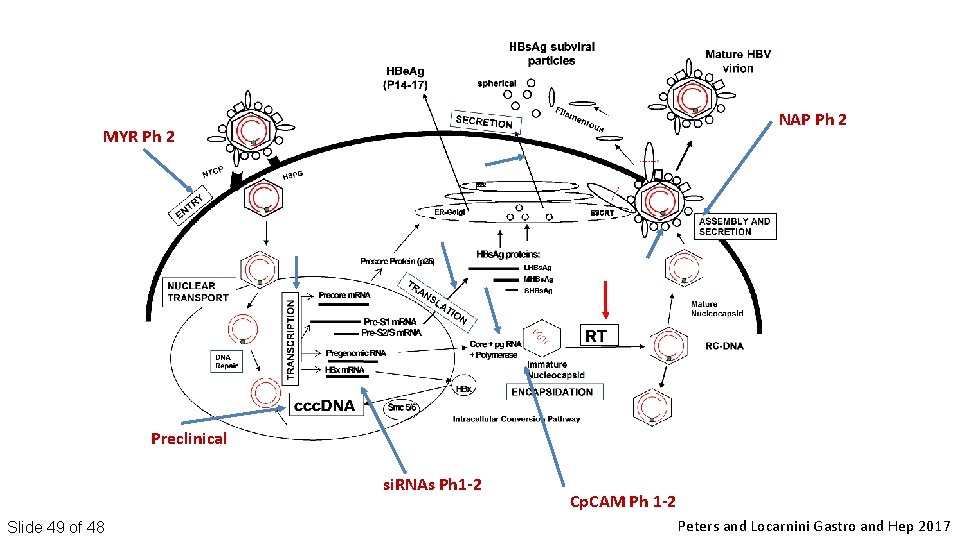
NAP Ph 2 MYR Ph 2 Preclinical si. RNAs Ph 1 -2 Slide 49 of 48 Cp. CAM Ph 1 -2 Peters and Locarnini Gastro and Hep 2017

Question-and-Answer Slide 50 of 48
- Slides: 50