Irreversible Reactions Reactions completely change reactants into products


§ Irreversible Reactions § Reactions completely change reactants into products § Example: Once it is chocolate, it will always only be chocolate a. A + b. B -> c. C + d. D

§ Most reactions are reversible reactions § Both forward and reverse directions § Example: Changes into chocolate, but also changes back into cacao and sugar at the same time § Product is made, but some of the product is also changing back to reactants a. A + b. B -> c. C + d. D
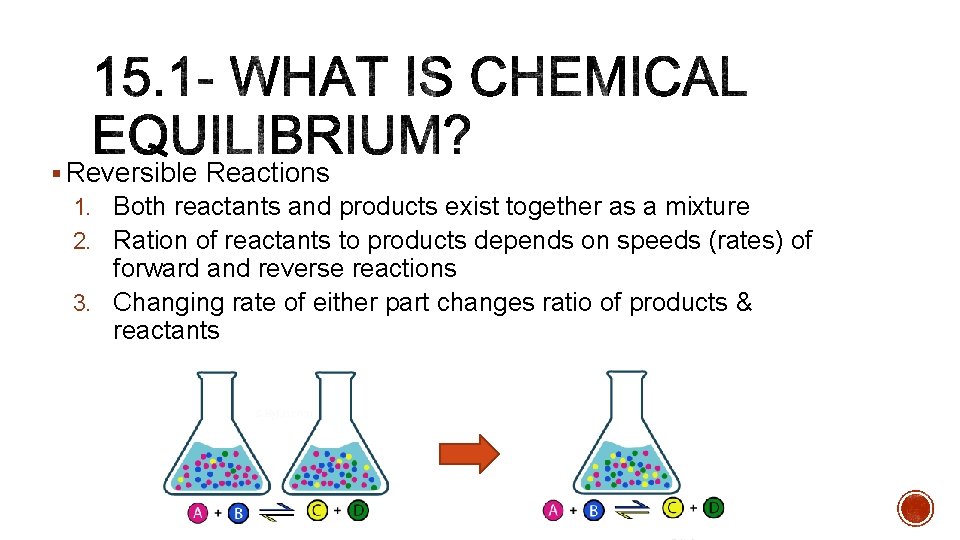
§ Reversible Reactions 1. Both reactants and products exist together as a mixture 2. Ration of reactants to products depends on speeds (rates) of forward and reverse reactions 3. Changing rate of either part changes ratio of products & reactants

§ Chemical equilibrium§ forward & reverse occur at same rate, balance each other

§ Chemical equilibrium is dynamic. Particles involved constantly move and react. § Ethane (C 2 H 6) reacts with Oxygen to produce water and carbon dioxide 2 C 2 H 6 + 7 O 2 6 H 2 O + 4 CO 2 § But some water also collides with carbon dioxide to form ethane and oxygen 6 H 2 O + 4 CO 2 2 C 2 H 6 + 7 O 2


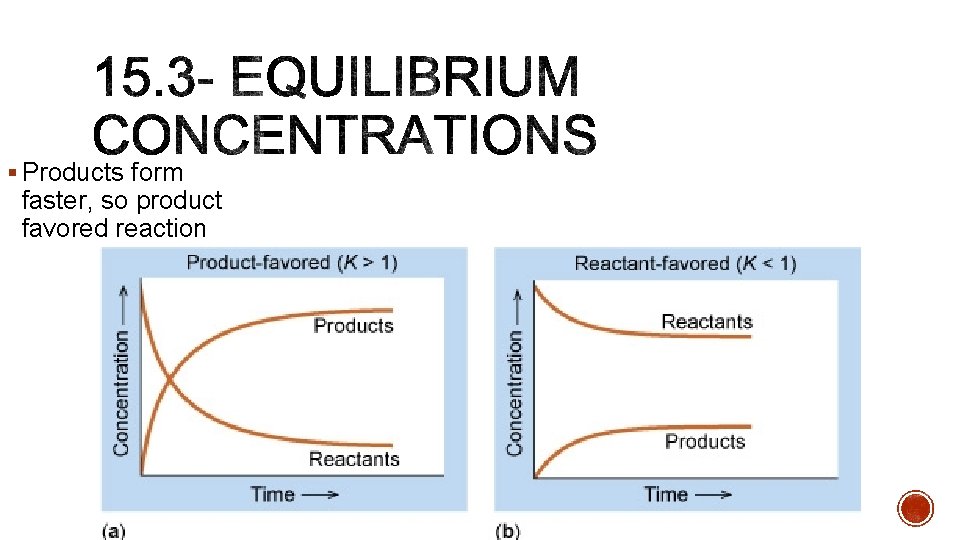
§ Products form faster, so product favored reaction
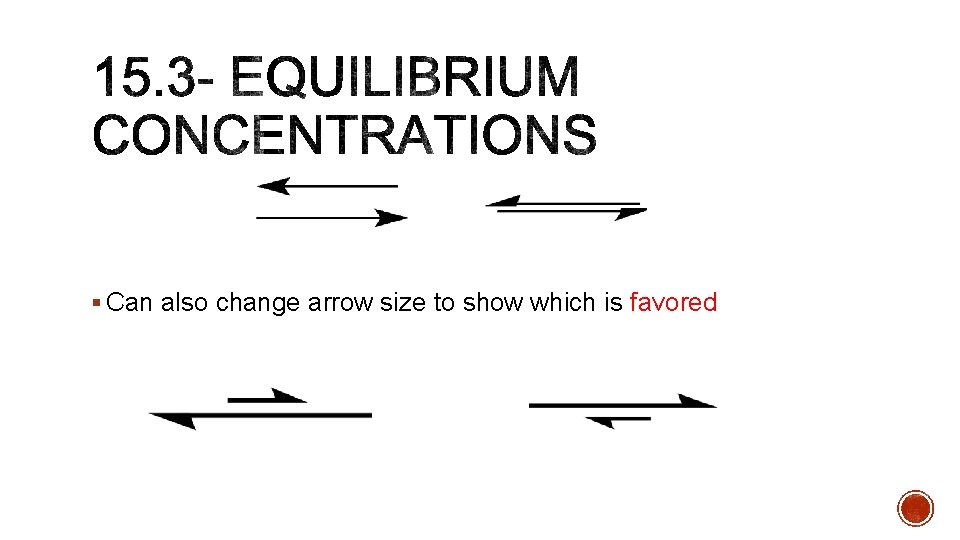
§ Can also change arrow size to show which is favored





![§ Good to use something called an ICEbox [A] Initial Change Equilibrium [B] [C] § Good to use something called an ICEbox [A] Initial Change Equilibrium [B] [C]](http://slidetodoc.com/presentation_image_h2/83261e8dd89eb3d03f4c3a5184020a74/image-16.jpg)
§ Good to use something called an ICEbox [A] Initial Change Equilibrium [B] [C]


![§ [H 2] Initial Change Equilibrium [I 2] [HI] § [H 2] Initial Change Equilibrium [I 2] [HI]](http://slidetodoc.com/presentation_image_h2/83261e8dd89eb3d03f4c3a5184020a74/image-19.jpg)
§ [H 2] Initial Change Equilibrium [I 2] [HI]
![§ Initial Change Equilibrium [H 2] [I 2] [HI] 0. 200 0 § Initial Change Equilibrium [H 2] [I 2] [HI] 0. 200 0](http://slidetodoc.com/presentation_image_h2/83261e8dd89eb3d03f4c3a5184020a74/image-20.jpg)
§ Initial Change Equilibrium [H 2] [I 2] [HI] 0. 200 0
![§ [H 2] [I 2] [HI] Initial 0. 200 0 Change -x -x + § [H 2] [I 2] [HI] Initial 0. 200 0 Change -x -x +](http://slidetodoc.com/presentation_image_h2/83261e8dd89eb3d03f4c3a5184020a74/image-21.jpg)
§ [H 2] [I 2] [HI] Initial 0. 200 0 Change -x -x + 2 x Equilibrium
![§ [H 2] [I 2] [HI] Initial 0. 200 0 Change -x -x + § [H 2] [I 2] [HI] Initial 0. 200 0 Change -x -x +](http://slidetodoc.com/presentation_image_h2/83261e8dd89eb3d03f4c3a5184020a74/image-22.jpg)
§ [H 2] [I 2] [HI] Initial 0. 200 0 Change -x -x + 2 x Equilibrium 0. 200 – x 2 x





- Slides: 27