Iron Making Blast Furnace Reactions BASICS OF IRON

Iron Making – Blast Furnace Reactions BASICS OF IRON MAKING

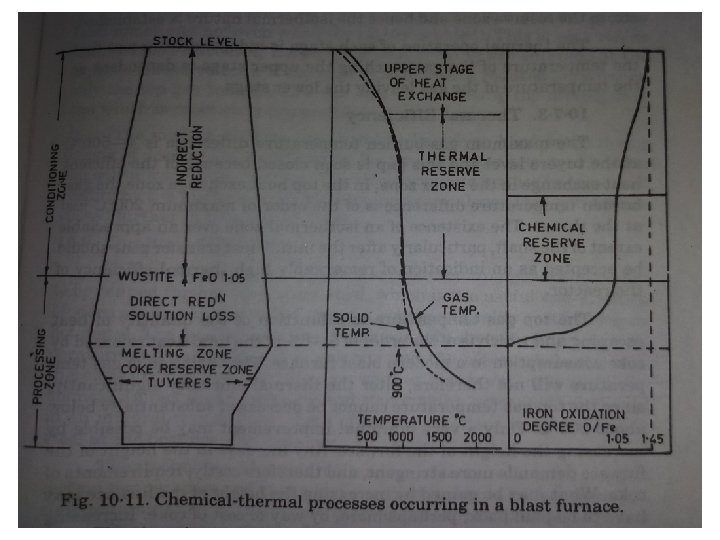


Reactions in Stack 1. Progressively reduce the iron oxide to iron. 2. Extract sensible heat from ascending gases. Gases at stock line must be at atmospheric temperature & CO/CO 2 ratio should be in equilibrium with iron oxide as 3 Fe 2 O 3 + CO = 2 Fe 3 O 4 + CO 2 Effluent gases temp – 200 o. C CO/CO 2 = 1. 5 – 3. 0

CO/CO 2 ratio in contact with Fe. O & Carbon at 950 o. C. 1. CO 2 + C = 2 CO Equilibrium is in proportion to 100% CO 2. Fe + O 2 = 2 Fe. O Pco/pco 2= (-1018/T) + 1. 181 Equilibrium is in proportion to 70%/30% - CO/CO 2


Reduction & Regeneration : Air enters BF at 1000 o. C & O 2 potential is normal atmosphere i. e, p. O 2 = 0. 21 atmos. - Oxygen combine with Coke to form CO and develops temperature 2000 o. C (Sharp falling Curve). - Later before the formation of CO , O 2 comes in contact with iron oxide, there may be Fe. O to Fe 3 O 4 there by raising the oxygen potential.

Reaction in Bosh Reduction of iron oxide by CO contained in the ascending gases in stack and directly by solid carbon or by Naumann reversion reaction continues as direct reduction. Out of which the gas-solid reaction is faster and dominates in stack region(Indirect reduction). - Gangue mixed Fe. O is formed. - Highly oxidising and low melting slag (Fe. O) + <C> = <Fe> + {CO}

Initial slag will have high Fe. O content later on reacting with Ca. O will reduce its acidic nature. Reduction of Si. O 2, P 2 O 5, Cr 2 O 3 are reduced.
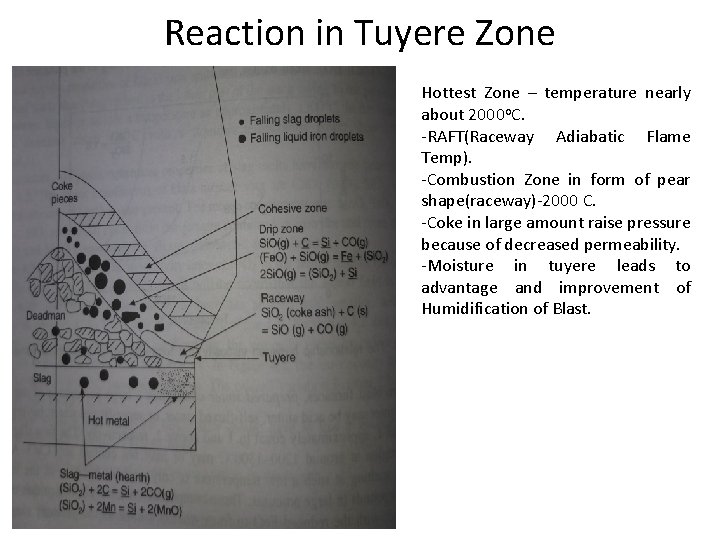
Reaction in Tuyere Zone Hottest Zone – temperature nearly about 2000 o. C. -RAFT(Raceway Adiabatic Flame Temp). -Combustion Zone in form of pear shape(raceway)-2000 C. -Coke in large amount raise pressure because of decreased permeability. -Moisture in tuyere leads to advantage and improvement of Humidification of Blast.
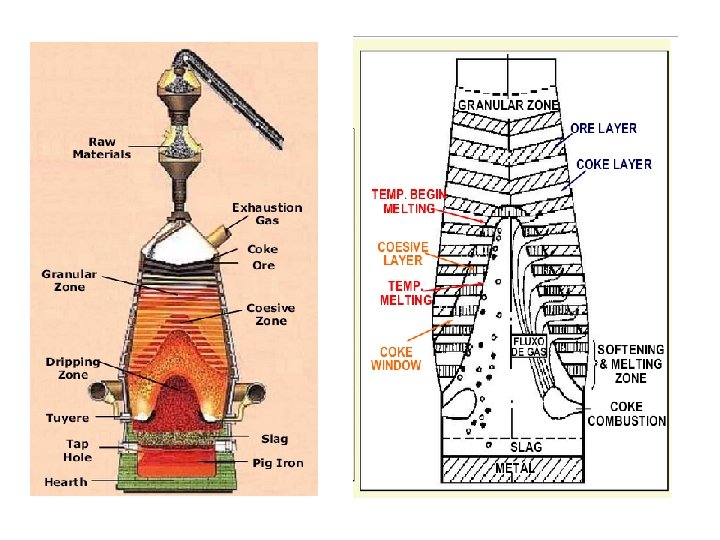
- Slides: 12