IRON DEFICIENCY ANAEMIA Dr Vivek Veeram Reddy Background

IRON DEFICIENCY ANAEMIA Dr. Vivek Veeram Reddy

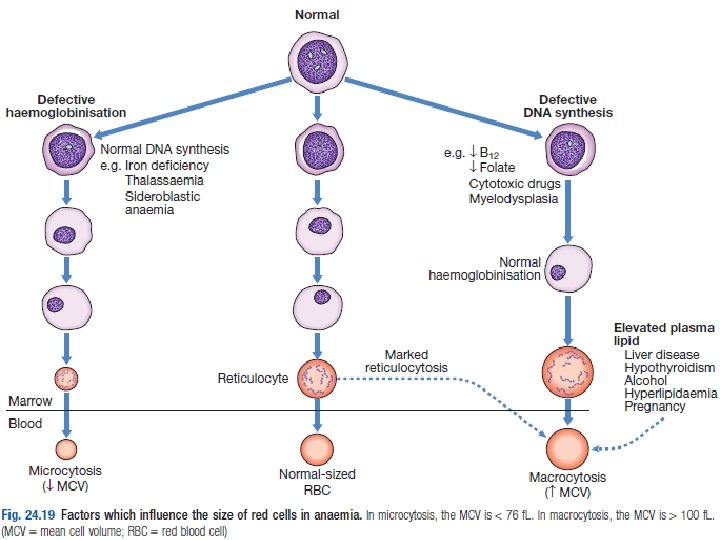
Background • Commonest cause of anaemia worldwide • Caused by defective synthesis of haemoglobin, resulting in red cells that are smaller than normal (microcytic) and contain reduced amounts of haemoglobin (hypochromic).

IRON CYCLE & METABOLISM

The Iron Cycle in Humans 1. 2. 3. 4. 5. 6. 7. 8. Nutritional Iron Balance and Absorption Iron Absorption at Molecular level Regulation of Iron Absorption by Hepcidin Iron Transport Cellular Iron uptake and release Iron Utilization Iron Storage Iron re-cycling (Reticulo-endothelial system)

1. Nutritional Iron Balance • No regulated excretory pathway for iron. • The margin between the amount of iron available for absorption and the requirement for iron in growing infants and the adult female is narrow. • Iron bioavailability is affected by the nature of the foodstuff. • An individual with iron deficiency can increase iron absorption to approximately 20% of the iron present in a meat-containing diet but only 5– 10% of the iron in a vegetarian diet.

1. Iron Absorption • Site : Proximal small intestine (Duodenum & Jejunum) • Facilitated by intraluminal acidic p. H of the stomach • Phytates and Phosphates reduce iron absorption by approximately 50%. • Foods containing Ascorbic acid increase non-heme iron absorption. • Therapeutic ferrous iron is well absorbed on empty stomach. • Heme iron is not affected by ingestion of other food items. • For an equivalent amount of ferrous salt, iron in vegetables is only about onetwentieth as available, egg iron one-eighth, liver iron one-half, and heme iron one-half to two-thirds.
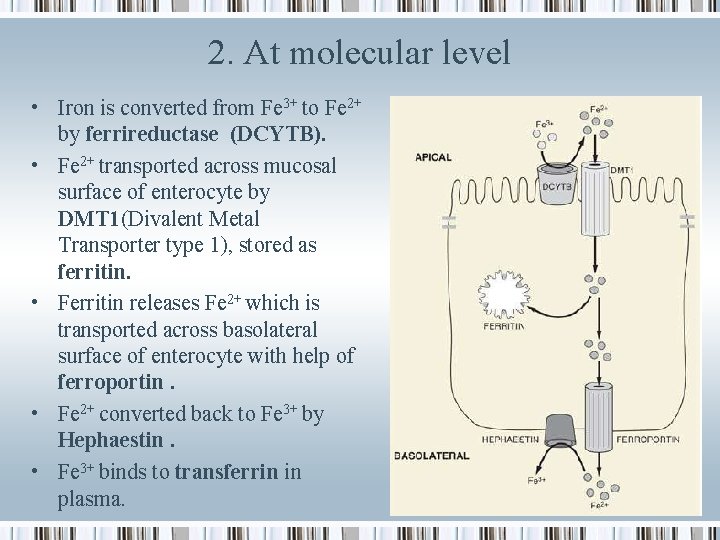
2. At molecular level • Iron is converted from Fe 3+ to Fe 2+ by ferrireductase (DCYTB). • Fe 2+ transported across mucosal surface of enterocyte by DMT 1(Divalent Metal Transporter type 1), stored as ferritin. • Ferritin releases Fe 2+ which is transported across basolateral surface of enterocyte with help of ferroportin. • Fe 2+ converted back to Fe 3+ by Hephaestin. • Fe 3+ binds to transferrin in plasma.

3. Regulation of Iron Absorption • Regulated at two stages – Mucosal uptake – At stage of transfer to blood 1. HIF-2 ά- a mediator of cellular adaptation to hypoxia, regulates DMT 1 transcription and thus regulates mucosal uptake of iron. 2. Iron transfer to the plasma depends on the requirements of the erythron for iron and the level of iron stores. • This regulation is mediated directly by hepcidin.
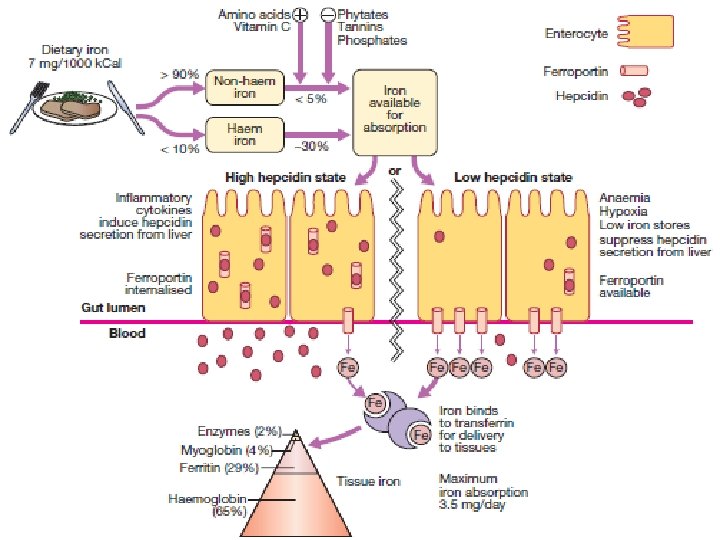

Hepcidin • Increased erythropoiesis causes decreased hepcidin. – Blocks ferroportin – Prevents absorption of iron from enterocytes. – Prevents iron exportation from macrophages. – Increased in inflammation. – Leads to reduced serum iron, microcytic anemia, and incomplete response to iron therapy

Ferroportin • Transporter protein of iron in enterocytes and macrophages. • Blocked by hepcidin.

4. Iron Transport • Transferrin is the major protein responsible for transporting iron in the body • Transferrin receptors, located in almost all cells of the body, can bind two molecules of transferrin. • One molecule of transferrin binds two molecules of iron. • Transferrin, when incompletely saturated with iron, exists in four forms: 1. Apotranferrin 2. Monoferrric transferrin A 3. Monoferrric transferrin B 4. Diferric Transferrin
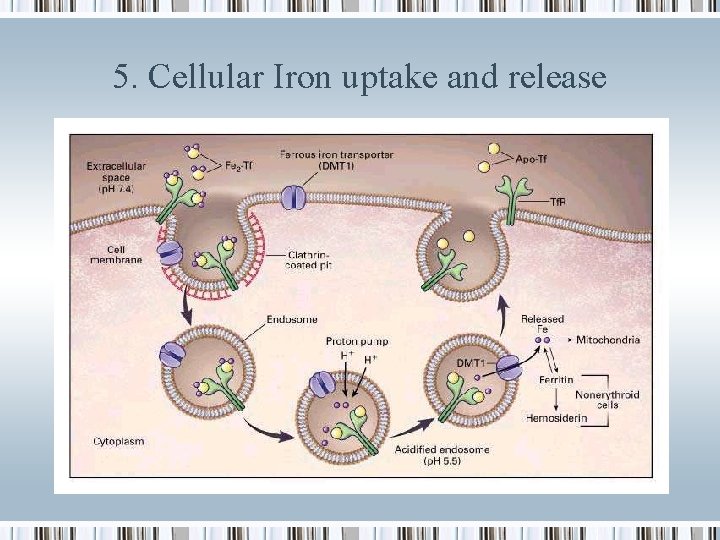
5. Cellular Iron uptake and release

6. Iron Utilization • Within the erythroid cell, iron in excess of the amount needed for hemoglobin synthesis binds to a storage protein, apoferritin, forming ferritin. • This mechanism of iron exchange also takes place in other cells of the body expressing transferrin receptors, especially liver parenchymal cells where the iron can be incorporated into heme-containing enzymes or stored. • The iron incorporated into hemoglobin subsequently enters the circulation as new red cells are released from the bone marrow. • The iron is then part of the red cell mass and will not become available for reutilization until the red cell dies.

7. Iron Storage • Iron stored in two forms – Soluble ferritin – Insoluble hemosiderin- denatured form of ferritin in which the protein shells have partly degraded, allowing the iron cores to aggregate. Hemosiderin deposits are seen on Prussian-blue positivity after staining of tissue sections with potassium ferrocyanide in acid.

8. Reticulo-Endothelial System • The reticuloendothelial macrophages play a major role in recycling iron resulting from the degradation of haemoglobin from senescent erythrocytes. • They engulf red blood cells and release the iron within using haem oxygenase. • The globin and other proteins are returned to the amino acid pool, and the iron is shuttled back to the surface of the RE cell, where it is presented to circulating transferrin. • It is the efficient and highly conserved recycling of iron from senescent red cells that supports steady-state erythropoiesis.

RISK FACTORS & CAUSES

Risk factors in the development of IDA
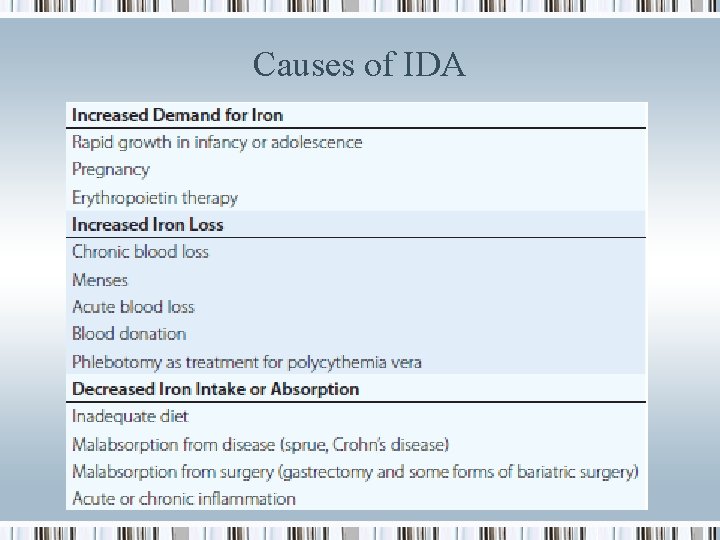
Causes of IDA

CLINICAL PRESENATION
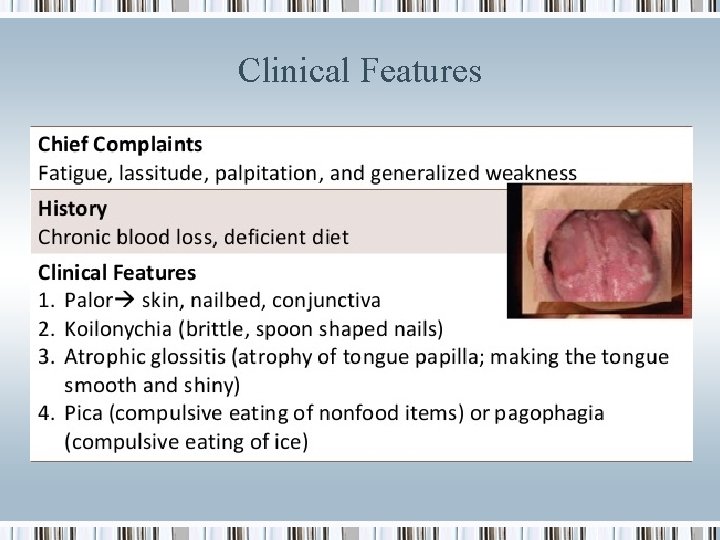
Clinical Features

Symptoms and Signs

Rare Manifestations • • Decreased Immunity Menstrual Irregularities Restless Leg Syndrome Plummer-Vinson Syndrome

Stages of IDA • Negative Iron Balance • Iron-Deficient Erythropoiesis • Iron Deficiency Anaemia


INVESTIGATIONS
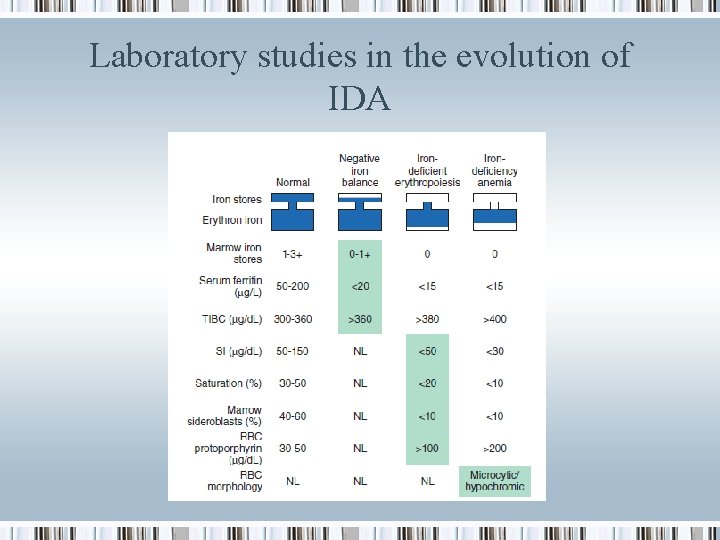
Laboratory studies in the evolution of IDA

Investigations in IDA

Diagnosis of IDA

Differential Diagnoses 1. Anaemias of Chronic Disease 2. Thalassemias 3. Sideroblastic Anaemia (Myelodysplastic syndromes)
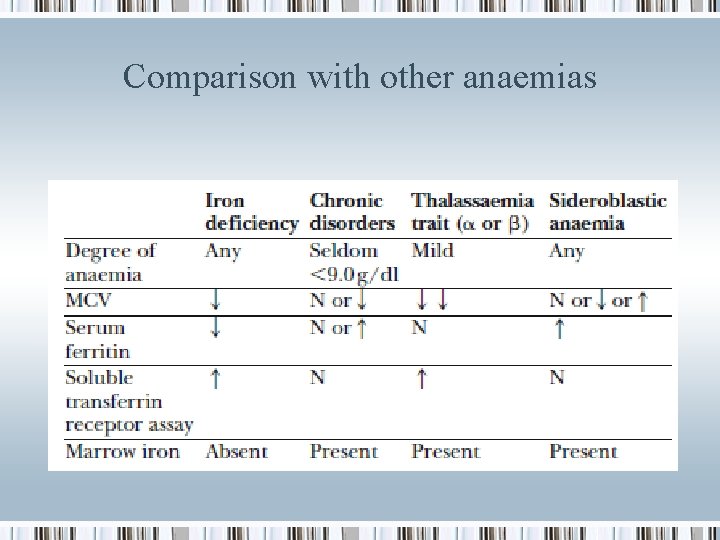
Comparison with other anaemias

EVALUATION OF IDA
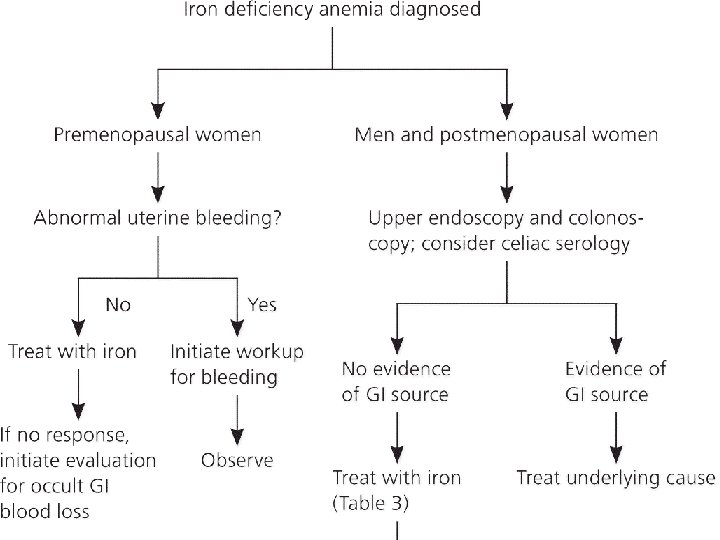
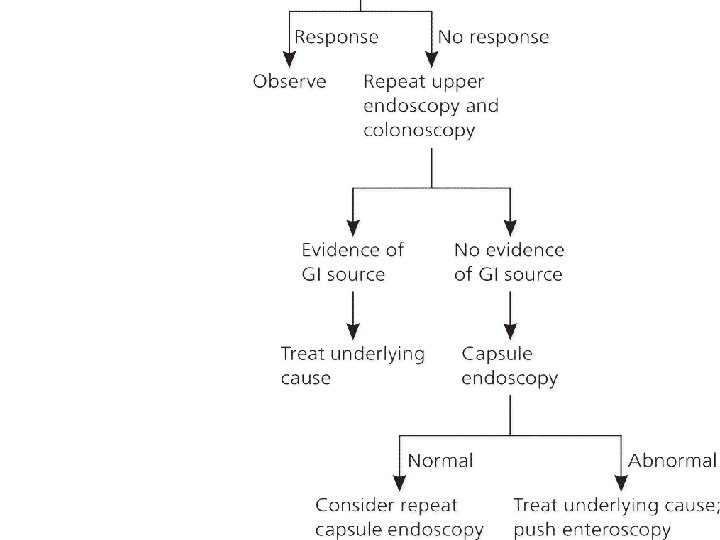

TREATMENT

• Once the diagnosis of iron-deficiency anemia and its cause is made, there are three major therapeutic approaches. 1. Red Cell Transfusion 2. Oral Iron Therapy 3. Parenteral Iron Therapy
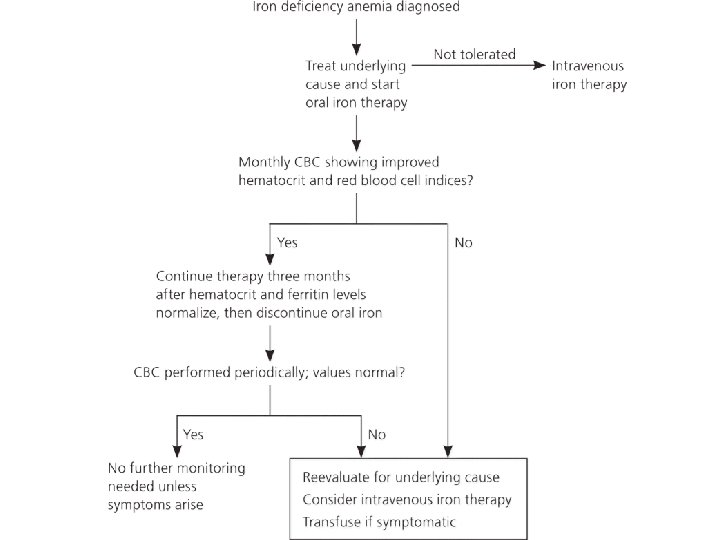

1. Red Cell Transfusion • Reserved for individuals who have symptoms of anemia, cardiovascular instability, and continued and excessive blood loss from whatever source and who require immediate intervention. • Correct the anemia acutely • The transfused red cells provide a source of iron for reutilization
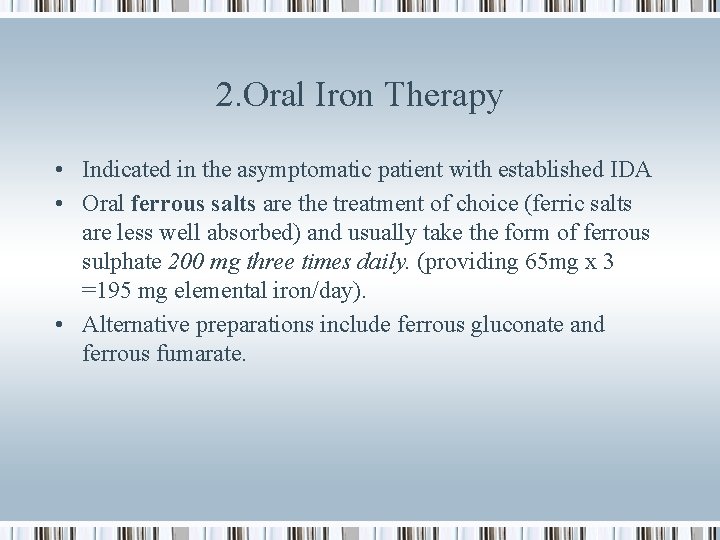
2. Oral Iron Therapy • Indicated in the asymptomatic patient with established IDA • Oral ferrous salts are the treatment of choice (ferric salts are less well absorbed) and usually take the form of ferrous sulphate 200 mg three times daily. (providing 65 mg x 3 =195 mg elemental iron/day). • Alternative preparations include ferrous gluconate and ferrous fumarate.

• A dose of 200 mg of elemental iron per day should result in the absorption of iron up to 50 mg/d. • This supports a red cell production level of two to three times normal in an individual with a normally functioning marrow and appropriate erythropoietin stimulus. • The goal of therapy in individuals with iron-deficiency anemia is not only to repair the anemia, but also to provide stores of at least 0. 5– 1 g of iron. • Sustained treatment for a period of 6– 12 months after correction of the anemia will be necessary to achieve this

• Ideally, oral iron preparations should be taken on an empty stomach, since food may inhibit iron absorption. • Some patients with gastric disease or prior gastric surgery require special treatment with iron solutions, because the retention capacity of the stomach may be reduced. The retention capacity is necessary for dissolving the shell of the iron tablet before the release of iron. • Abdominal pain, nausea, vomiting, or constipation may lead to noncompliance.
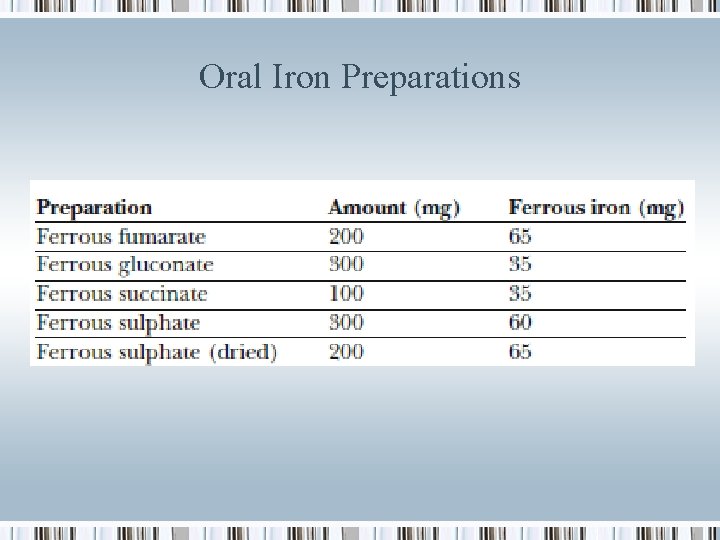
Oral Iron Preparations

Monitoring response to therapy • Typically, the reticulocyte count should begin to increase within 4– 7 days after initiation of therapy and peak at 1– 1. 5 weeks. • The absence of a response may be due to poor absorption, noncompliance (which is common), or a confounding diagnosis. Iron tolerance test. • Two iron tablets are given to the patient on an empty stomach, and the serum iron is measured serially over the subsequent 2 h. • Normal absorption will result in an increase in the serum iron of at least 100 μg/ d. L. • If iron deficiency persists despite adequate treatment, it may be necessary to switch to parenteral iron therapy.

3. Parenteral Iron Therapy • • Indicated in patients who are unable to tolerate oral iron whose needs are relatively acute who need iron on an ongoing basis (due to persistent GI loss) Recombinant EPO therapy produces a large demand for iron • The amount of iron needed by an individual patient is calculated by the following formula: Body weight (kg) × 2. 3 × (15 – patient’s hemoglobin, g/d. L) + 500 or 1000 mg (for stores).

• Parenteral iron is used in two ways: 1. one is to administer the total dose of iron required to correct the hemoglobin deficit and provide the patient with at least 500 mg of iron stores; 2. the second is to give repeated small doses of parenteral iron over a protracted period.

Iron Preparations • Iron dextran (not used anymore) • Safer preparations like Ferumoxytol (Feraheme), sodium ferric gluconate (Ferrlecit), iron sucrose (Venofer), and ferric carboxymaltose (Injectafer) • Ferumoxytol delivers 510 mg of iron per injection; ferric gluconate 125 mg per injection, ferric carboxymaltose 750 mg per injection, and iron sucrose 200 mg per injection.

Adverse effects • In administering intravenous iron dextran, anaphylaxis is a concern. • Anaphylaxis is much rarer with the newer preparations. • Generalized symptoms appearing several days after the infusion of a large dose of iron can include arthralgias, skin rash, and low-grade fever. • The iron preparation should be diluted in 5% dextrose in water or 0. 9% Na. Cl solution and can then be infused over a 60 - to 90 -min at a rate convenient for monitoring. • If chest pain, wheezing, a fall in blood pressure, or other systemic symptoms occur, the infusion of iron should be stopped immediately.

Dietary Advice • Foods rich in Iron must be taken in patients who have no symptoms of malabsorption • Heme iron rich foods have higher bioavailability than nonheme iron rich foods! • For an equivalent amount of ferrous salt, iron in vegetables is only about one-twentieth as available, egg iron oneeighth, liver iron one-half, and heme iron one-half to twothirds.

THANK YOU!
- Slides: 50