Iron Chemistry sources and sinks Iron hypothesis Martin



- Slides: 9

Iron : Chemistry, sources and sinks.

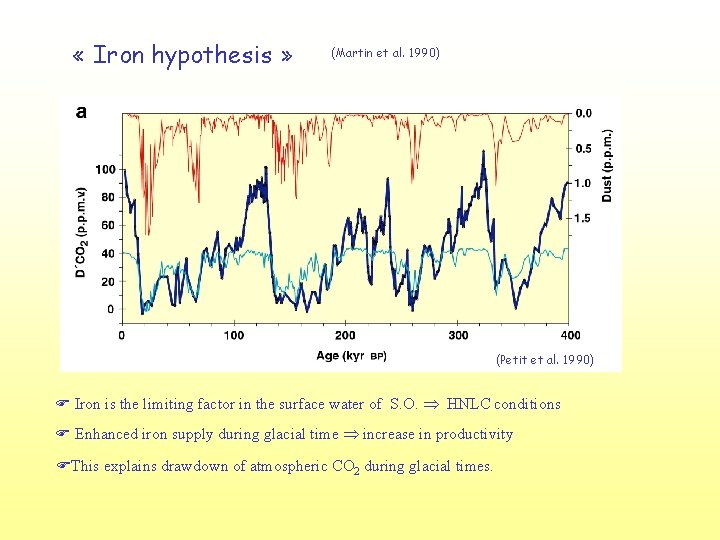
« Iron hypothesis » (Martin et al. 1990) (Petit et al. 1990) F Iron is the limiting factor in the surface water of S. O. HNLC conditions F Enhanced iron supply during glacial time increase in productivity FThis explains drawdown of atmospheric CO 2 during glacial times.

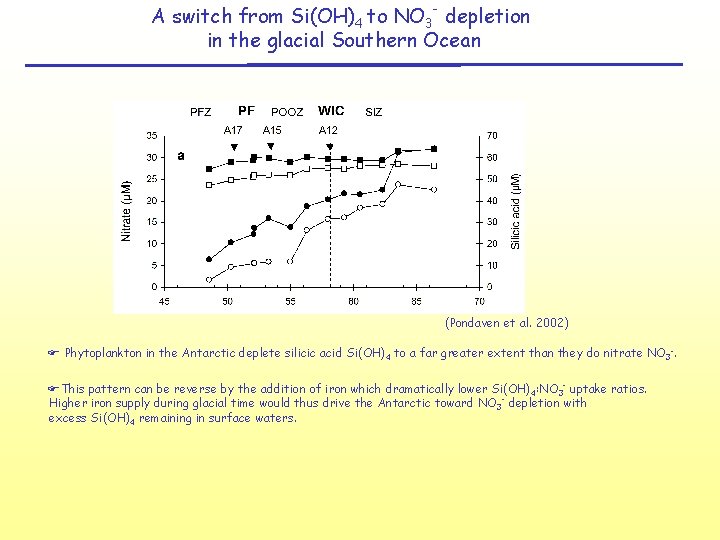
A switch from Si(OH)4 to NO 3 - depletion in the glacial Southern Ocean (Pondaven et al. 2002) F Phytoplankton in the Antarctic deplete silicic acid Si(OH)4 to a far greater extent than they do nitrate NO 3 -. FThis pattern can be reverse by the addition of iron which dramatically lower Si(OH)4: NO 3 - uptake ratios. Higher iron supply during glacial time would thus drive the Antarctic toward NO 3 - depletion with excess Si(OH)4 remaining in surface waters.

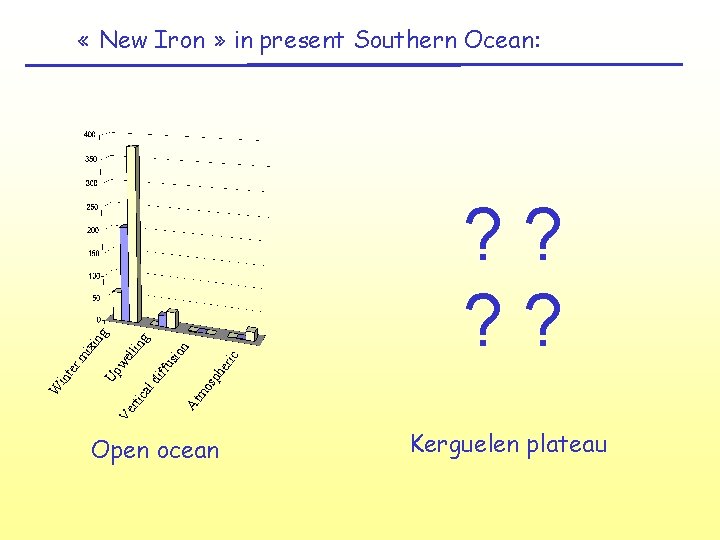
mo sp he ? ? At rti ca ld iff us ric ion ng lli we Up Ve W int er mi xin g « New Iron » in present Southern Ocean: Open ocean Kerguelen plateau

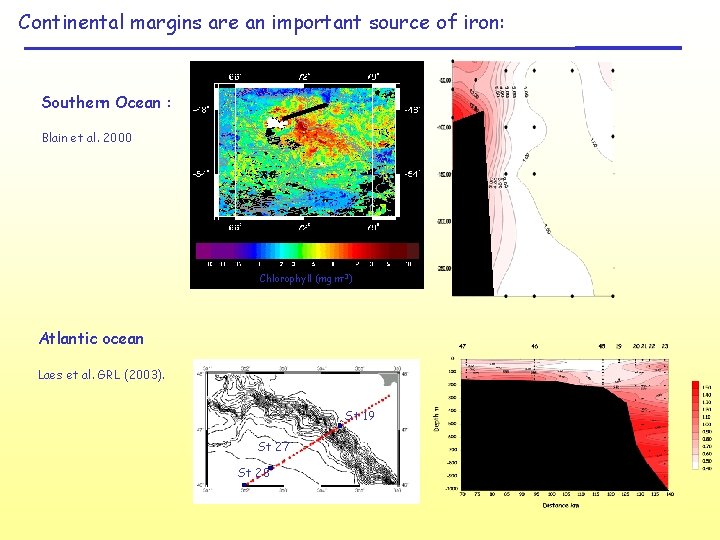
Continental margins are an important source of iron: Southern Ocean : Blain et al. 2000 Chlorophyll (mg m-3) Atlantic ocean Laes et al. GRL (2003). St 19 St 27 St 28

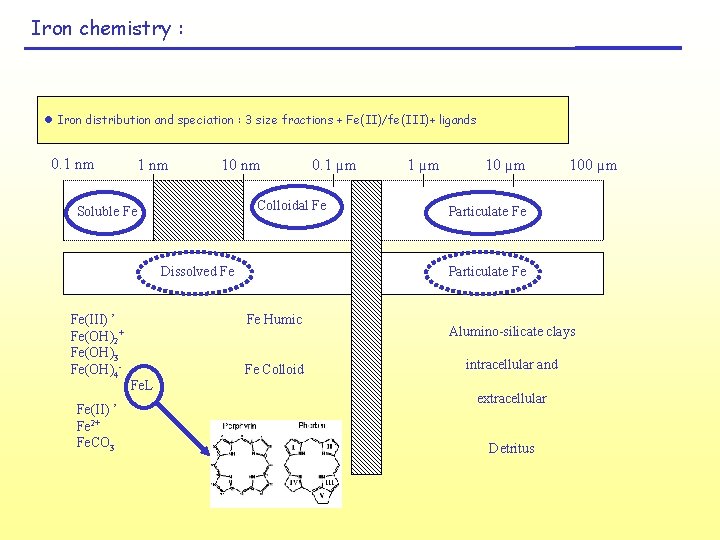
Iron chemistry : l Iron distribution and speciation : 3 size fractions + Fe(II)/fe(III)+ ligands 0. 1 nm 10 nm Colloidal Fe Soluble Fe Dissolved Fe Fe(III) ’ Fe(OH)2+ Fe(OH)3 Fe(OH)4 Fe(II) ’ Fe 2+ Fe. CO 3 1 µm 100 µm Particulate Fe Fe Humic Fe Colloid Fe. L 0. 1 µm Alumino-silicate clays intracellular and extracellular Detritus

IRON TEAM: • Water Column : international team. LEMAR, LOB, NIOZ and ARC • long experience in iron work • have already work together at sea • SAFE cruise in October 2004. • On the Marion Dufresne: • sampling and onboard analysis. • To collect samples for other trace metal determination • To provide clean seawater for OBEX • Sediment : LGE and LSCE

IRON TEAM: • Atmosphere : LOV and LISA ØTo determine the particles abundance of the area ØTo determine the size distribution in order to attempt a IRON FLUX calculation. ØTo determine the mineralogical composition. ØTo determine some geochemical tracers (Al, Pb, …) and elements of biogeochemical interest such as Fe and P. ØTo quantify dissolved iron originating from these particles in the surface seawater

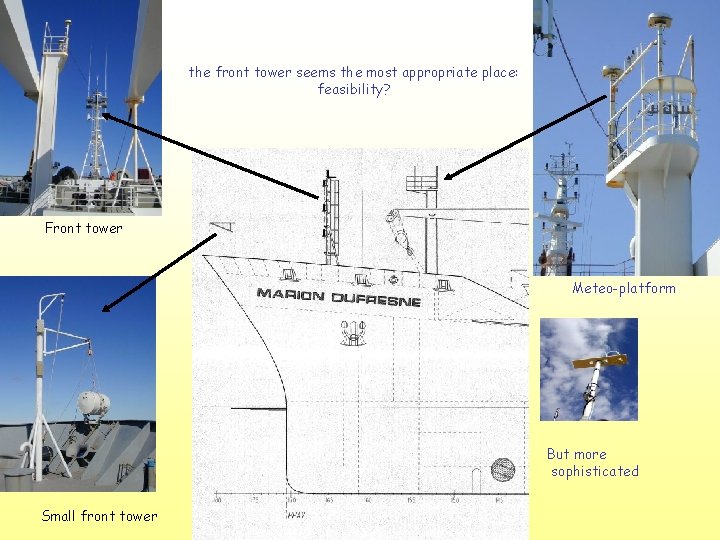
the front tower seems the most appropriate place: feasibility? Front tower Meteo-platform But more sophisticated Small front tower