Irbesartan Diabetic Nephropathy Trial IDNT Collaborative Study Group

Irbesartan Diabetic Nephropathy Trial (IDNT) Collaborative Study Group N Eng J Med 345: 851 -860, 2001 Edmund J. Lewis, M. D. Muehrcke Family Professor of Nephrology Section of Nephrology Rush University Medical Center Chicago, IL

RC Atkins et al: Proteinuria reduction and progression to renal failure in patients with type 2 diabetes mellitus and overt nephropathy. American Journal of Kidney Diseases 45: 281 -287, 2005

Definitions (IDNT) measure of proteinuria: baseline and quarterly 24 hr urine protein outcomes: primary: composite time to doubling serum creatinine plus end stage renal failure (Cr>6 mgdl) plus all cause mortality

IDNT – Secondary Outcome Renal outcome: doubling of serum creatinine or end stage renal disease (creatinine ≥ 6 mg/dl, dialysis or transplantation).

Analyses IDNT baseline proteinuria vs CKD progression X early change in proteinuria vs CKD progression X effect of treatment On CKD progression X

Irbesartan Diabetic Nephropathy Trial (IDNT) DOUBLE MASKED RANDOMIZED PLACEBO CONTROLLED IRBESARTAN (300 MG) AMLODIPINE (10 MG) CONTROL TOTAL N 579 567 569 1715 TARGET BP 135/85 MMHG MEDIAN FOLLOW-UP 30 MONTHS PRIMARY COMPOSITE END POINT: DOUBLING SERUM CREATININE END STAGE RENAL FAILURE (CR>6) MORTALITY
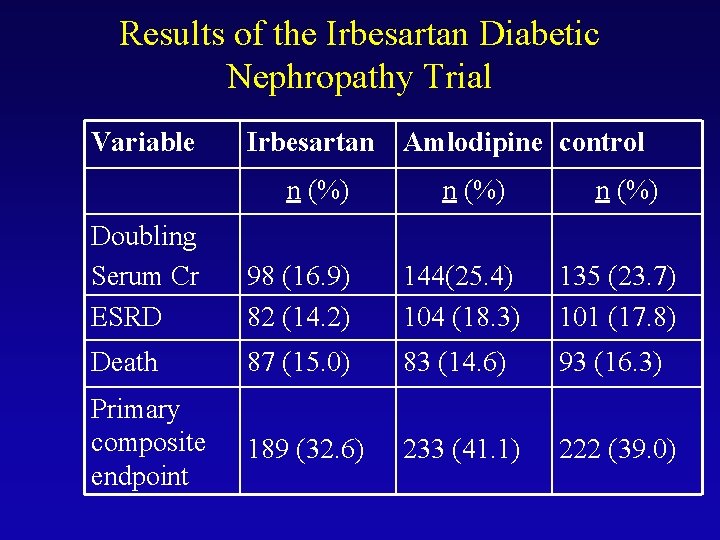
Results of the Irbesartan Diabetic Nephropathy Trial Variable Irbesartan n (%) Amlodipine control n (%) Doubling Serum Cr ESRD 98 (16. 9) 82 (14. 2) 144(25. 4) 104 (18. 3) 135 (23. 7) 101 (17. 8) Death 87 (15. 0) 83 (14. 6) 93 (16. 3) Primary composite endpoint 189 (32. 6) 233 (41. 1) 222 (39. 0)

Irbesartan Diabetic Nephropathy Trial Baseline N = Median proteinuria 1715 2. 9 G/24 hr (interquartile range 1. 6 -5. 2) Median albuminruia 1. 9 G/24 hr (interquartile range 1. 0. -3. 6) 12 months N = 50% decrease proteinuria 1550 29% irbesartan 40 amlodipine 20 control 25 Study end Doubling serum Cr ESRD first event Renal end point 377 10 387 (22%)

Is there an association between baseline proteinuria and outcome.

Irbesartan Diabetic Nephropathy Trial (IDNT) Association between baseline proteinuria and renal outcomes 24 hr urine protein at baseline % renal outcome at 36 months <1 G/day 7. 7% 1 -2 11. 4 2 -4 22. 9 4 -8 34. 3 >8 64. 9
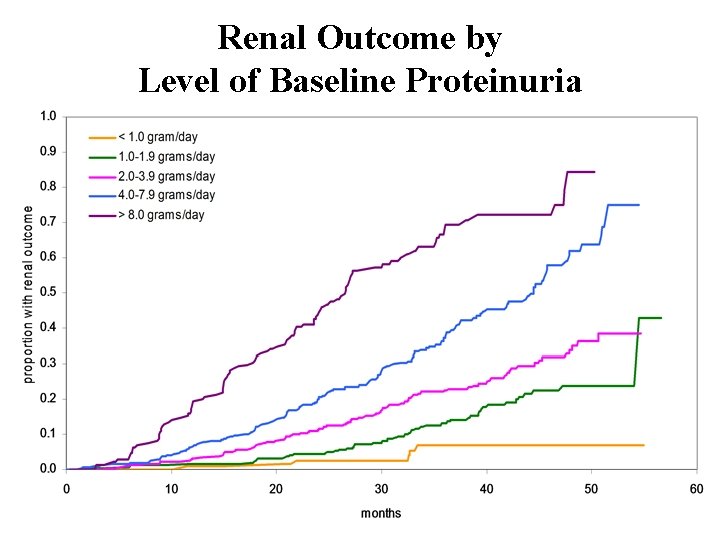
Renal Outcome by Level of Baseline Proteinuria

Irbesartan Diabetic Nephropathy Trial (IDNT) Association between baseline proteinuria and renal endpoints Doubling baseline proteinuria Hazard ratio 95% CI P 2. 04 1. 87 -2. 22 <0. 001


Was the amount of proteinuria altered during the course of the study?

Irbesartan Diabetic Nephropathy Trial (IDNT) Decrease in proteinuria at 12 months 95% CI Irbesartan (300 mg/day) 41% (37 -46) Amlodipine (10 mg/day) 11% (4 -17) Control 16% (9 -22) P <0. 001 0. 004 Proteinuria at 30 months Decrease Irbesartan 50% Amolidipine 23% Control 21%


Irbesartan Diabetic Nephropathy Trial (IDNT) 50% Decrease Proteinuria Levels at 12 Months Compared to Baseline Irbesartan 40% Amlodipine 20% (P<0. 001 vs IRB) Control (P<0. 001 vs IRB) 25%

Did alteration in the amount of proteinuria correlate with renal outcomes?

Irbesartan Diabetic Nephropathy Trial (IDNT) Impact of Change in Proteinuria at 36 Months Renal End-points Proteinuria (2 x Crs or ESRD) Greater than 50% reduction 9. 6% Zero to 50% reduction 26. 2% Zero to 50% increase 34. 5% More than 50% increase 38. 0%
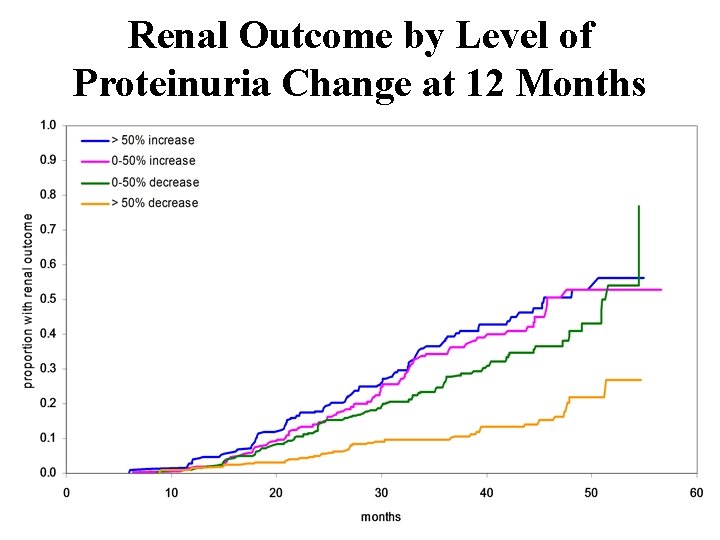
Renal Outcome by Level of Proteinuria Change at 12 Months

Irbesartan Diabetic Nephropathy Trial (IDNT) Reduction of Risk of Kidney Failure: Relationship to Halving of Proteinuria at 12 Months P vs those not halving Irbesartan (20 mg) 0. 39 (0. 32 -0. 47) <0. 001 Amlodipine (10 mg) 0. 49 (0. 42 -0. 58) <0. 001 Control 0. 44 (0. 38 -0. 51) <0. 001 • Amlodipine vs irbesartan p = 0. 48 • Control vs irb p = 0. 245

Decreased proteinuria is therefore a marker for an improved renal outcome. Can the proportion of the renoprotective effect of irbesartan which is associated with decreased proteinuria be estimated?
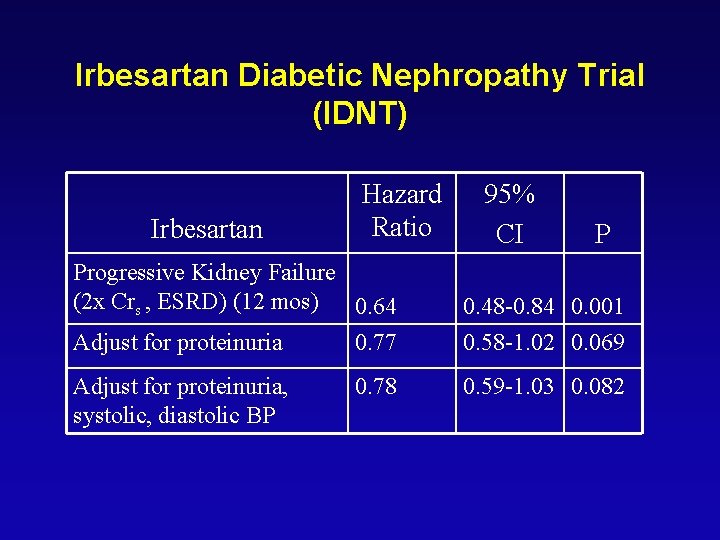
Irbesartan Diabetic Nephropathy Trial (IDNT) Irbesartan Hazard Ratio Progressive Kidney Failure (2 x Crs , ESRD) (12 mos) 0. 64 Adjust for proteinuria 0. 77 Adjust for proteinuria, systolic, diastolic BP 0. 78 95% CI P 0. 48 -0. 84 0. 001 0. 58 -1. 02 0. 069 0. 59 -1. 03 0. 082

Conclusions 1. Baseline proteinuria identifies patients at greatest risk for a poor renal outcome. 2. Proteinuria reduction is more common and greater when irbesartan is used in this patient population. 3. Halving proteinuria is associated with a marked decrease in the risk of a renal endpoint. 4. Proteinuria reduction in the first 12 months of follow-up accounts for some, but not all of the renoprotective effect of irbesartan.
- Slides: 24