IRB Process Effective January 19 th 2018 Institutional

IRB Process Effective January 19 th, 2018

Institutional Review Board • What is an IRB? • What is its job? • What are its guiding Principles? – Belmont Report (i. e. , Respect for Persons, Beneficence, Justice) – Code of Federal Regulations, Title 45 – Public Welfare, Part 46 Protection of Human Participants (aka The Common Rule) – USM Policies and Procedures, Section III and Section IV

Determine Whether Your Study Requires IRB Review IRB PROCESS STEP 1

What is Human Subjects’ Research? HUMAN SUBECT RESEARCH • Living individual about whom an investigator obtains, uses, studies, generates or analyzes: • Private information • Identifiable information • A systematic investigation, including research development, testing, and evaluation, designed to develop or contribute to generalizable knowledge • Through either • Intervention • Interaction

Ensure All Study Personnel Have Completed Human Subjects’ Protections Training IRB PROCESS STEP 2

Human Subjects’ Protections Training CITI PROGRAM • Complete all modules • Should take several hours • Score at least 80% • Print out and submit copy of Completion Certificate NOTE: Studies will not be submitted for review UNTIL evidence of Human Subjects’ Protection Training has been provided by ALL study personnel involved in data collection or analysis New User • Click on Register to set up a free account • Click on Affiliate with Another Institution; follow instructions • Under Towson University Courses, Click on Add a Course – Question 1: Responsible Conduct of Research – optional – Question 2: Skip – Question 3: Faculty/Staff Select Social & Behavioral Research OR Biomedical Research Students Conducting No More than Minimal Risk Research – Question 4: Not at this time, thank you

Determine Level of Review IRB PROCESS STEP 3

Minimal Risk • As defined in 45 CFR 46. 102. j, minimal risk. . . – …means that the probability and magnitude of harm or discomfort, anticipated in the research are not greater in and of themselves than those risks ordinarily encountered in daily life or during the performance of routine physical or psychological tests.
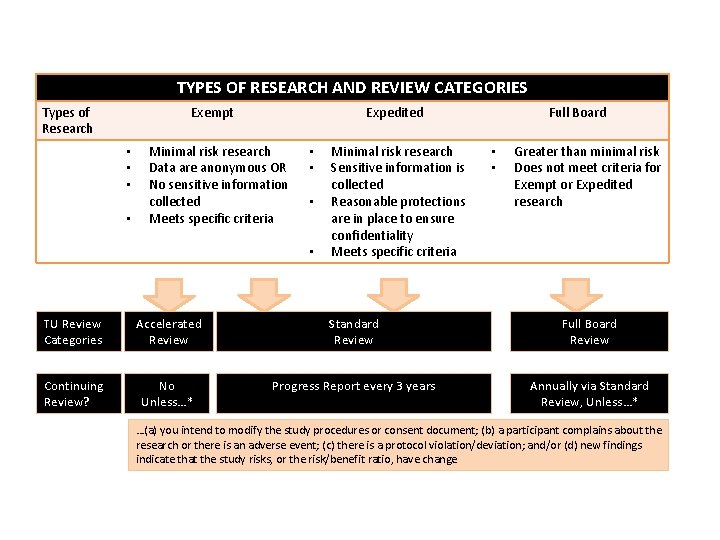
TYPES OF RESEARCH AND REVIEW CATEGORIES Types of Research Exempt • • Expedited Minimal risk research Data are anonymous OR No sensitive information collected Meets specific criteria • • Minimal risk research Sensitive information is collected Reasonable protections are in place to ensure confidentiality Meets specific criteria Full Board • • Greater than minimal risk Does not meet criteria for Exempt or Expedited research TU Review Categories Accelerated Review Standard Review Full Board Review Continuing Review? No Unless…* Progress Report every 3 years Annually via Standard Review, Unless…* …(a) you intend to modify the study procedures or consent document; (b) a participant complains about the research or there is an adverse event; (c) there is a protocol violation/deviation; and/or (d) new findings indicate that the study risks, or the risk/benefit ratio, have change
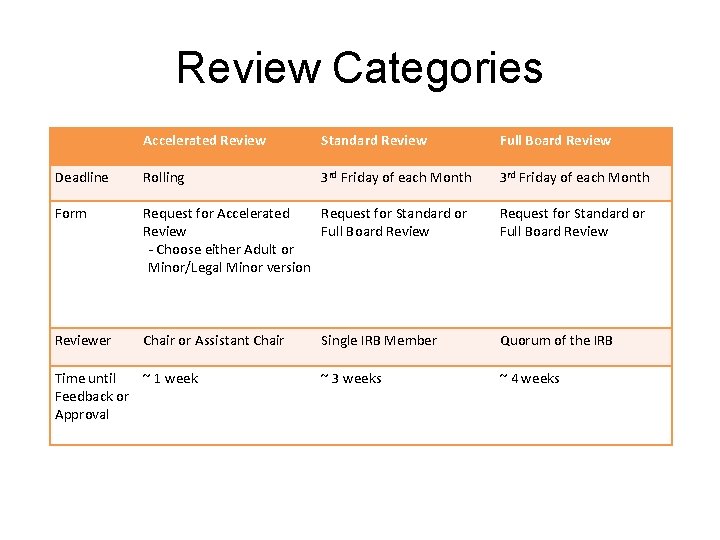
Review Categories Accelerated Review Standard Review Full Board Review Deadline Rolling 3 rd Friday of each Month Form Request for Accelerated Request for Standard or Review Full Board Review - Choose either Adult or Minor/Legal Minor version Request for Standard or Full Board Reviewer Chair or Assistant Chair Single IRB Member Quorum of the IRB ~ 3 weeks ~ 4 weeks Time until ~ 1 week Feedback or Approval

Complete and Submit the Application IRB PROCESS STEP 4

The Successful IRB Application • Provides detail about: – Study rationale as well as aims or hypotheses – Anticipated benefits – Inclusion/Exclusion criteria – Recruitment plan – How informed consent will be obtained – Study procedures (i. e. , in chronological order, what will subjects be asked to do) – Measures to be used – Anticipated risks and plans for mitigating or managing risks • If applicable, a statement that there are no foreseeable risks

The Successful IRB Application • Provides detail about: – How confidentiality or anonymity will be assured – How data will be stored to ensure security and who has access to data – If/when/how data will be destroyed – If/how subjects will be compensated • Informed Consent/Assent** – Includes all required elements (use TU Consent/Assent template) – 8 th grade reading level (adults) – Appropriate reading level (children/adolescents) – Sufficient detail so that participants understand risks and what is expected of them

The Successful IRB Application • At minimum, attach the following: – Informed consent form or information sheet – Measures (or detailed description of questions, if proprietary), interview questions – Human Subjects’ Protections Training certification – If student PI, Faculty Advisor signature • If appropriate also attach: – Informed assent form – Recruitment materials, including: • Content of e-mails or letters • Scripts for verbal recruitment • Fliers • Content of ads and where they will be placed – Links to relevant websites

Revising Your Application Based on Reviewer Comments IRB PROCESS STEP 5

Revising your Application • Address ALL reviewer concerns – Submit a cover letter with the reviewer’s comments and your response/revision • If you disagree with a suggested revision In your response, provide a detailed explanation for why you disagree with the comment; be advised the reviewer may still require the revision if the rationale is not convincing • Submit a revised protocol AND consent form – Use track changes, highlighting, or different colored font for revisions • DO NOT BEGIN DATA COLLECTION UNTIL APPROVAL FOR THE STUDY HAS BEEN GRANTED!! – Once approved, you will receive notification from the IRB office – you may begin data collection!

Amending Your Application After Approval IRB PROCESS STEP 6

Amending Applications • All substantive changes to consent forms or protocols MUST be reviewed and approved by the IRB – If not sure if change is substantive, ask • Submit – E-mail describing the change and its rationale – Protocol with track changes, highlighting, or different colored font – Consent form with track changes, highlighting, or different colored font – Include: new questionnaires; recruitment materials, etc • NOTE: You may not implement protocol changes

Some Final Thoughts What to do if. . . • • • I want to include 17 -year-old college students I want to pay participants I want to use data panels Someone complains about the research A participant experiences an adverse event There is a protocol deviation/violation NOTE: For more detailed information on all of these topics, see the Principal Investigator’s Instruction Manual which is available on the IRB website
- Slides: 19