IR spectroscopy of firstrow transition metal clusters and

![Depletion of elemental species in Interstellar Medium [10 -15 mbar] 10 -30 K Relative Depletion of elemental species in Interstellar Medium [10 -15 mbar] 10 -30 K Relative](https://slidetodoc.com/presentation_image_h2/615737104b7fa5700d94e6e17a9c1ecc/image-2.jpg)







- Slides: 26

IR spectroscopy of first-row transition metal clusters and their complexes with simple molecules Denis M. Kiawi FELIX facility, Radboud University Nijmegen, the Netherlands Anton Pannekoek Astronomical Institute, University of Amsterdam, the Netherlands
![Depletion of elemental species in Interstellar Medium 10 15 mbar 10 30 K Relative Depletion of elemental species in Interstellar Medium [10 -15 mbar] 10 -30 K Relative](https://slidetodoc.com/presentation_image_h2/615737104b7fa5700d94e6e17a9c1ecc/image-2.jpg)
Depletion of elemental species in Interstellar Medium [10 -15 mbar] 10 -30 K Relative atomic abundance (H=1) in -19 mbar] molecular cloud [10 dense 30 -100 K Williams & Herbst, Surf. Sci. 500, 823 (2002

In what form is Fe present in the ISM? Hypothesis: Fe. S formation • Fe → Fen • Fen + S→ Fen. Sm • n. Fe + m. S → Fen. Sm If true: Efficient conversion of Fe to Fe. S Clue: cometary fragments contain Fe. S grains Williams & Herbst, Surf. Sci. 500, 823 (2002)

Goals of my Ph. D project • Get an insight in interaction between Fe and S on the scale of clusters by studying: § Fen. Sm § Fen-SX (S-containing ligands) Methods: § IR spectroscopy geometric structure § DFT calculations Possible outcome § Insight in mechanism of Fe. S formation in ISM § Compare lab results with astronomical observations § Identification of IR spectrum of Fe/Fe. S species in the ISM

Why clusters? § Model systems for Fe particles in ISM § Isolated species in gas phase § Size range: n=3 -100 § Transition between atomic and bulk properties § Strong size-dependence of properties

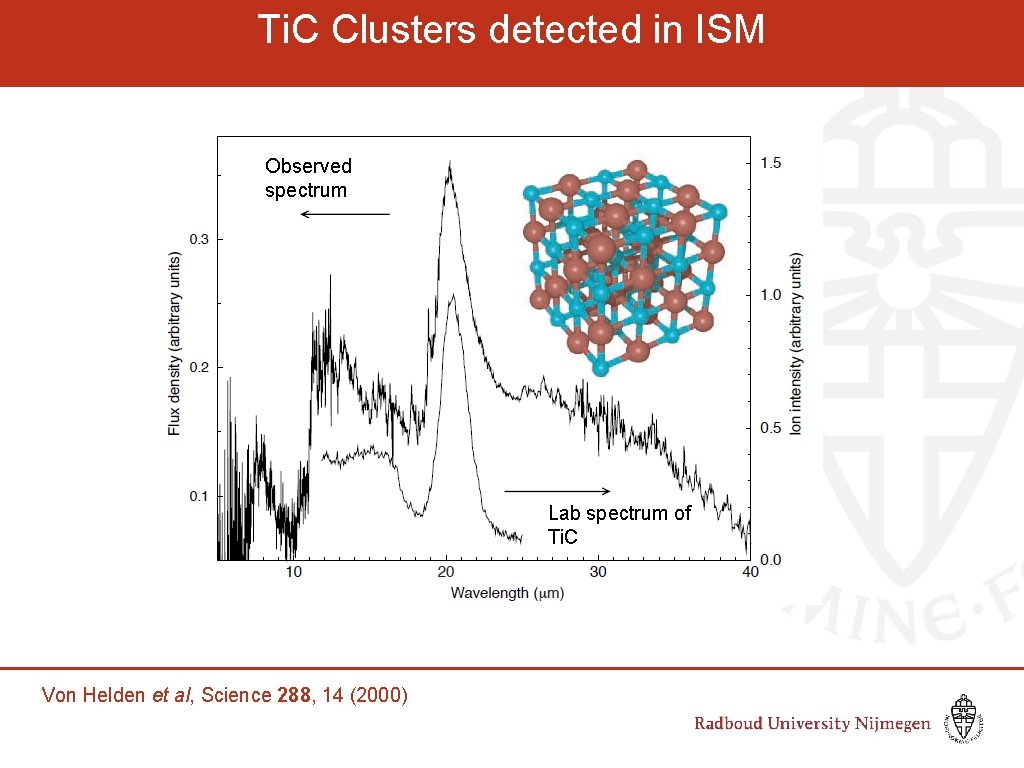
Ti. C Clusters detected in ISM Observed spectrum Lab spectrum of Ti. C Von Helden et al, Science 288, 14 (2000)

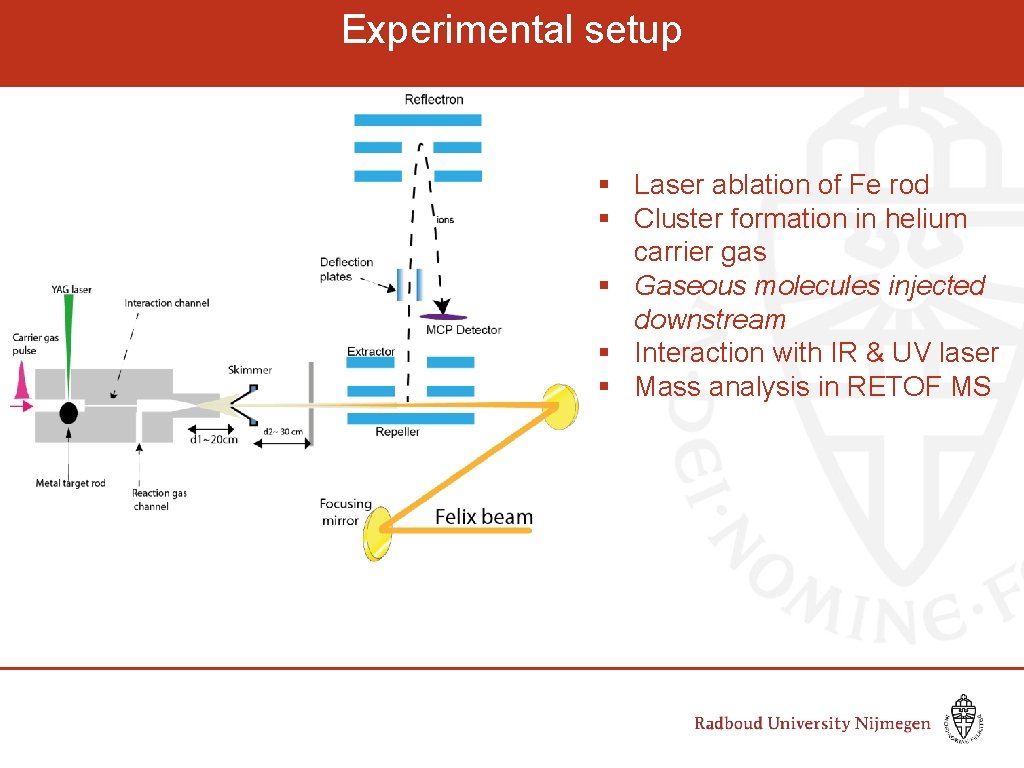
Experimental setup § Laser ablation of Fe rod § Cluster formation in helium carrier gas § Gaseous molecules injected downstream § Interaction with IR & UV laser § Mass analysis in RETOF MS

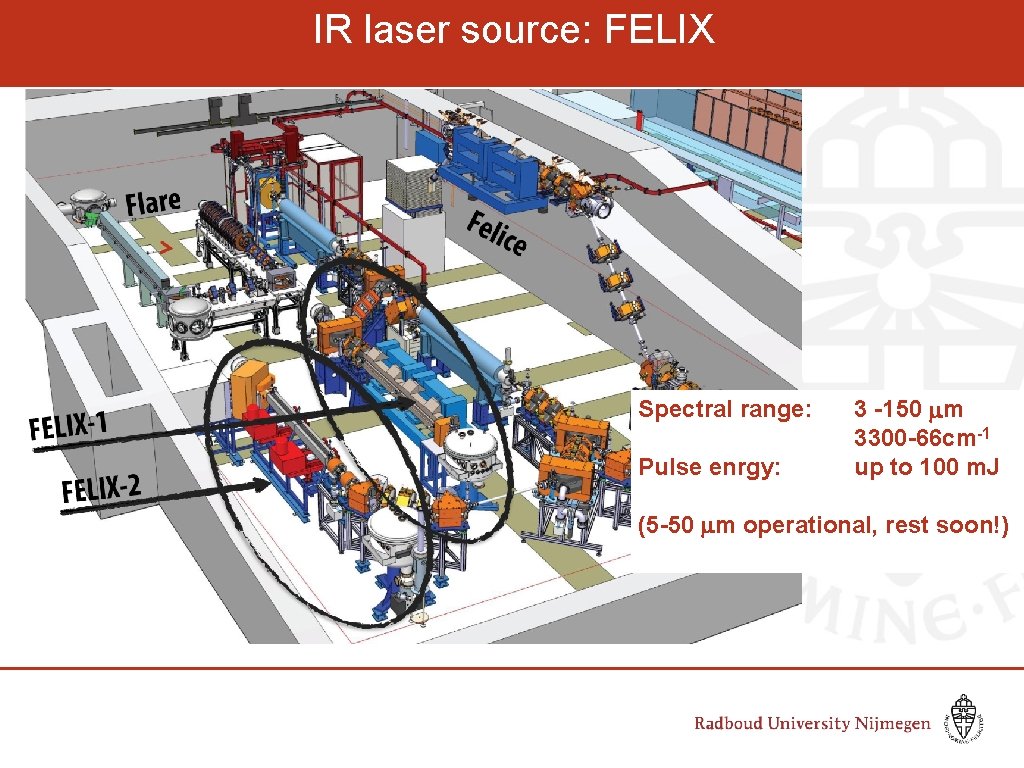
IR laser source: FELIX Inter-Cavity Spectral range: Pulse enrgy: 3 -150 mm 3300 -66 cm-1 up to 100 m. J (5 -50 mm operational, rest soon!)

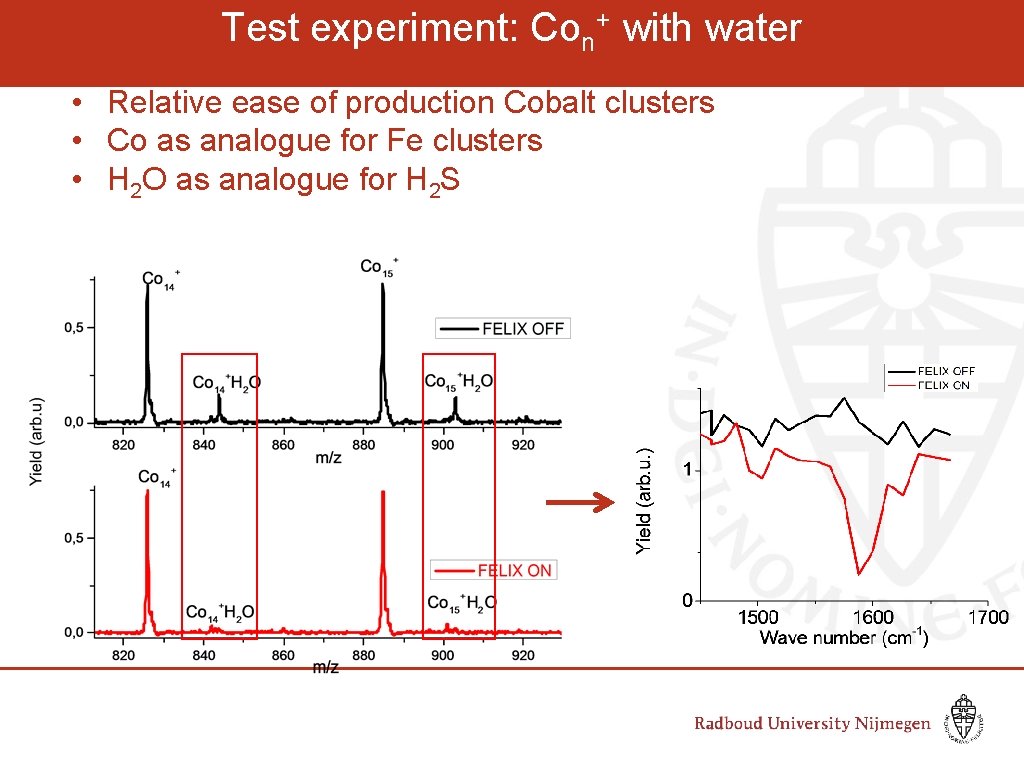
Test experiment: Con+ with water • Relative ease of production Cobalt clusters • Co as analogue for Fe clusters • H 2 O as analogue for H 2 S

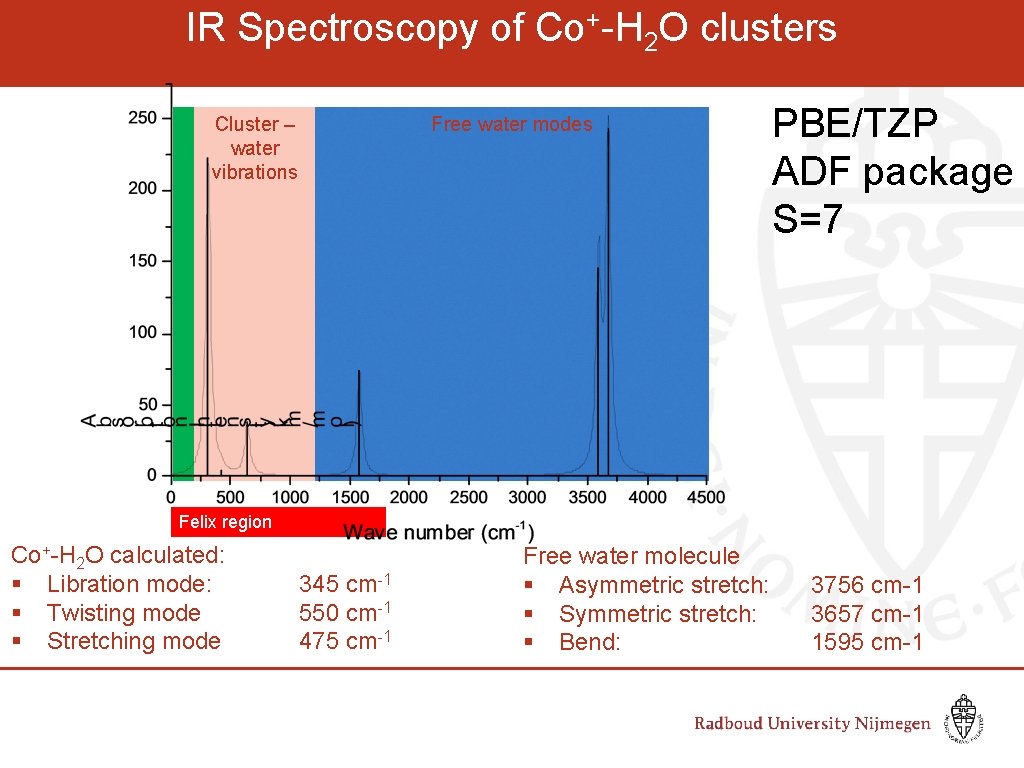
IR Spectroscopy of Co+-H 2 O clusters Cluster – water vibrations Free water modes PBE/TZP ADF package S=7 Felix region Co+-H 2 O calculated: § Libration mode: § Twisting mode § Stretching mode 345 cm-1 550 cm-1 475 cm-1 Free water molecule § Asymmetric stretch: § Symmetric stretch: § Bend: 3756 cm-1 3657 cm-1 1595 cm-1

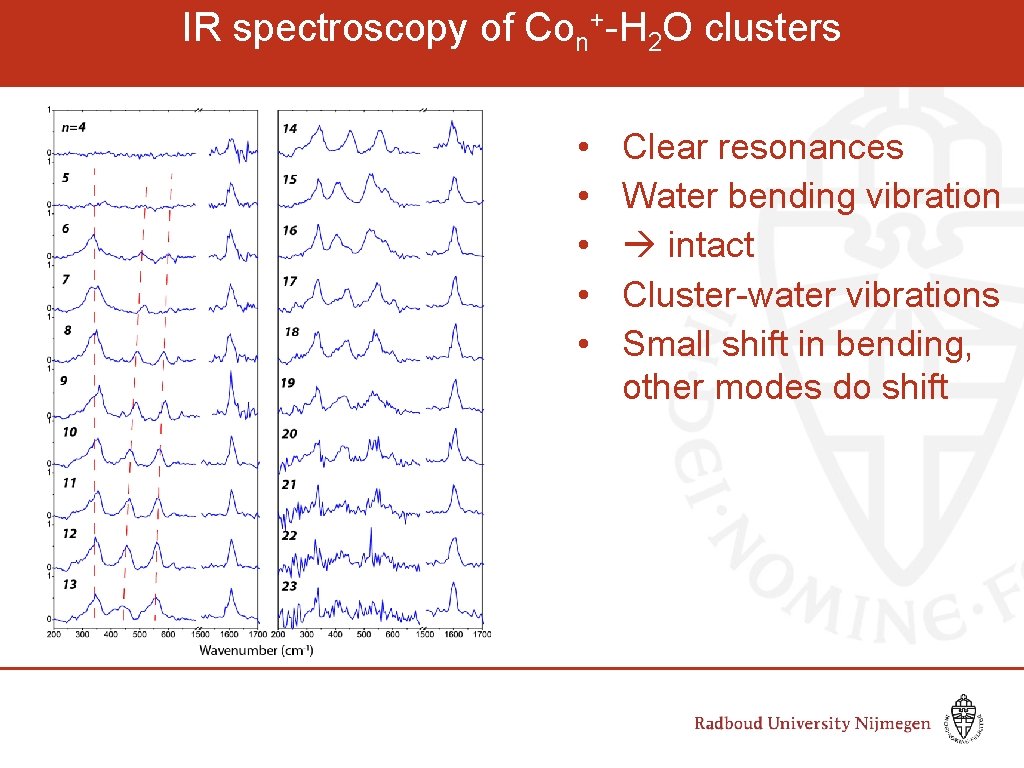
IR spectroscopy of Con+-H 2 O clusters • • • Clear resonances Water bending vibration intact Cluster-water vibrations Small shift in bending, other modes do shift

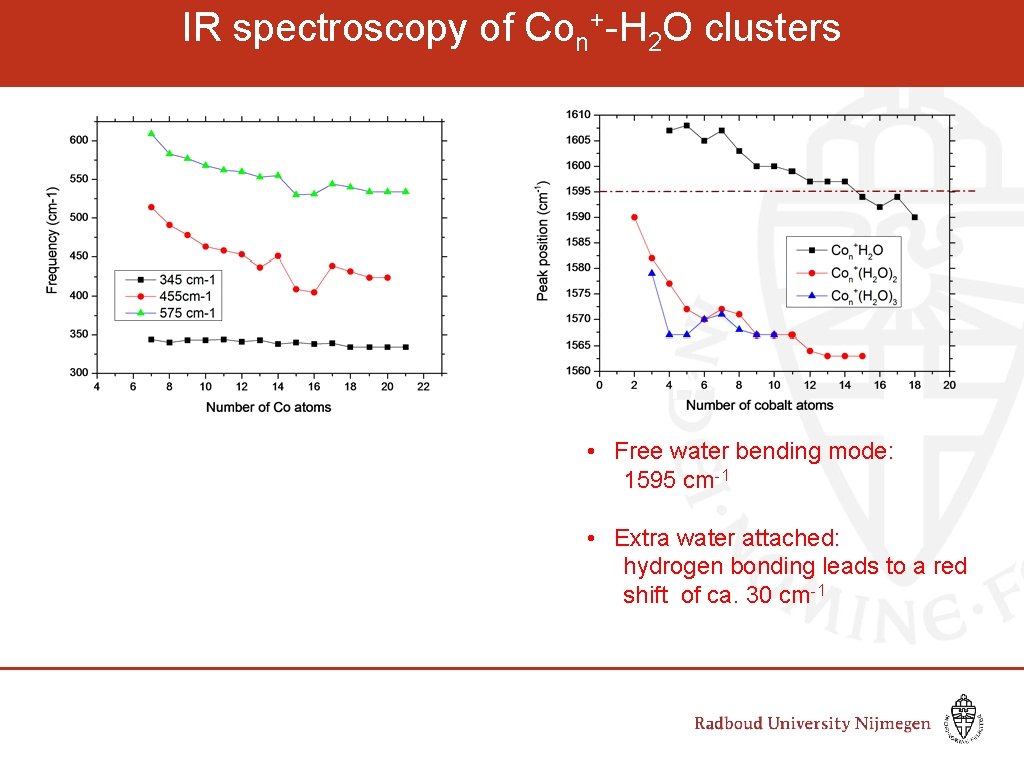
IR spectroscopy of Con+-H 2 O clusters Wave number (cm-1) • Free water bending mode: 1595 cm-1 • Extra water attached: hydrogen bonding leads to a red shift of ca. 30 cm-1

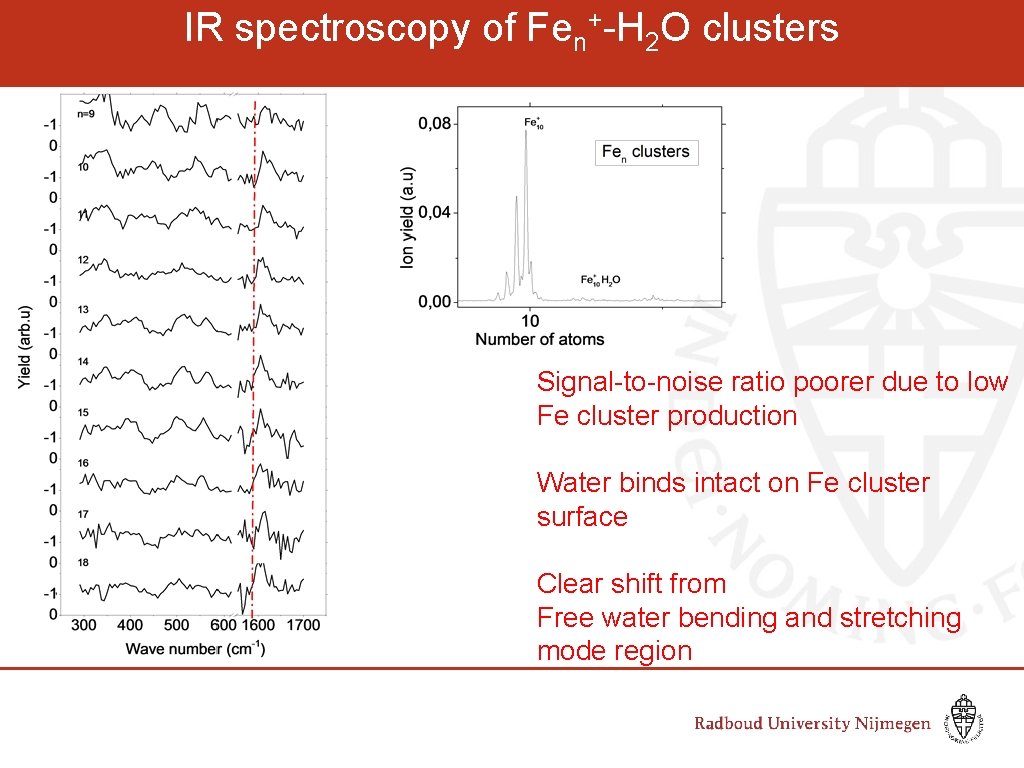
IR spectroscopy of Fen+-H 2 O clusters Signal-to-noise ratio poorer due to low Fe cluster production Water binds intact on Fe cluster surface Clear shift from Free water bending and stretching mode region

Conclusions • Astrophysical motivation to study Fe and Fe. S clusters • Laboratory IR spectra of Con+-H 2 O and Fen+-H 2 O clusters • H 2 O bending mode proves that water is bonded molecularly to the cluster • Clear size dependence of cluster-molecule vibrational frequencies Outlook: • DFT of Con+-H 2 O and Fen+-H 2 O • IR spectroscopy of § Fen-H 2 S § Fen. Sm § Fen

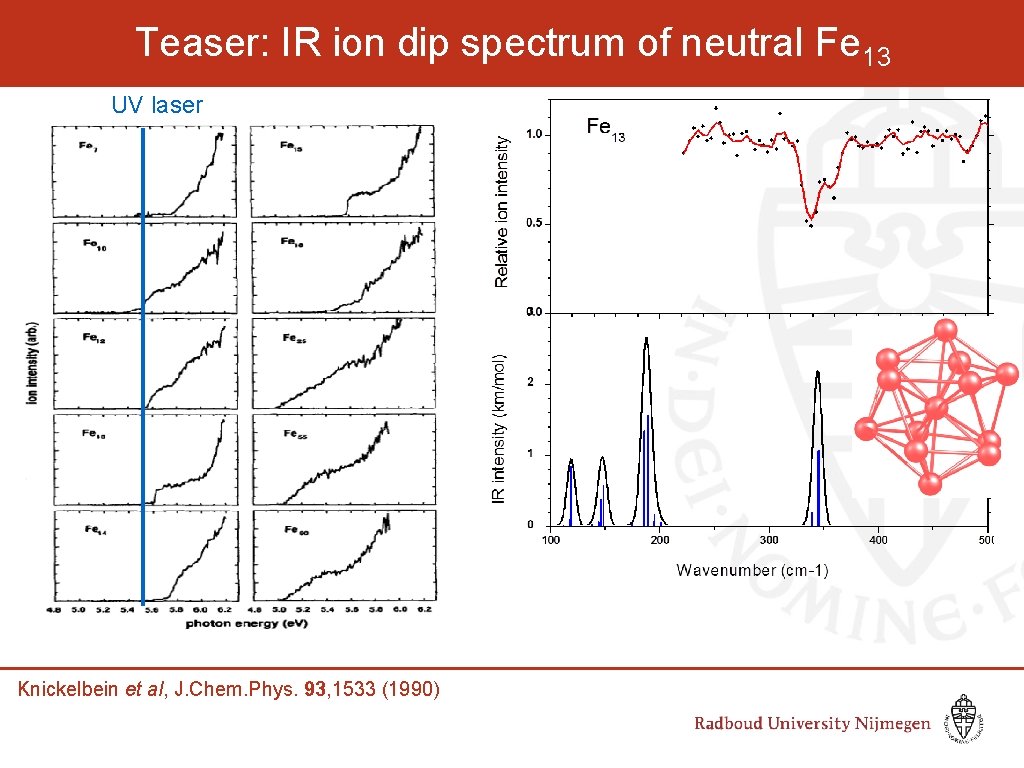
Teaser: IR ion dip spectrum of neutral Fe 13 UV laser Knickelbein et al, J. Chem. Phys. 93, 1533 (1990)

Acknowledgement Nijmegen Jos Oomens Joost Bakker + entire FELIX Team University of Amsterdam Rens Waters Wybren-Jan Buma Free University Amsterdam Luuk Visscher $:

The END

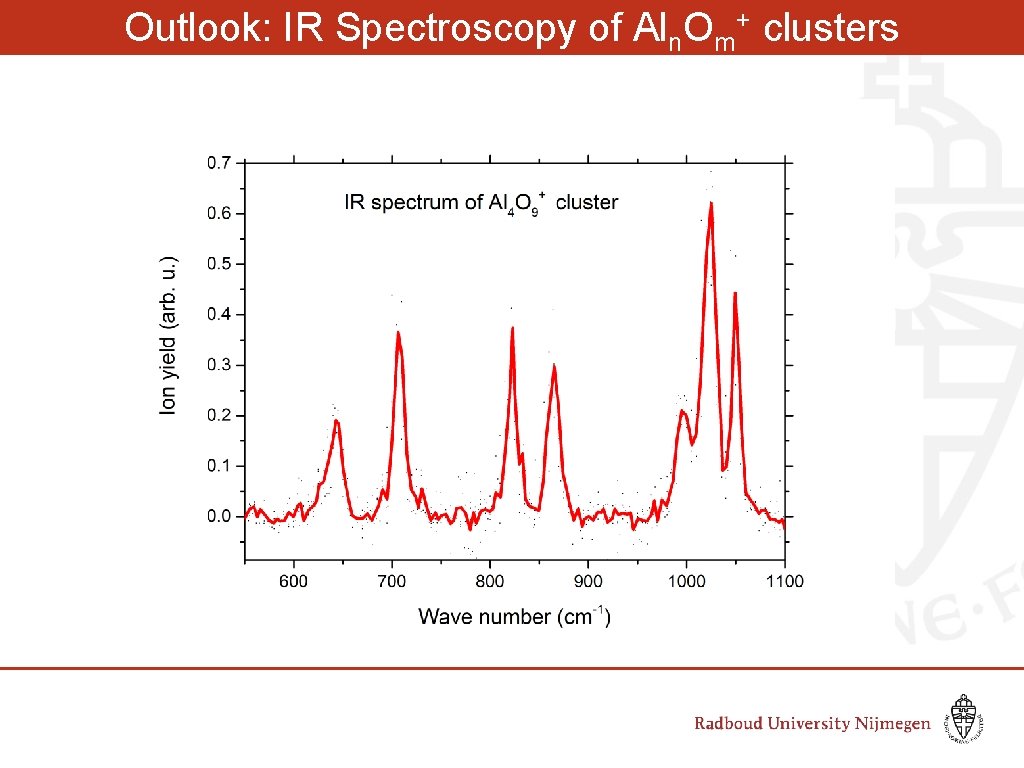
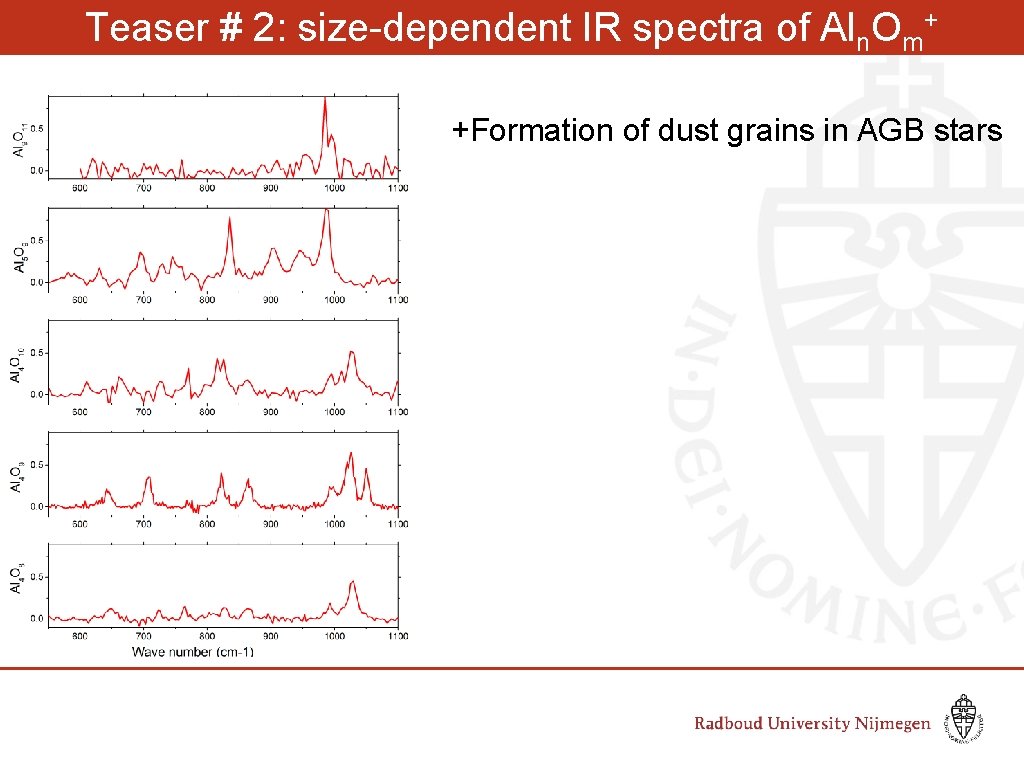
Outlook: IR Spectroscopy of Aln. Om+ clusters

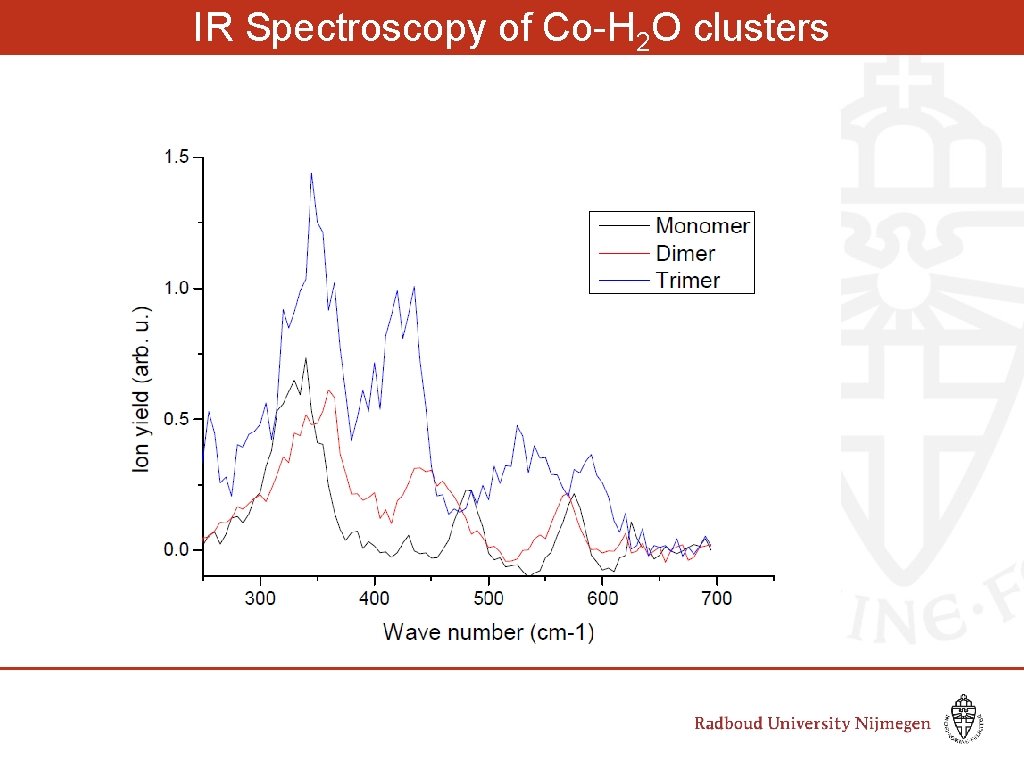
IR Spectroscopy of Co-H 2 O clusters Wave number (cm-1)

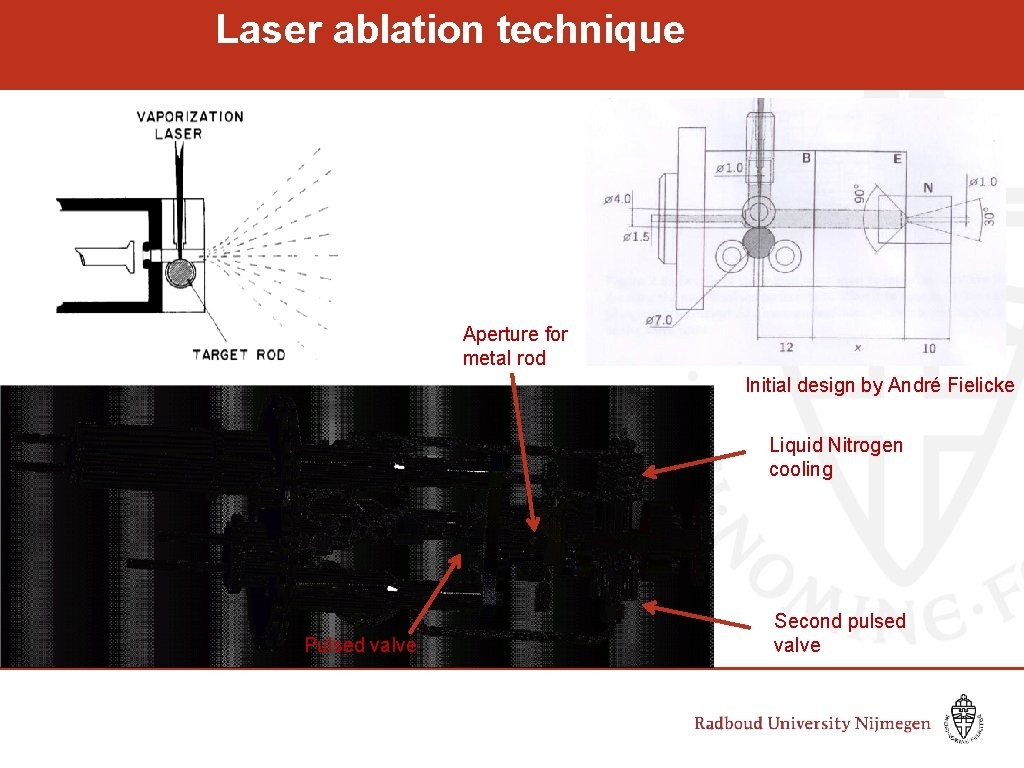
Laser ablation technique Aperture for metal rod Initial design by André Fielicke Liquid Nitrogen cooling Pulsed valve Second pulsed valve

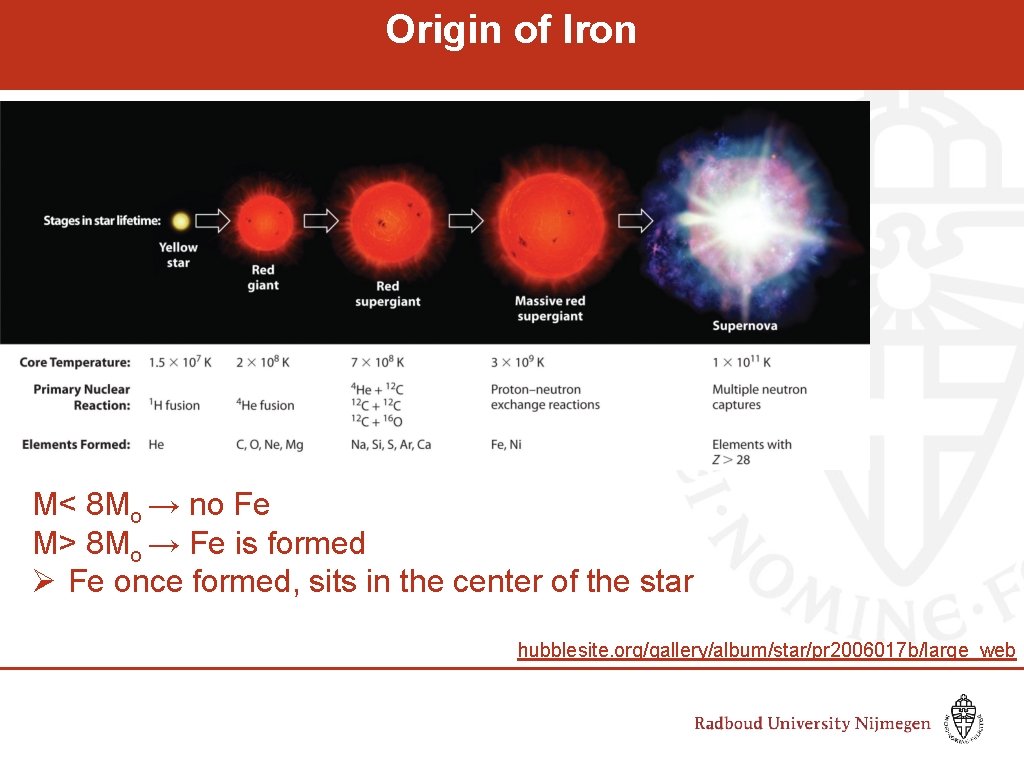
Origin of Iron M< 8 Mo → no Fe M> 8 Mo → Fe is formed Ø Fe once formed, sits in the center of the star hubblesite. org/gallery/album/star/pr 2006017 b/large_web

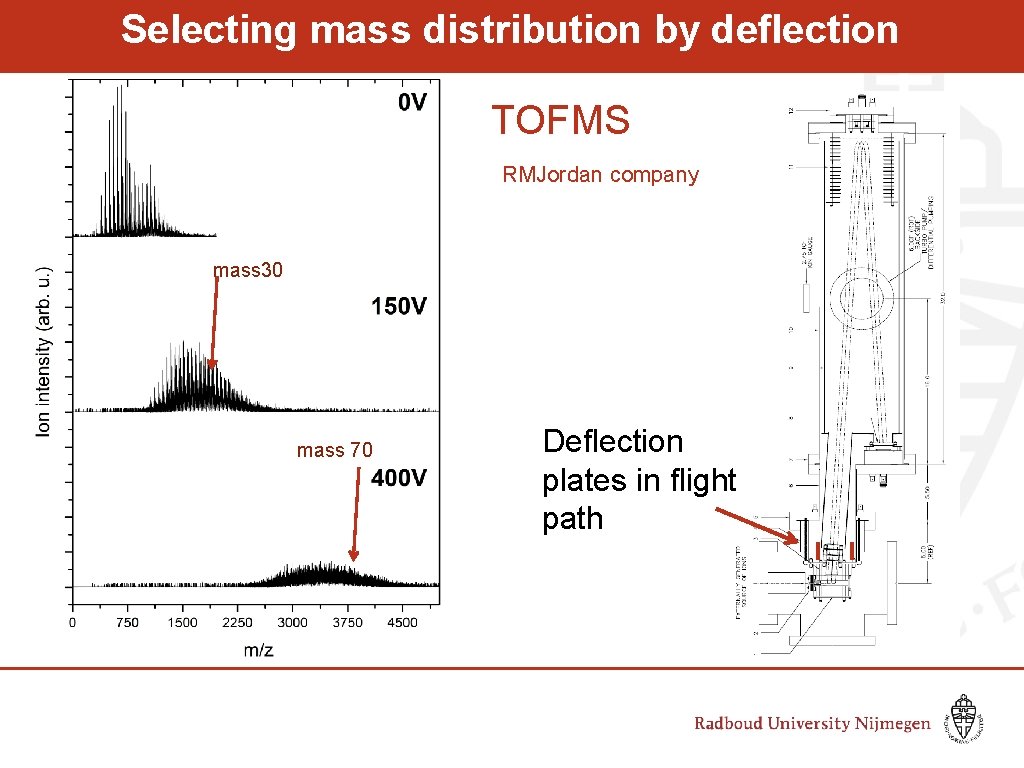
Selecting mass distribution by deflection TOFMS RMJordan company mass 30 mass 70 Deflection plates in flight path

Why is FEL is needed for such experiments ?

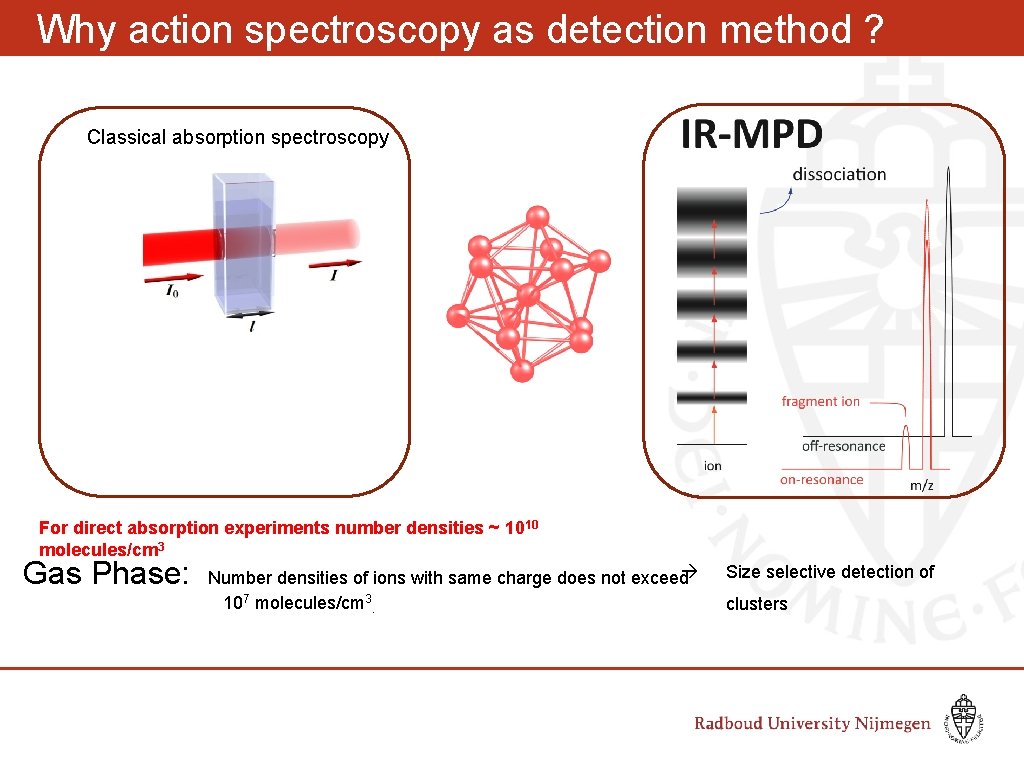
Why action spectroscopy as detection method ? Classical absorption spectroscopy For direct absorption experiments number densities ~ 1010 molecules/cm 3 Gas Phase: Number densities of ions with same charge does not exceed 10 7 molecules/cm 3. Size selective detection of clusters

DFT Calculations Theory Outcome • DFT geometry optimization • Evaluate many spin states! • Functionals - PBE - TPSS • Basis set - TZP • Analytical frequency calculations • Binding energy of various cluster geometries • IR spectra Commercial package ADF Amsterdam Density Functional

Teaser # 2: size-dependent IR spectra of Aln. Om+ +Formation of dust grains in AGB stars