Ions with Lewis Dot structures Abbreviating Lewis Dot

Ions with Lewis Dot structures

Abbreviating Lewis Dot Structures It can sometime be tedious writing out all the dots, and they can also be difficult to see. n So it is very common to use a dash to represent a pair of electrons. n So… n . . H : O. . : Cl: . . becomes H-O-Cl

If it’s a double bond single bond is a single dash, double bond is a double dash. n So CO 2 n O=C=O n Make sure bonds connect the elements, and unpaired electrons don’t connect to another element

Lewis dot structures with ions

First things first! Determine whether the bond will be ionic or covalent! n A nonmetal and a metal is ionic if they are both nonmetals it is covalent, hydrogen is not a metal. n To draw an ionic bond remove the electron(s) from the cation and add them to the anion. Show the. . charges on +each. . ion. n Na. . Cl: . . Na [: Cl: . . ]

For Polyatomic Ions… The polyatomic ion is a covalently bonded structure with an ionic bond to another substance. n NH 4 F n H. F H-N-H. H That goes to Fluoride It has an extra electron

For Polyatomic Ions… The polyatomic ion is a covalently bonded structure with an ionic bond to another substance. n NH 4 F n + H F H-N-H H That goes to Fluoride It has an extra electron

Polyatomic Ions we need to know
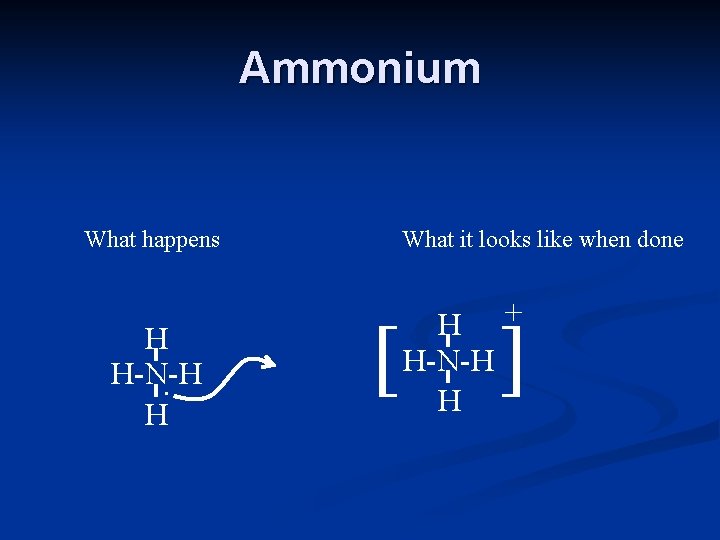
Ammonium What happens H H-N-H. H What it looks like when done [ H + H-N-H H ]
![Hypochlorite What happens . - - ] - [ -O-Cl- - - O-Cl - Hypochlorite What happens . - - ] - [ -O-Cl- - - O-Cl -](http://slidetodoc.com/presentation_image_h2/76939865f556efb246286fb090c42333/image-10.jpg)
Hypochlorite What happens . - - ] - [ -O-Cl- - - O-Cl - - What it looks like when done
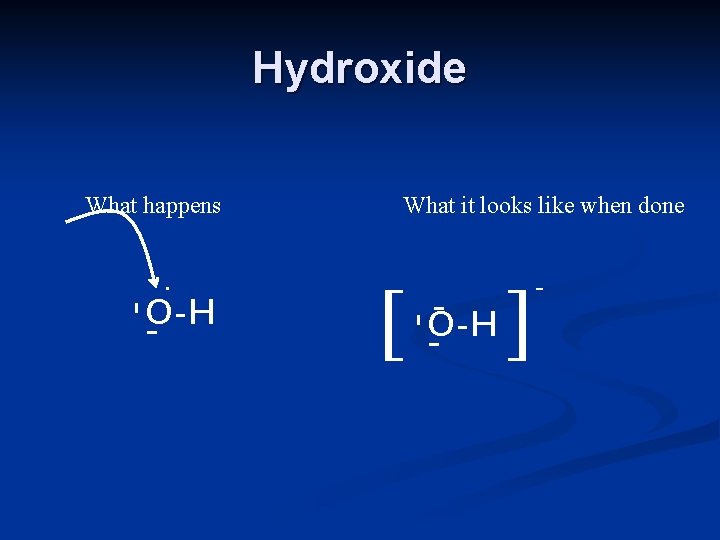
Hydroxide What happens . [ -O-H - - - O-H - What it looks like when done ] -
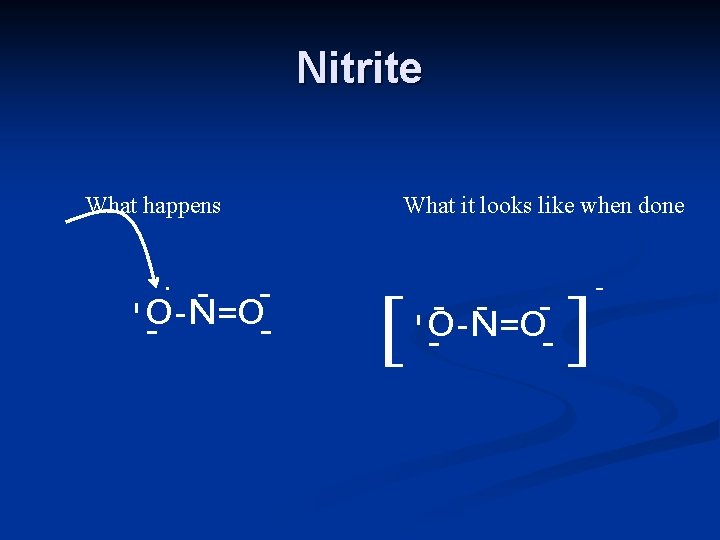
Nitrite What happens - O-N=O - [ -O-N=O - - - . - What it looks like when done ] -
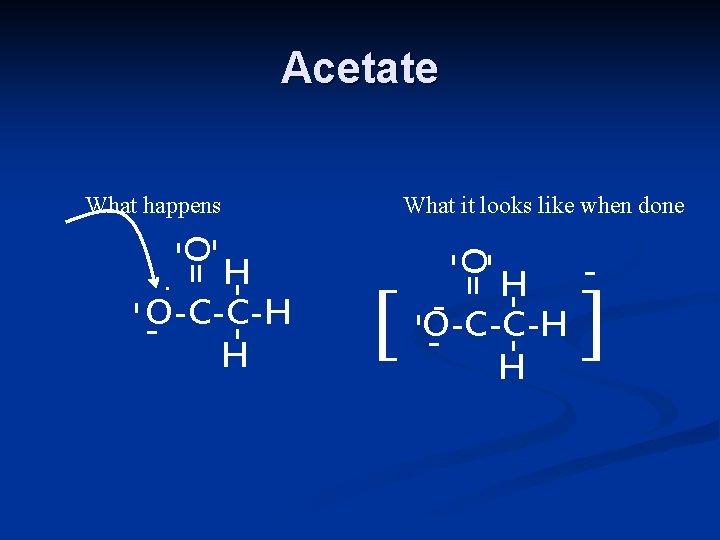
Acetate What happens - -O= - [ H O-C-C-H H - - -O= H O-C-C-H H. What it looks like when done - ]

Homework n Redo the Lewis Dot worksheet with dashes
- Slides: 14