Ions Continued Unit 3 Topic 2 Charges Because

Ions Continued Unit 3 Topic 2

Charges ¡ ¡ Because elements in the same group (column) of the Periodic Table have the same number of valence electrons They also tend to form ions with the same charge

Charges ¡ Elements in columns 1 -13 tend to undergo Oxidation (lose electrons) and form Positive ions. Oxidize (lose electrons) to form Positive Ions

Charges ¡ Elements in columns 15 -17 tend to undergo Reduction (gain electrons) and form Negative ions. Reduces (gains electrons) to form Negative Ions

Oxidation vs Reduction ¡ How am I going to remember these terms? l l l O -Oxidation I -Is L -Loss R -Reduction I -Is G -Gain

Oxidation States ¡ The common oxidation state (ion charge) for elements can be predicted by their position on the Periodic Table

Common Oxidation States (Charges) s e rg a h ~C y~ r a V Color Page 2 of your PT Coloring book. +1 +2 +3 + or -4 -3 -2 -1 0

Octet Rule When elements react by losing or gaining electrons, they will lose or gain the number of electrons needed to become more stable ¡ The most stable electron configuration is a completely filled energy level ¡ Which means you have a filled valence shell of 8 valence electrons known as the octet rule (except Hydrogen and Helium) ¡

8 is Great ¡ Which means you have a filled valence shell of 8 valence electrons known as the octet rule Or in the case of Hydrogen and Helium 2 electrons or zero electrons

Bonding Positive and negative ions are attracted to each other (because of their opposite charges) and combine to form ionic compounds. ¡ When ions combine to form compounds they must balance out their charges so that the compound ends up with no charge (electrically neutral) ¡

Predicting… ¡ We can predict the formulas for ionic compounds by determining the number of each ion (cation and anion) that is needed to cancel each other’s charges Note: The POSTIVE ion (cation) always goes FIRST!
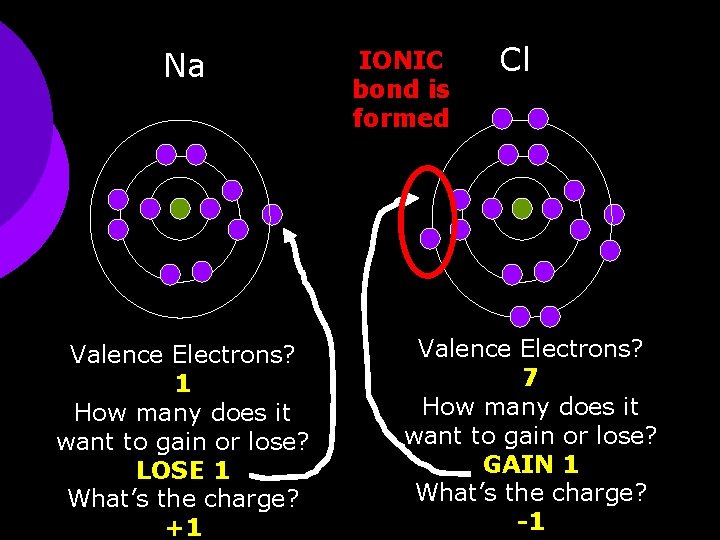
Na Valence Electrons? 1 How many does it want to gain or lose? LOSE 1 What’s the charge? +1 IONIC bond is formed Cl Valence Electrons? 7 How many does it want to gain or lose? GAIN 1 What’s the charge? -1

Sodium Chloride ¡ Na+1 + Cl-1 Na. Cl ¡ (+1) + (-1) = 0
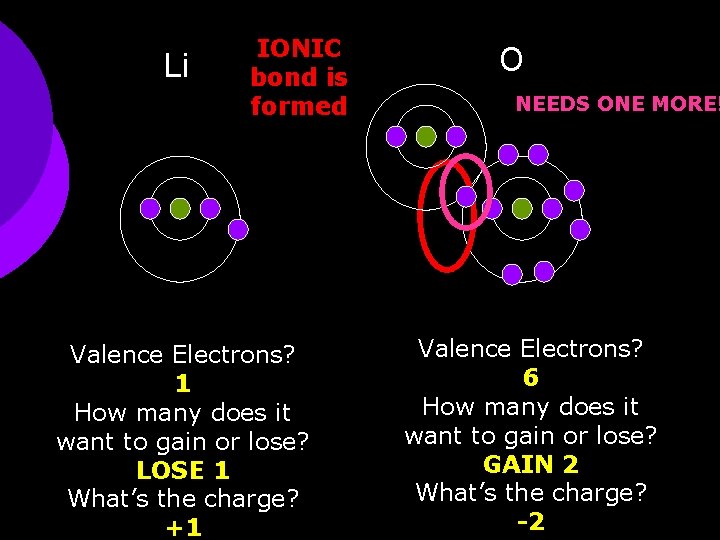
Li IONIC bond is formed Valence Electrons? 1 How many does it want to gain or lose? LOSE 1 What’s the charge? +1 O NEEDS ONE MORE! Valence Electrons? 6 How many does it want to gain or lose? GAIN 2 What’s the charge? -2

Lithium oxide ¡ Li+1 + O-2 Criss. Cross Method Li 2 O ¡ 2(+1) + 1(-2) = 0

Barium Iodide ¡ Ba+2 + I-1 Ba. I 2 ¡ (+2) + 2(-1) = 0

Magnesium phosphide ¡ Mg+2 + P-3 Mg 3 P 2 ¡ 3(+2) + 2(-3) = 0

Magnesium phosphide ¡ Mg+2 + P-3 Mg 3 P 2 ¡ 3(+2) + 2(-3) = 0

Ionic Bonding Ionic bonding occurs when a metal transfers one or more electrons to a nonmetal in an effort to attain a stable octet of electrons. For example, the transfer of an electron from sodium to chlorine can be depicted by a LDD. Cl Na Cl Ca Cl
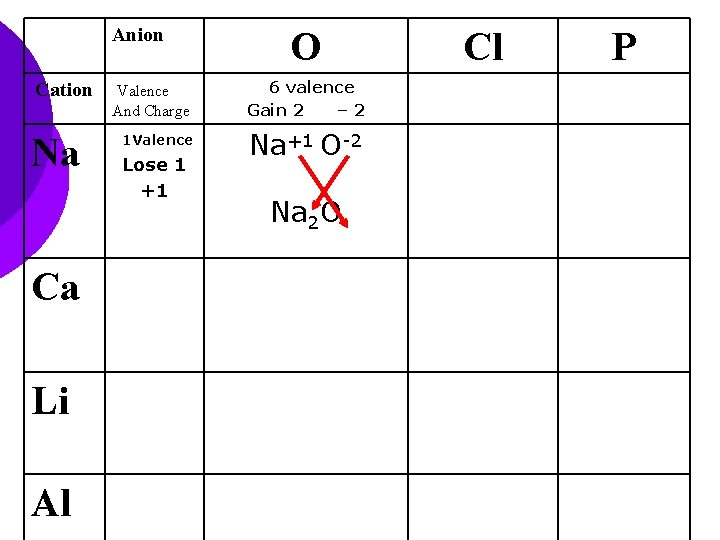
Anion Cation Na Ca Li Al O Valence And Charge 6 valence Gain 2 – 2 1 Valence Na+1 O-2 Lose 1 +1 Na 2 O Cl P
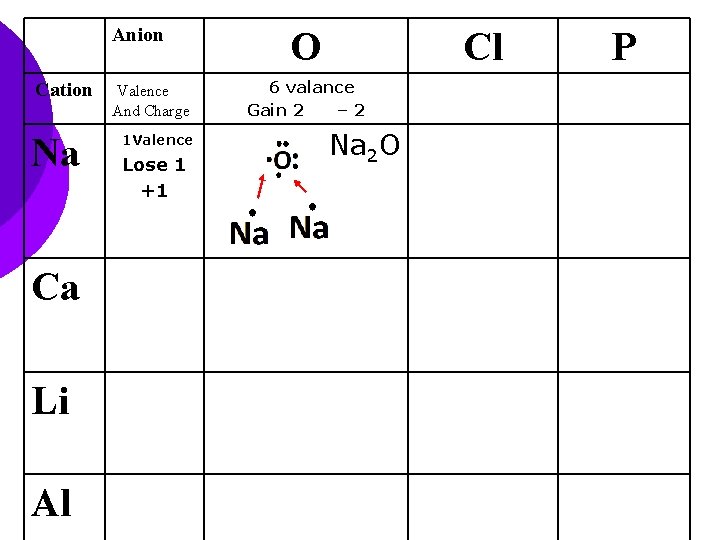
Anion Cation Na Ca Li Al Valence And Charge 1 Valence Lose 1 +1 O Cl 6 valance Gain 2 – 2 Na 2 O P

Naming Ionic Compounds What is an Ionic Compound? ¡ Positive Metal + Negative Nonmetal ¡ To name compounds: ¡ Positive Metal name + Negative nonmetal name (drop ending and add “ide” ¡ Na. Cl = sodium chloride ¡ Ba 3 P 2 = barium phosphide ¡
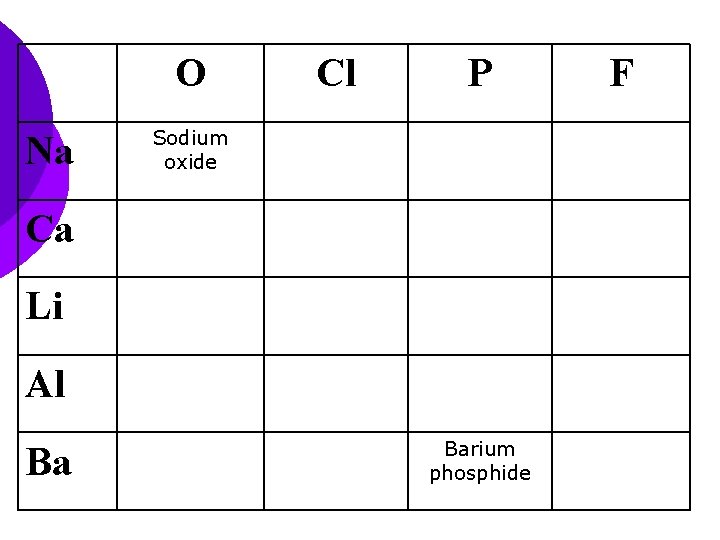
O Na Cl P Sodium oxide Ca Li Al Ba Barium phosphide F
- Slides: 23