Ions and Ionic bonding Ionic Compounds metal nonmetal

Ions and Ionic bonding

Ionic Compounds (metal + non-metal) The main goal an atom has is to obtain a full outer shell. To do this atoms must lose or gain electrons, thereby becoming ions. Ions are atoms with charges. When a metal ion and a non-metal ion combine an ionic bond is formed. An ionic bond contains a positively charged cation and a negatively charged anion. Electrostatic forces of attraction are in place. Cations are metal ions that have a positive charge and have lost electrons in order to obtain a full outer shell. Anions are non-metal ions that have a negative charge and have gained electrons in order to obtain a full outer shell. Sodium (Na) must lose 1 electron in order to gain a full outer shell. Chlorine (Cl) must gain an electron in order to gain a full outer shell.

Ionic Compounds (metal + non-metal) Ionic compounds have the following properties in common: They are usually solids at room temperature They normally have high melting and boiling points They dissolve in water to form aqueous solutions These aqueous solutions conduct electricity
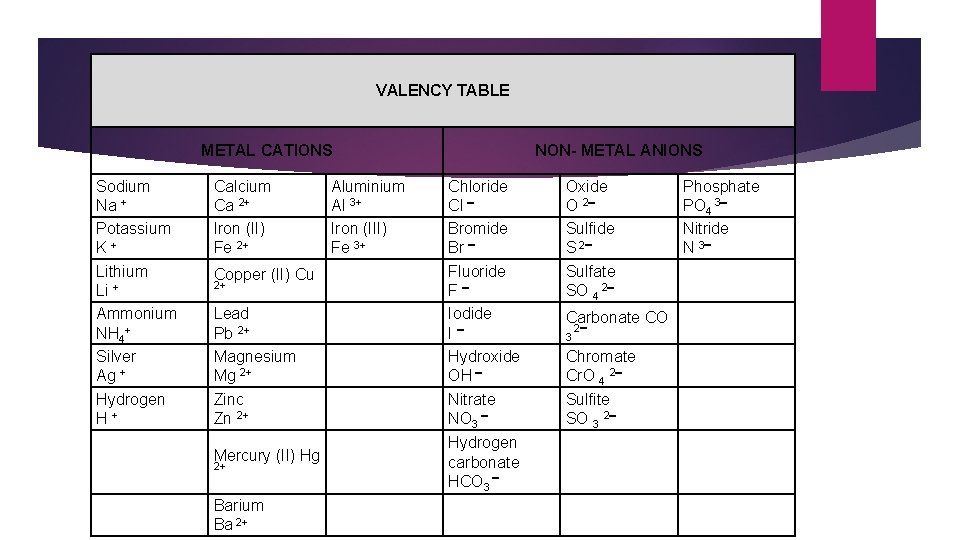
VALENCY TABLE Ionic Compounds METAL CATIONS Sodium Na + Potassium K+ Lithium Li + Ammonium NH 4+ Silver Ag + Hydrogen H+ Calcium Ca 2+ Iron (II) Fe 2+ Copper (II) Cu 2+ Lead Pb 2+ Magnesium Mg 2+ Zinc Zn 2+ Mercury (II) Hg 2+ Barium Ba 2+ Aluminium Al 3+ Iron (III) Fe 3+ NON- METAL ANIONS Chloride Cl Bromide Br Fluoride F Iodide I Hydroxide OH Nitrate NO 3 Hydrogen carbonate HCO 3 Oxide O 2 Sulfide S 2 Sulfate SO 4 2 Carbonate CO 3 2 Chromate Cr. O 4 2 Sulfite SO 3 2 Phosphate PO 4 3 Nitride N 3

Finding the right formula- Ionic Compounds: 1. Use the electrovalency (shows the positive or negative charges of an ion) to help you work out how many electrons need to be gained or lost in order to obtain a full outer shell for each element in the compound. 2. Determine the ratio of ions required to create electrically neutral compounds by using the SWAP & DROP method. The number at the top of each ion is dropped to the bottom of the opposite ion ignoring the charge. 3. Write the formula for the compound using the numbers in the ratios as the subscripts.

Writing the right formula when forming Ionic Compounds (metal cation + non-metal anion) Swap and drop rules: 1. If the two ions have the same number but opposite charge then they come together at a 1: 1 ratio. E. g. Na+ Cl- =Na. Cl Sodium Chloride, Ca 2+ O 2 - =Ca. O Calcium Oxide, Al 3+ N 3 -=Al. N Aluminium Nitride

Writing the right formula when forming Ionic Compounds (metal cation + non-metal anion) 2. If the two ions have different numbers but opposite charges then you need to take the number from the top of the cation and drop it down the bottom of the anion. Then take the number from the top of the anion and drop it down the bottom of the cation. E. g. Mg 2+ N 3 - = Mg 3 N 2 Magnesium Nitride Na+ O 2 - = Na 2 O Sodium Oxide

Writing the right formula when forming Ionic Compounds (metal cation + non-metal anion) 3. If one of the ions is a polyatomic ion (more than 1 element bound together) and you need more than 1 of it then you must put the whole thing in brackets and put the amount you need outside the brackets. This can occur for both cations and anions. E. g. NH 4+ PO 43 - = (NH 4)3 PO 4 Ammonium Phosphate Zn 2+ HCO 3 - = Zn(HCO 3)2 Zinc Hydrogen Carbonate

Writing the right formula when forming Ionic Compounds (metal cation + non-metal anion) When non-metals become anions they change their suffix to –ide. Nothing happens to the cation’s names. Transition metals charge is determined by the roman numeral in the brackets Lead (III) will be Pb 3+
- Slides: 9