IONISATION ENERGY OBJECTIVES To define the term ionisation

IONISATION ENERGY OBJECTIVES: To define the term ‘ionisation energy’ To describe and explain the trends in ionisation energy across period 3 and down group 2 FIRST THOUGHTS… KEY WORDS: What do you think we might mean by ionisation energy? IONISATION ENERGY NUCLEAR CHARGE TREND

IONISATION ENERGY I CAN… I AM… State what is meant by ionisation energy C Describe the trend across period 3 and down group 2 B Explain why this trend occurs A Suggest how these trends provide evidence for the existence of electron levels and sub-levels A*

IONISATION ENERGY WHAT IS IONISATION ENERGY? “The energy required to remove 1 electron from each atom in 1 mole of gaseous atoms to form 1 mole of gaseous 1+ ions” Energy required to remove electrons Measured in k. Jmol-1 Given the abbreviation IE Values tell us a lot about the electronic configuration of elements
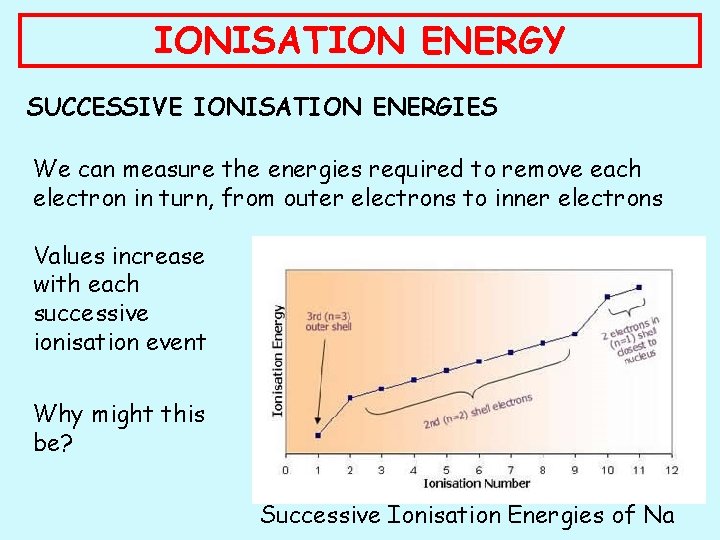
IONISATION ENERGY SUCCESSIVE IONISATION ENERGIES We can measure the energies required to remove each electron in turn, from outer electrons to inner electrons Values increase with each successive ionisation event Why might this be? Successive Ionisation Energies of Na
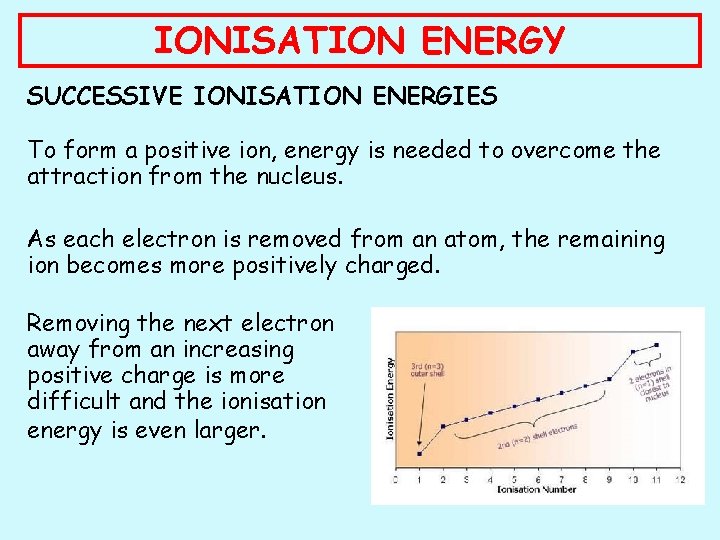
IONISATION ENERGY SUCCESSIVE IONISATION ENERGIES To form a positive ion, energy is needed to overcome the attraction from the nucleus. As each electron is removed from an atom, the remaining ion becomes more positively charged. Removing the next electron away from an increasing positive charge is more difficult and the ionisation energy is even larger.
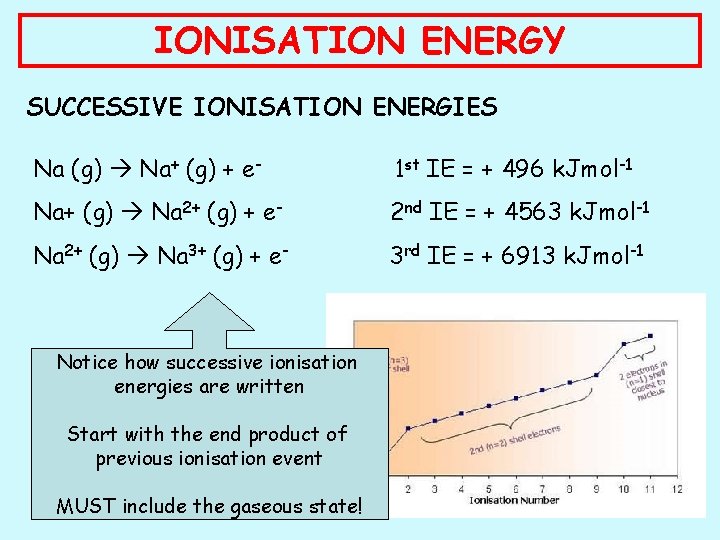
IONISATION ENERGY SUCCESSIVE IONISATION ENERGIES Na (g) Na+ (g) + e- 1 st IE = + 496 k. Jmol-1 Na+ (g) Na 2+ (g) + e- 2 nd IE = + 4563 k. Jmol-1 Na 2+ (g) Na 3+ (g) + e- 3 rd IE = + 6913 k. Jmol-1 Notice how successive ionisation energies are written Start with the end product of previous ionisation event MUST include the gaseous state!
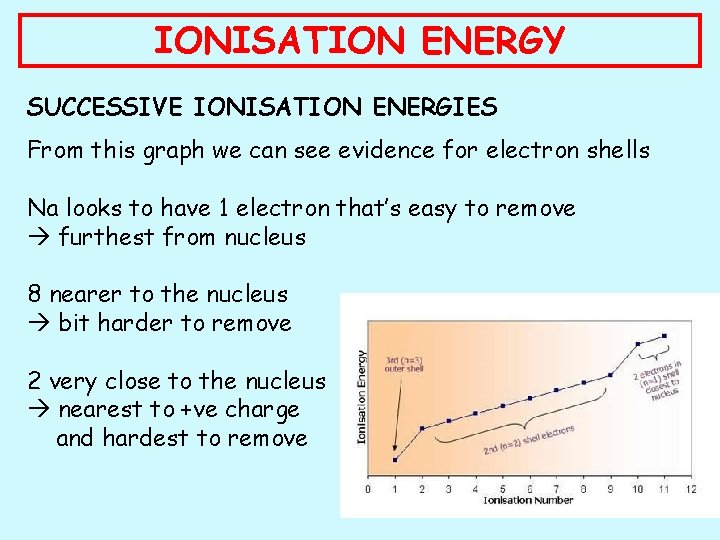
IONISATION ENERGY SUCCESSIVE IONISATION ENERGIES From this graph we can see evidence for electron shells Na looks to have 1 electron that’s easy to remove furthest from nucleus 8 nearer to the nucleus bit harder to remove 2 very close to the nucleus nearest to +ve charge and hardest to remove

IONISATION ENERGY FACTORS AFFECTING IONISATION ENERGY As electrons are negatively charged and protons in the nucleus are positively charged, there will be an attraction between them. The greater the pull of the nucleus, the harder it will be to pull an electron away from an atom. Nuclear attraction of an electron depends on: • Atomic radius • Nuclear Charge • Electron shielding or screening

IONISATION ENERGY ATOMIC RADIUS: Greater the atomic radius, the smaller the nuclear attraction experienced by the outer electrons NUCLEAR CHARGE: The greater the nuclear charge, the greater the attractive force on the outer electrons Hydrogen 1310 k. J mol-1 Helium Lithium 2370 k. J mol-1 519 k. J mol-1

IONISATION ENERGY ELECTRON SHIELDING Inner shells of electrons repel the outer-shell electrons Known as ‘Electron shielding’ or ‘screening’ More inner shells larger the screening and smaller the nuclear attraction of outer electrons Hydrogen 1310 k. J mol-1 Helium Lithium 2370 k. J mol-1 519 k. J mol-1
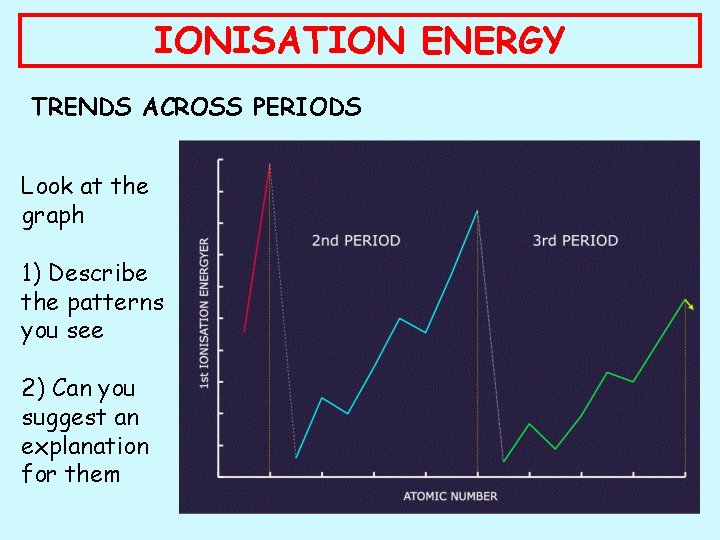
IONISATION ENERGY TRENDS ACROSS PERIODS Look at the graph 1) Describe the patterns you see 2) Can you suggest an explanation for them
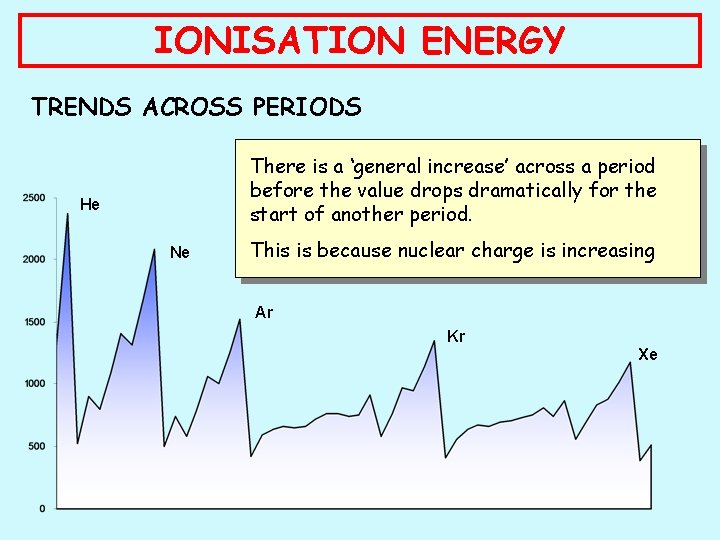
IONISATION ENERGY TRENDS ACROSS PERIODS There is a ‘general increase’ across a period before the value drops dramatically for the start of another period. He Ne This is because nuclear charge is increasing Ar Kr Xe
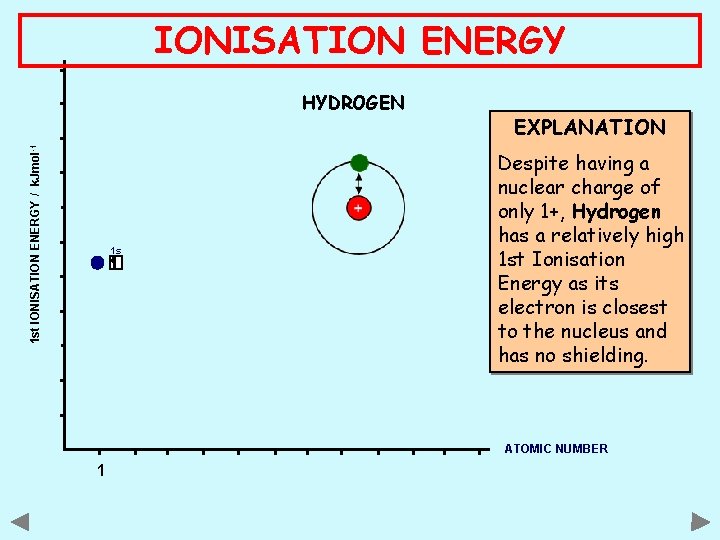
IONISATION ENERGY 1 st IONISATION ENERGY / k. Jmol -1 HYDROGEN 1 s EXPLANATION Despite having a nuclear charge of only 1+, Hydrogen has a relatively high 1 st Ionisation Energy as its electron is closest to the nucleus and has no shielding. ATOMIC NUMBER 1
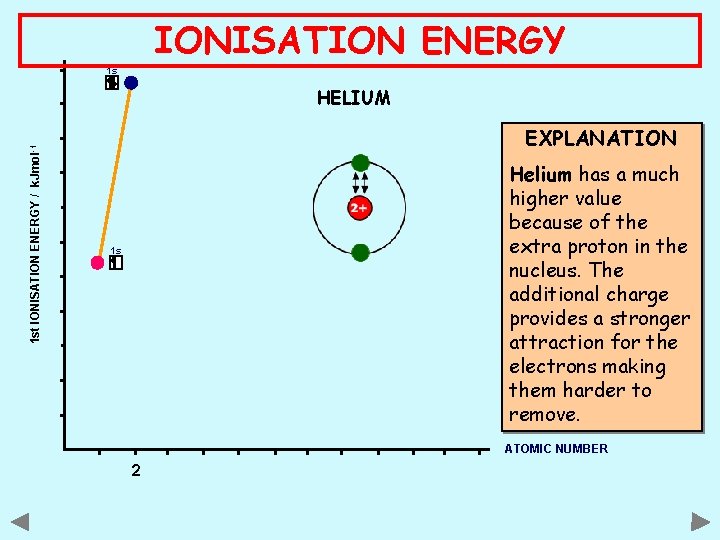
IONISATION ENERGY 1 s 1 st IONISATION ENERGY / k. Jmol -1 HELIUM EXPLANATION Helium has a much higher value because of the extra proton in the nucleus. The additional charge provides a stronger attraction for the electrons making them harder to remove. 1 s ATOMIC NUMBER 2
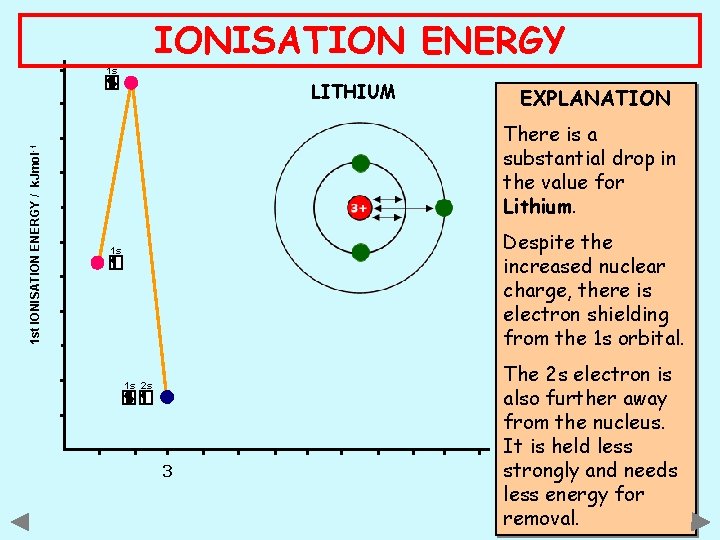
IONISATION ENERGY 1 s 1 st IONISATION ENERGY / k. Jmol -1 LITHIUM EXPLANATION There is a substantial drop in the value for Lithium. Despite the increased nuclear charge, there is electron shielding from the 1 s orbital. 1 s 1 s 2 s 3 The 2 s electron is also further away from the nucleus. ATOMIC NUMBER It is held less strongly and needs less energy for removal.
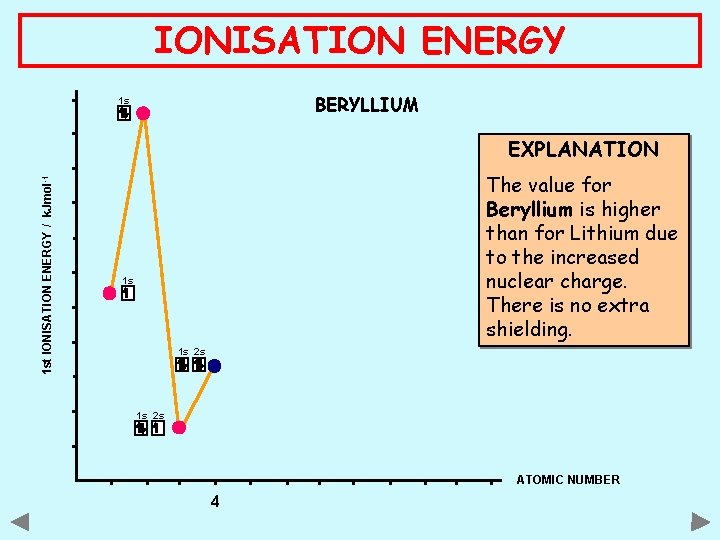
IONISATION ENERGY BERYLLIUM 1 s 1 st IONISATION ENERGY / k. Jmol -1 EXPLANATION The value for Beryllium is higher than for Lithium due to the increased nuclear charge. There is no extra shielding. 1 s 1 s 2 s ATOMIC NUMBER 4
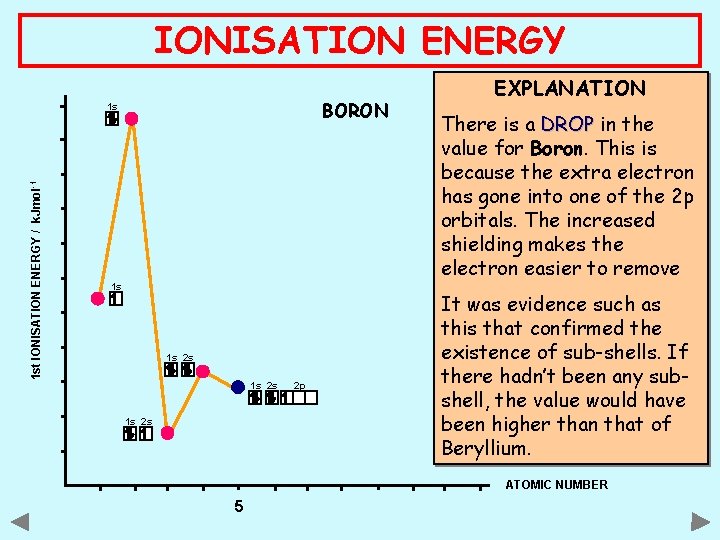
IONISATION ENERGY BORON 1 st IONISATION ENERGY / k. Jmol -1 1 s 1 s 1 s 2 s 2 p EXPLANATION There is a DROP in the value for Boron. This is because the extra electron has gone into one of the 2 p orbitals. The increased shielding makes the electron easier to remove It was evidence such as this that confirmed the existence of sub-shells. If there hadn’t been any subshell, the value would have been higher than that of Beryllium. ATOMIC NUMBER 5
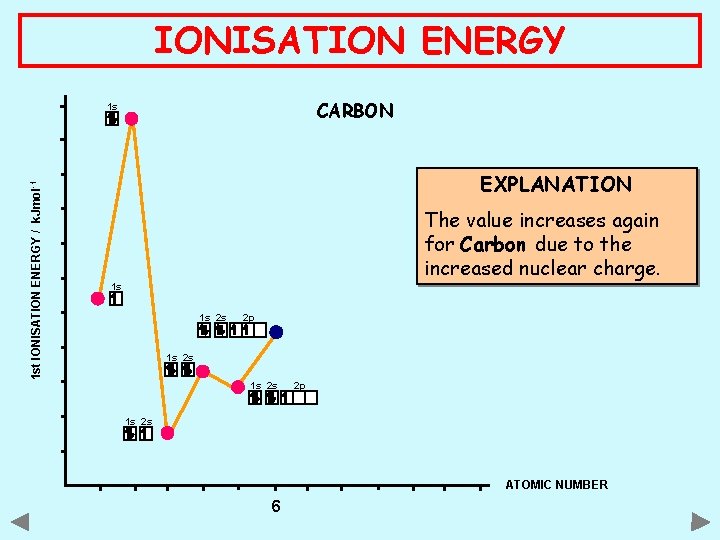
IONISATION ENERGY CARBON 1 st IONISATION ENERGY / k. Jmol -1 1 s EXPLANATION The value increases again for Carbon due to the increased nuclear charge. 1 s 1 s 2 s 2 p 1 s 2 s ATOMIC NUMBER 6
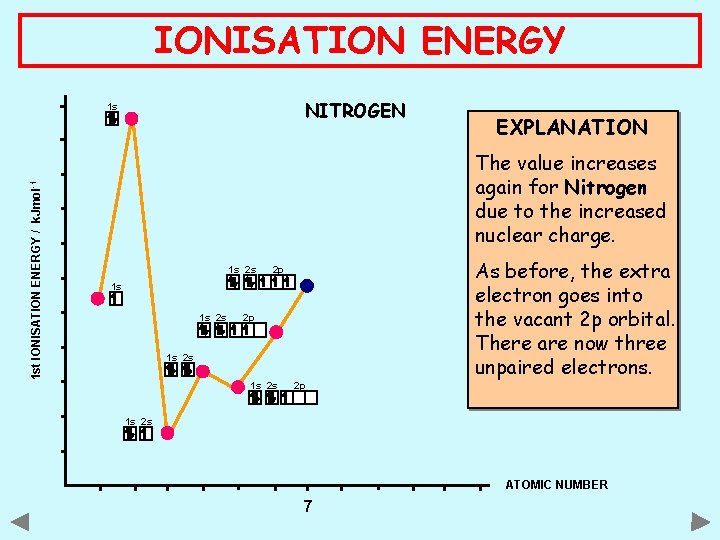
IONISATION ENERGY NITROGEN 1 st IONISATION ENERGY / k. Jmol -1 1 s EXPLANATION The value increases again for Nitrogen due to the increased nuclear charge. 1 s 2 s 2 p 1 s 2 s 2 p As before, the extra electron goes into the vacant 2 p orbital. There are now three unpaired electrons. 1 s 2 s ATOMIC NUMBER 7
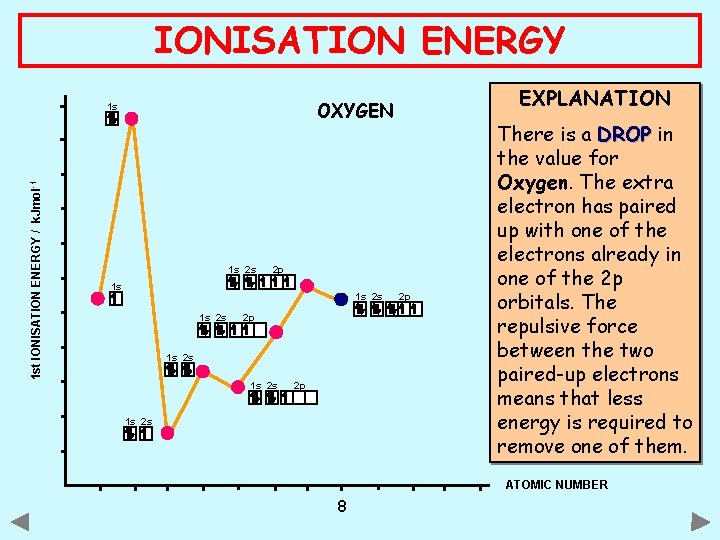
IONISATION ENERGY 1 st IONISATION ENERGY / k. Jmol -1 EXPLANATION OXYGEN 1 s 1 s 2 s 2 p 1 s 2 s 2 p There is a DROP in the value for Oxygen. The extra electron has paired up with one of the electrons already in one of the 2 p orbitals. The repulsive force between the two paired-up electrons means that less energy is required to remove one of them. ATOMIC NUMBER 8
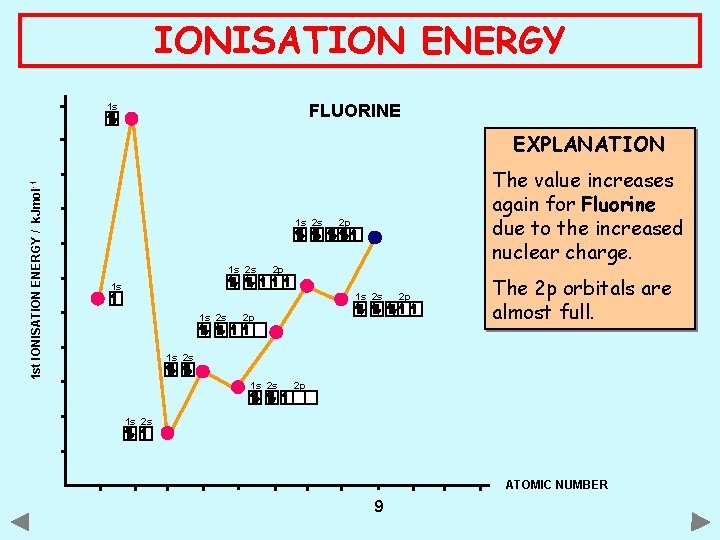
IONISATION ENERGY 1 s FLUORINE 1 st IONISATION ENERGY / k. Jmol -1 EXPLANATION 1 s 2 s The value increases again for Fluorine due to the increased nuclear charge. 2 p 2 p 1 s 1 s 2 s 2 p 2 p The 2 p orbitals are almost full. 1 s 2 s 2 p 1 s 2 s ATOMIC NUMBER 9
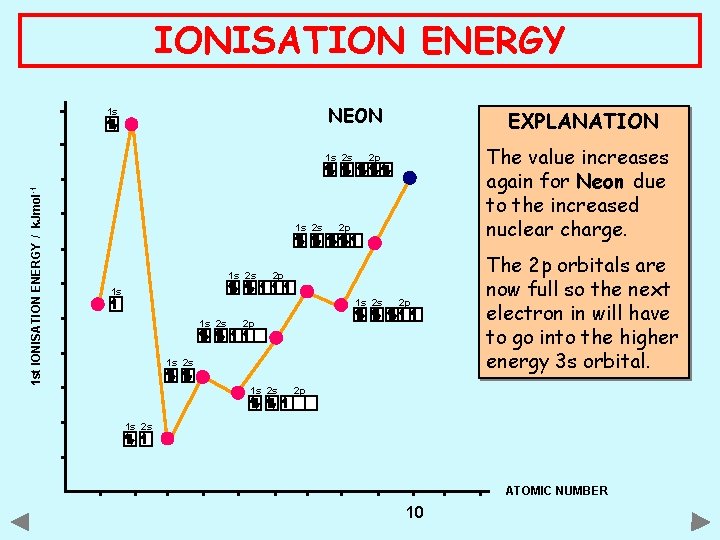
IONISATION ENERGY NEON 1 s 1 st IONISATION ENERGY / k. Jmol -1 1 s 2 s The value increases again for Neon due to the increased nuclear charge. 2 p 2 p 2 p 1 s 1 s 2 s EXPLANATION 2 p 2 p 1 s 2 s The 2 p orbitals are now full so the next electron in will have to go into the higher energy 3 s orbital. 2 p 1 s 2 s ATOMIC NUMBER 10
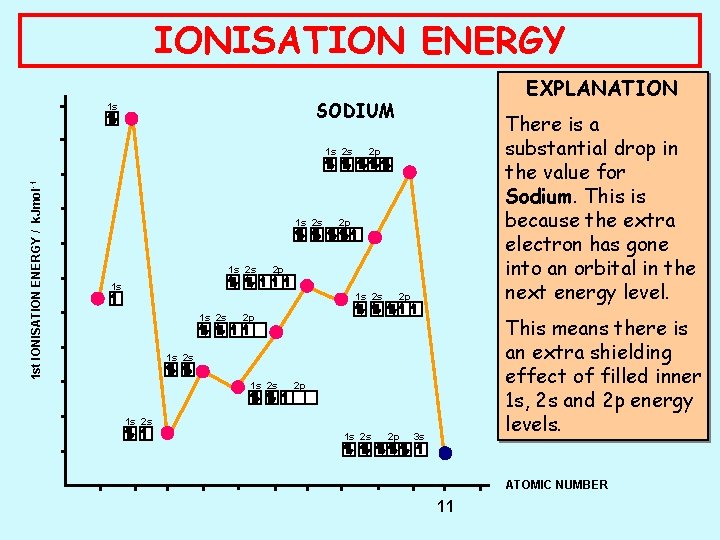
IONISATION ENERGY SODIUM 1 s 1 s 2 s 1 st IONISATION ENERGY / k. Jmol -1 EXPLANATION 1 s 2 s 2 p 2 p 2 p 1 s 1 s 2 s There is a substantial drop in the value for Sodium. This is because the extra electron has gone into an orbital in the next energy level. 2 p 2 p This means there is an extra shielding effect of filled inner 1 s, 2 s and 2 p energy levels. 1 s 2 s 2 p 3 s ATOMIC NUMBER 11
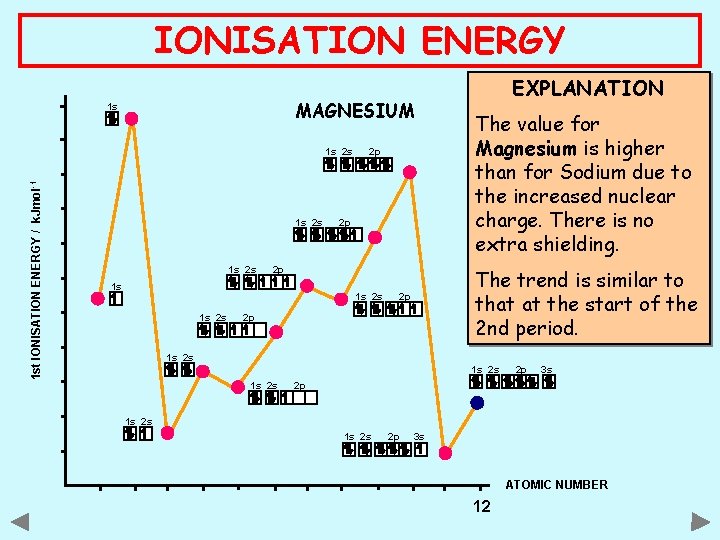
IONISATION ENERGY MAGNESIUM 1 s 1 st IONISATION ENERGY / k. Jmol -1 1 s 2 s 2 p 2 p 2 p 1 s 1 s 2 s EXPLANATION The value for Magnesium is higher than for Sodium due to the increased nuclear charge. There is no extra shielding. The trend is similar to that at the start of the 2 nd period. 2 p 2 p 1 s 2 s 2 p 3 s 2 p 1 s 2 s 2 p 3 s ATOMIC NUMBER 12
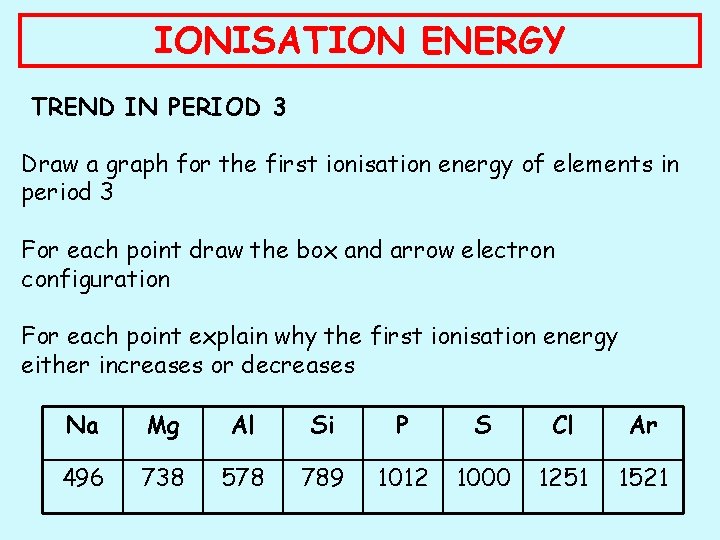
IONISATION ENERGY TREND IN PERIOD 3 Draw a graph for the first ionisation energy of elements in period 3 For each point draw the box and arrow electron configuration For each point explain why the first ionisation energy either increases or decreases Na Mg Al Si P S Cl Ar 496 738 578 789 1012 1000 1251 1521

IONISATION ENERGY TREND IN PERIOD 3 Drop from Mg Al: Mg: 1 s 2 2 p 6 3 s 2 Al: 1 s 2 2 p 6 3 s 2 3 p 1 Drop from P S: P: 1 s 2 2 p 6 3 s 2 3 p 3 S: 1 s 2 2 p 6 3 s 2 3 p 4 The outer electron in Al has moved into the 3 p orbital. It takes less energy to remove an electron from here than the 3 s In P each of the 3 p orbitals has 1 electron. In S one must have 2. The repulsion between these 2 paired electrons makes it easier to remove one
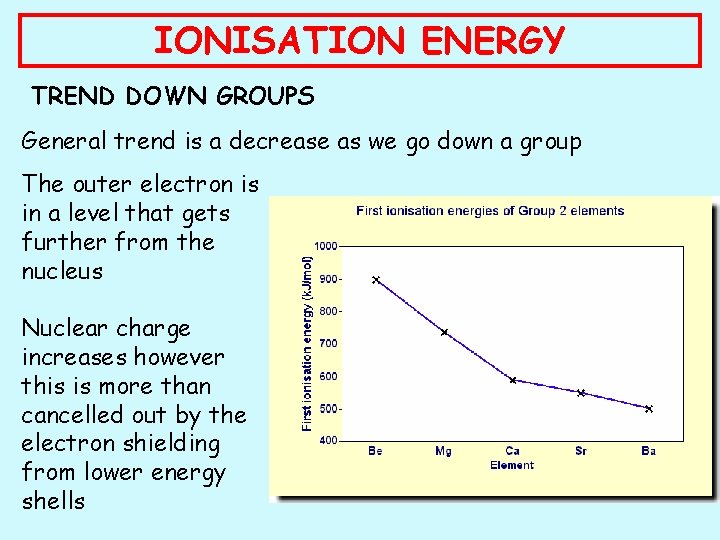
IONISATION ENERGY TREND DOWN GROUPS General trend is a decrease as we go down a group The outer electron is in a level that gets further from the nucleus Nuclear charge increases however this is more than cancelled out by the electron shielding from lower energy shells

IONISATION ENERGY EXTENSION QUES: Which has the higher value, the 1 st I. E. of sodium or the 2 nd I. E. of magnesium? A: The 2 nd I. E. of magnesium The 1 st I. E. of sodium involves the following change Na(g) 1 s 2 2 p 6 3 s 1 Na+(g) 1 s 2 2 p 6 The 2 nd I. E. of magnesium involves the same change in electron configuration… Mg+(g) 1 s 2 2 p 6 3 s 1 Mg 2+(g) 1 s 2 2 p 6 However, magnesium has 12 protons in its nucleus, whereas sodium only has 11. The greater nuclear charge means that the electron being removed is held more strongly and more energy must be put in to remove it.

IONISATION ENERGY HOMEWORK: Revise for an end of chapter test next lesson

IONISATION ENERGY I CAN… I AM… State what is meant by ionisation energy C Describe the trend across period 3 and down group 2 B Explain why this trend occurs A Suggest how these trends provide evidence for the existence of electron levels and sub-levels A* How low can you go? ? Write what you can do and what grade this is show some proof you can do this!
- Slides: 30