Ionic Compounds Writing Formulas Empirical Formulas smallest wholenumber

Ionic Compounds: Writing Formulas

Empirical Formulas • = smallest whole-number ratio of elements • Ionic compounds: empirical formulas only

Identify the empirical formulas • H 2 O Empirical • H 2 O 2 Molecular • Ca. F 2 Empirical (Ionic) • Na. Cl Empirical (Ionic) • CH 4 Empirical • C 2 H 6 • Fe. Cl 3 Molecular Empirical (Ionic)

Binary Ionic Compounds • Composed of two different elements – Positive monatomic metal ion – Negative monatomic nonmetal ion Note: binary compound may contain more than 2 ions but only 2 kinds of ions like Al 2 O 3

Oxidation Number • Charge of a monatomic ion O-2 • Some elements form only 1 ion Na+1 Ca+2 • Some elements can form more than 1 ion Fe+2 and Fe+3

Oxidation State of Metal Elements • • Group 1 ions always +1 Group 2 ions always +2 MOST group 13 ions are +3 Groups 14, 15, 16, and 17: multiple oxidation states • Transition metals (Group B) can have more than 1 oxidation state

Writing Formulas: Binary Ionic Compounds • first rule is: POSITIVE ION FIRST (metal) How do you get subscripts? start by writing the ions

Compounds are neutral: • Sum of all charges must = 0 • (+) and (-) charges must = each other

Charges: Equal but Opposite • Na+1 and Cl-1: Na. Cl • Mg+2 and O-2: Mg. O • Al+3 and P-3: Al. P +1 + (-1) = 0 +2 + (-2) = 0 +3 + (-3) = 0 • Rule: write the symbols, (+) FIRST! FIRST

Try a few formulas: • • • Li+1 and I-1 Ca+2 and O-2 Al+3 and N-3 K+1 and F-1 Ba+2 and S-2 Li. I Ca. O Al. N KF Ba. S
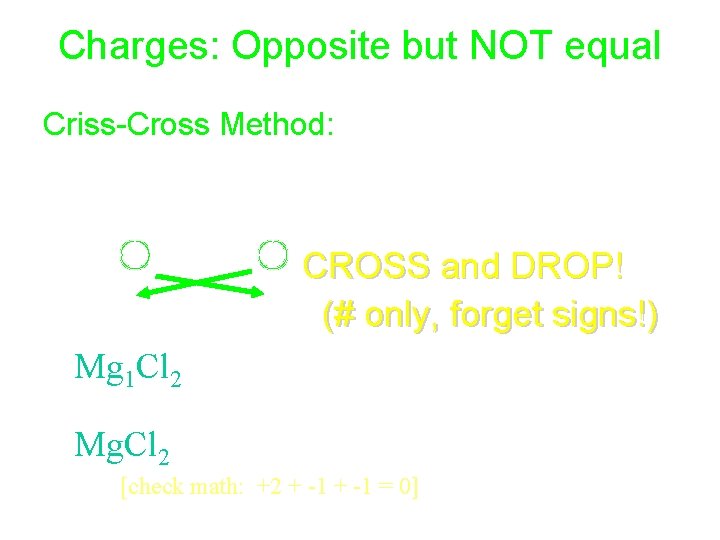
Charges: Opposite but NOT equal Criss-Cross Method: • Mg+2 and Cl-1, CROSS and DROP! (# only, forget signs!) • Mg 1 Cl 2 (if subscript is 1, forget it) • Mg. Cl 2 (1 Mg+2 and 2 Cl-1) [check math: +2 + -1 = 0]
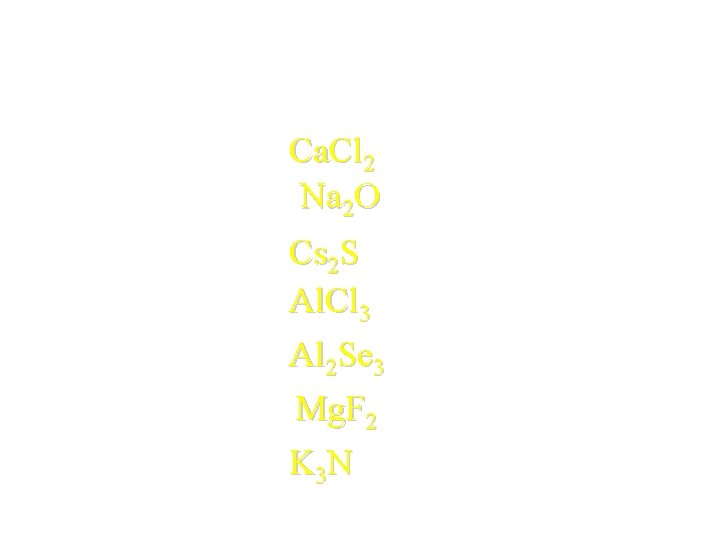
Try a few formulas: • • Ca+2 + Cl-1 Na+1 + O-2 Cs+1 + S-2 Al+3 + Cl-1 Al+3 + Se-2 Mg+2 + F-1 K+1 + N-3 Ca. Cl 2 Na 2 O Cs 2 S Al. Cl 3 Al 2 Se 3 Mg. F 2 K 3 N
- Slides: 12