IONIC COMPOUNDS WHAT IS AN ION An ion























- Slides: 25

IONIC COMPOUNDS

WHAT IS AN ION? An ion is an atom that has gained or lost an electron Cation – lost electron – name stays the same – sodium ion Anion – gained an electron – adds –ide to the end - chloride

HOW MANY ELECTRONS DOES AN ATOM START WITH? Atomic number: Number of protons Number of electrons

HOW DO YOU KNOW IF AN ATOM WILL LOSE OR GAIN? Metals lose electrons until they reach the electron configuration of a noble gas Nonmetals gain electrons until they reach electron configuration of a noble gas


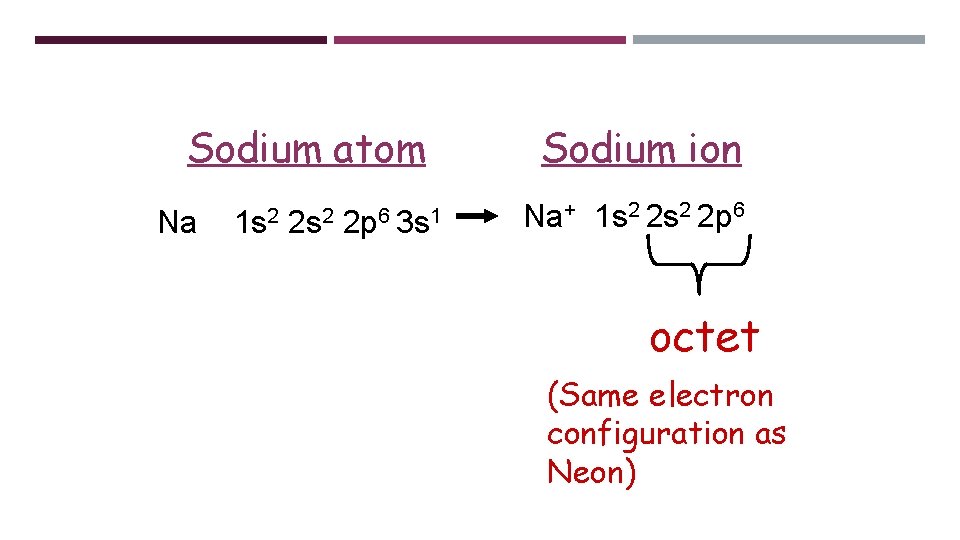
Sodium atom Na 1 s 2 2 p 6 3 s 1 Sodium ion Na+ 1 s 2 2 p 6 octet (Same electron configuration as Neon)

OXIDATION NUMBERS Oxidation numbers are used to keep track of how many electrons are lost or gained Written as a superscript after the element symbol + indicates electrons lost (less electrons) - indicates electrons gained (more electrons) Ex: Na+1 Ex: Cl-1


PROPERTIES OF IONIC COMPOUNDS Brittle solids High melting points Electronically neutral compounds Soluble in water When dissolved in water, they are known as electrolytes Ionic compounds are known as salts

IONIC BONDS • Formed between a Cation (+)(“giver”) or (metal) and an anion(-) (“taker”) or (nonmetal) • Electrons transfer from one atom to another

2 TYPES OF IONIC COMPOUNDS Binary – has just two elements with total charge equaling zero Ex. Na. Cl Ternary – involve three (or more) elements, usually forming a polyatomic ion group

FORMULA FOR BINARY IONIC COMPOUND Ex: magnesium chloride Magnesium ion has a +2 charge Chloride ion has a -1 charge So we need 2 Cl for every 1 Mg Mg. Cl 2 Ratio of Mg to Cl is 1: 2

CRISS CROSS METHOD Write symbol for + ion (metal) and its charge. Write symbol for – ion (nonmetal) and its charge Reduce to lowest terms Criss-cross (If subscript is a 1, no need to put it in formula)

CRISS CROSS METHOD EXAMPLE Sodium oxide Sodium and oxygen Na 1+ Na 2 O O = Na 2 O

FORMULA UNIT A formula unit smallest part of an ionic compound Ex. Na. Cl is 1 formula unit of table salt. 3 Na. Cl is 3 formula units of table salt

TRANSITION METALS Stock name and classical name Stock – preferred naming system, uses Roman numerals in parentheses to placed after the name of the element to indicated the numerical value of the charge ex: Fe 2+ would be iron(II) Classical- older method uses root word with different suffix and –ous -ic is used for the higher of the two ionic charges -ous is used for the smaller of the two charges -ic

LIST OF COMMON METAL IONS WITH MORE THAN ONE IONIC CHARGE Copper (I) Copper (II) Iron (III) Mercury (I) - Hg 22+ diatomic elemental ion Mercury (II) – Hg 2+ Cobalt (II) Cobalt (III) Lead (IV) Tin (II) Tin (IV) Chromium (II) Chromium (IV) Manganese (III)

SUMMARY How do you know what the charge is? Practice grasshoppers, just practice Cu 1+ and 2+ Fe, Mn, Cr, Co all 2+ and 3+ Pb and Sn are 2+ and 4+ Hg 2 2+ mercury(I) Hg 2+ mercury(II)

OTHER EXCEPTIONS Group 4 A: usually form ions with 4+ charge. Sn and Pb also produce ions with 2+ charge- may only lose 2 electrons from psublevel Ag is always 1+ Cd is always 2+ Zn is always 2+

TERNARY IONIC COMPOUNDS Involve polyatomic ions: Two or more atoms that are bound together by predominantly covalent bonds, but have gained or lost an electron in order to become more stable Act as a whole when forming an ionic compound

POLYATOMIC IONS Chromate Cr. O 4 2 - Sulfate SO 42 Phosphate PO 4 3 - Carbonate CO 32 Nitrate NO 31 Bromate Br. O 31 Chlorate Cl. O 31 - Acetate C 2 H 3 O 21 Permangenate Mn. O 41 Hydroxide OH 1 Ammonium NH 41+

WRITING TERNARY IONIC COMPOUND FORMULAS Potassium sulfate K+1 SO 42 - K 2 SO 4

Ex. Magnesium and hydroxide Mg and (OH) Mg 2+ and (OH)1 Use criss-cross method Mg(OH)2 *If there is more than one polyatomic ion group, you must put it in parentheses.

Calcium sulfate Potassium phosphate Ammonium sulfite Ammonium hydroxide

NAMING IONIC COMPOUNDS WITH POLYATOMIC ION GROUPS Same rules as binary ionic compounds, but if it has a Polyatomic Ion Group, use group name