Ionic Compounds What do these have in common








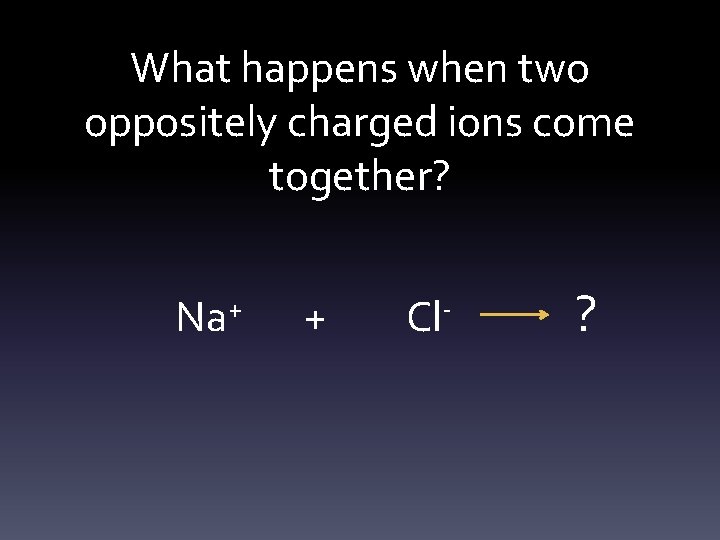













- Slides: 23

Ionic Compounds

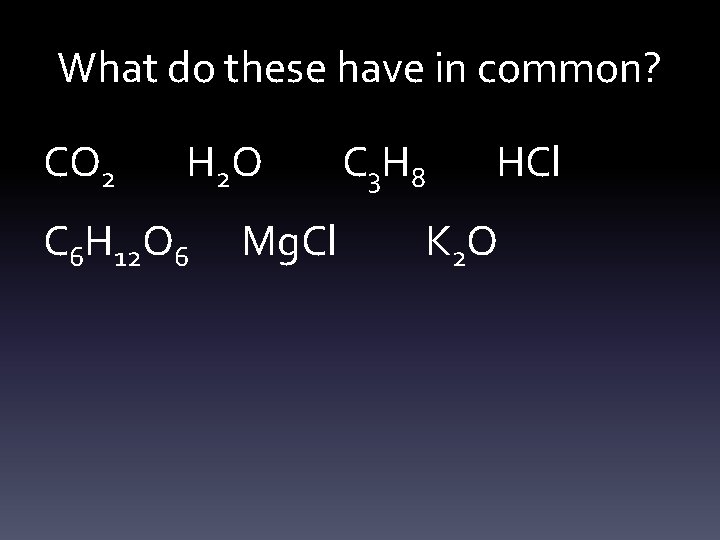
What do these have in common? CO 2 H 2 O C 6 H 12 O 6 Mg. Cl C 3 H 8 HCl K 2 O

• They are all compounds – 2 or more elements chemically bound together

Ionic Compounds • Ionic compounds are compounds that consist of a positive ion and a negative ion that are attracted to one another because of their opposite electrical charges.

What are ions? • An atom or group of atoms that has gained or lost electrons and has a negative or positive charge as a result.

Positively charged ions (cations) • Ca 2+ Na+ K+ Mg 2+ Al 3+ Sr 2+ The first one is a calcium ion. What are the other ones called?

Ionic Compound Note Sheet On your handout, write the names of the cations (positively charged atoms) in the space provided. If you need help, the sub has a key. But, seriously, this is easy. USE YOUR PERIODIC TABLE!

Negatively charged ions (anions) Cl- F- O 2 - P 3 - S 2 - N 3 - The first one is a chloride ion. What are the other ones called?

Ionic Compound Note Sheet On your handout, write the names of the anions (negatively charged atoms) in the space provided. If you need help, the sub has a key. But, seriously, this is easy. Just use -ide USE YOUR PERIODIC TABLE!
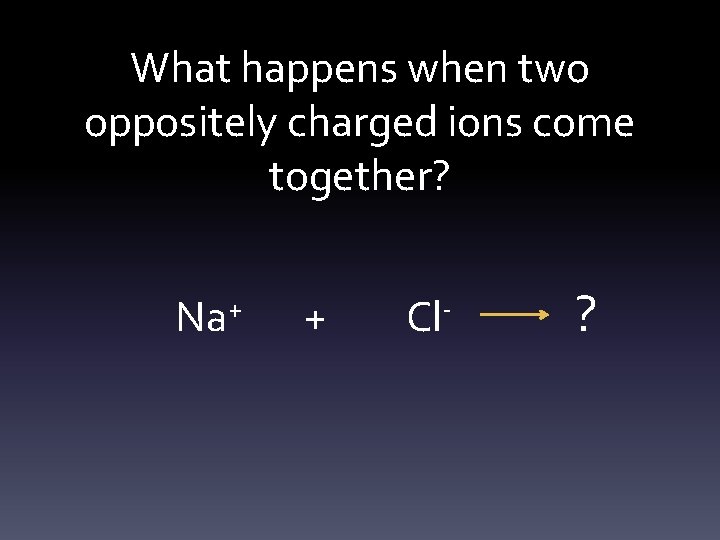
What happens when two oppositely charged ions come together? Na+ + Cl- ?

They form an ionic compound that has a neutral charge! Na. Cl

Ionic Compound Note Sheet • Take all 6 positively charged atoms (cations) from the first table and combine them with the 6 negatively charged atoms (anions) from the second table and write their names in the third table of your handout.

Names are easy – positive ions first and negative ions last Sodium chloride You will have 36 names to write on the third table of your handout.

Formulas? Remember they have to be neutral after they combine 2+ Ca + Cl ?

This is a little trickier. The positive atoms must cancel out the negative atoms. You can have as many positive and negative atoms as you like to do this.

2+ Ca combines 2 Cl with to give: Ca. Cl 2 The 2 chlorides add up to -2, cancelling out the +2 charge of the calcium. It’s neutral!

You need 2 chlorides (Cl-1) for every calcium (Ca+2). The resulting formula is neutral (no charge). Ca. Cl 2

Ionic Compound Note Sheet On you handout, write down the formulas for all 36 of the ionic compounds you named in the previous table. Remember, they will all have a neutral charge. The positive charges will cancel out the negative charges.

Polyatomic Ions Polyatomic ions are groups of atoms that have either a positive or negative charge Positive: NH 4+ ammonium Negative: SO 42 NO 32 SCNCO 32 OHPO 43 - sulfate nitrate thiocyanate carbonate hydroxide phosphate

Ionic Compound Note Sheet On the back of the handout, write the formulas that would result in the positive ions and negative ions forming a compound. Remember, the positive charges must cancel out the negative charges.

This is where parentheses are used. If you have more than one of the polyatomic ions in a formula, use the parentheses. The number on the outside says how many of the polyatomic ions you have.

NH 4+1 is called ammonium NO 32 - is called nitrate When they combine, they form ammonium nitrate. Names are easy. You need 2 NH 4+1 to cancel out the NO 32 -

You need 2 NH 4+1 to cancel out the NO 322 +1 charges cancel out the 1 -2 charge and it’s neutral (NH 4)2 NO 3 See? Parentheses are used!