Ionic Compounds Previous Information to Remember n Charges

Ionic Compounds

Previous Information to Remember!!! n Charges of ions of representative elements n Which types of elements form positive ions vs negative ions n Polyatomic ions (on formula chart) – Their Lewis structures n Element symbols n Cations and anions
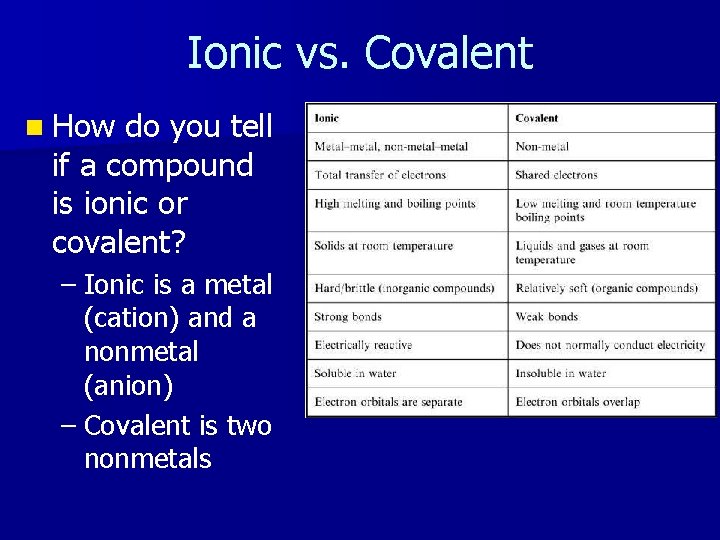
Ionic vs. Covalent n How do you tell if a compound is ionic or covalent? – Ionic is a metal (cation) and a nonmetal (anion) – Covalent is two nonmetals

Monatomic vs Polyatomic n Monatomic – 1 atom – Na+1, Al 3+, S 2 -, Cl 1– Anions end in “-ide” n Polyatomic – 2 or more atoms bonded together – NO 31 -, SO 42 -, OH 1 -, NH 41+ – Anions typically end in “-ate” or “-ite”

Ionic bond – Transfer of Electrons n What is the charge of the sodium ion? n What is the charge on the chloride ion? n What is the overall charge of the sodium chloride compound?

Ionic bond – Transfer of Electrons n What is the charge of the magnesium ion? n What is the charge on the oxide ion? n What is the overall charge of the magnesium oxide compound?

Ionic bond – Transfer of Electrons n What is the charge of the magnesium ion? n What is the charge on the fluoride ion? n What is the overall charge of the magnesium fluoride compound? Why was there 1 cation and 1 anion in magnesium oxide but 1 cation and 2 anions in magnesium fluoride?

Binary Ionic Compounds n Two elements – one metal, one nonmetal n Metal gets written first n Nonmetal gets written second, but change ending to –ide n e t it r n Examples: w nd – Be. Cl 2 – Beryllium chloride – Al 2 O 3 – Aluminum oxide it a re s i l 2 e C y e h B f th xes? W as i fi e 3 O pr l 2 A no e ar

Total charges of ions must equal zero!!! n Mg n How F many fluorines will you need? n Al O n What if the ions have the same numerical charge? n Ca S

Drop and Swap!!! n Write the ions n Drop the charges down (as subscripts) and swap charges with other atom Write as empirical formula!!!

Do Page 5 Odds!!!

Number Answer 1 Na. Br 3 Mg. S 5 Li 3 P 7 KI 9 Rb 3 N 11 Potassium oxide 13 Aluminum chloride 15 Sodium nitride 17 Barium phosphide 19 Strontium fluoride Check your Answers for the Odds!!!

Lewis Structures n Draw the Lewis dot structures of each element in compound n Transfer electrons from metal to nonmetal n Add enough of each element (as needed) to transfer the correct number of electrons n Brackets around the anion; put charges behind everything in compound

Polyatomic Ions § Formed when more than one atom are grouped together § Charge on polyatomic ions applies to entire group of atoms. § The polyatomic ion acts as an individual ion and is protected with parenthesize in the formula! § Typically end in –ate or -ite Sulfate Carbonate Common Polyatomic Ions NH 4+ Ammonium NO 3 - Nitrate OH- Hydroxide CO 32 - Carbonate SO 42 - Sulfate PO 43 - Phosphate NO 2 - Nitrite
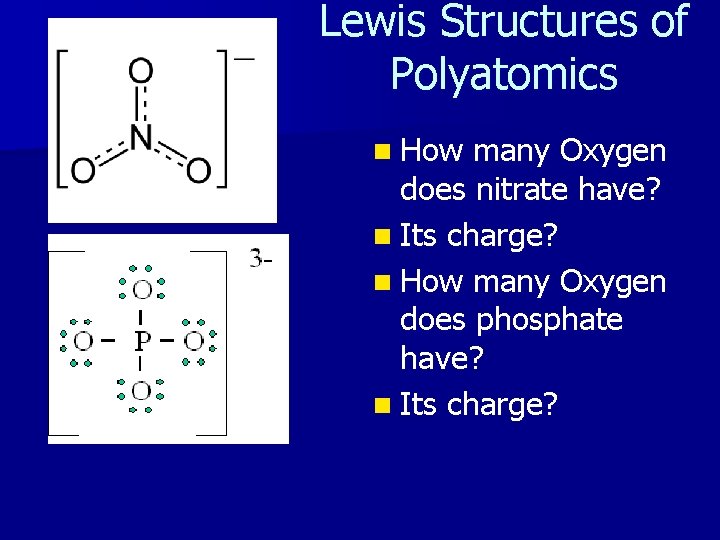
Lewis Structures of Polyatomics n How many Oxygen does nitrate have? n Its charge? n How many Oxygen does phosphate have? n Its charge?

Dropping and Swapping with Polyatomics n You have to keep the n. K+1 PO 43 same # of O!!! n. K 3 PO 4 n Drop and swap the charge!!! 2+ n. Ba NO 3 n Ba(NO ) 3 2 n What’s the difference between Mg. OH 2 and Mg(OH)2?

Practice Mg(Cl. O 3)2 chlorate: _______ NH 4 Cl n Ammonium chloride: _______ Magnesium carbonate n Mg. CO 3: ____________ Potassium sulfite n K 2 SO 3: ____________ n Aluminum dichromate: Al _______ 2(Cr 2 O 7)3 Ca(OH)2 n Calcium hydroxide: ________ Sodium nitrate n Na. NO 3: _____________ n Magnesium

Do Page 6 odds!!!

Number Answer 1 Na 2 Cr. O 4 3 Mg(NO 3)2 5 Li 3 PO 4 7 Cs. Cl. O 3 9 Ba(C 2 H 3 O 2)2 11 Potassium acetate 13 Aluminum chlorate 15 Strontium bicarbonate 17 Lithium carbonate 19 Cesium chromate Check your Answers!!!

Transition Metals and Roman Numerals n n n Transition metals (and Sn, Pb, Bi) do not have set charges Can form variable ions (ex. Fe 2+ or Fe 3+) Roman Numerals are used indicate charge of transition metals – Copper (II) = Copper +2 – Iron (III) = Iron +3 n Some transition metals do have set charges – Zn 2+, Cd 2+, Ag 1+

Writing Names and Formulas n Use Roman numeral in name to write formula – Example: Cobalt (III) chloride – Co. Cl 3 – Example: Tin (IV) sulfate– Sn(SO 4)2 n When going name to formula, include Roman numeral in name (except for Zn, Cd, Ag) – Example: Ni(NO 3)2 – Nickel (II) nitrate – Example: Au 2 O – Gold (I) oxide

Practice n Zn. Cl 2: ____________ Zinc chloride Fe. PO 4 n Iron (III) phosphate: ________ Cr. Br 2 n Chromium (II) bromide: _______ Lead (IV) oxide n Pb. O 2: ______________ Ag 2 SO 4 n Silver sulfate: __________ Copper (I) nitrate n Cu. NO 3: _____________ Nickel (III) sulfite n Ni 2(SO 3)3: ____________

Do Page 7 odds!!! Pay careful attention on when to use Roman numerals and when not to also when you have a polyatomic ion involved vs. a monatomic ion
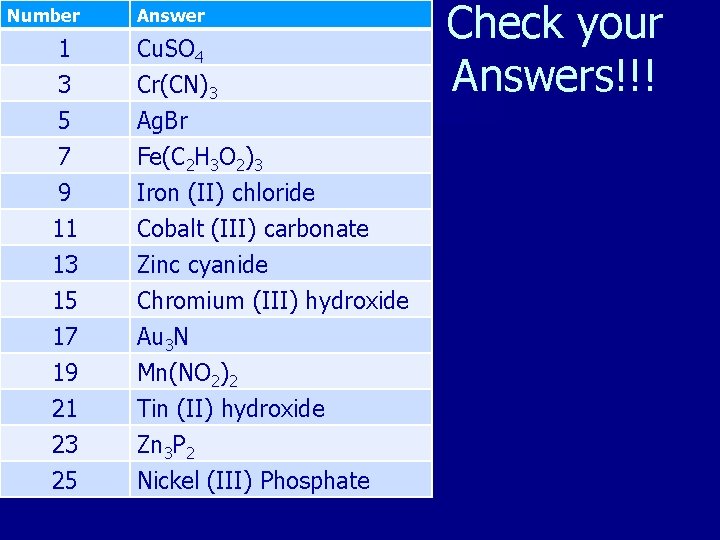
Number Answer 1 Cu. SO 4 3 Cr(CN)3 5 Ag. Br 7 Fe(C 2 H 3 O 2)3 9 Iron (II) chloride 11 Cobalt (III) carbonate 13 Zinc cyanide 15 Chromium (III) hydroxide 17 Au 3 N 19 Mn(NO 2)2 21 Tin (II) hydroxide 23 Zn 3 P 2 25 Nickel (III) Phosphate Check your Answers!!!
- Slides: 24