Ionic Bonds Two types of bonds n Ionic

Ionic Bonds

Two types of bonds n Ionic: between a metal and a nonmetal, electrons are transferred n Covalent: between two nonmetals, electrons are shared n All bonds help fulfill the octet rule=all elements want 8 valence electrons n Except H, He, Li, and Be that want only 2

Valence Electrons n Valence electrons are the electrons in the highest occupied energy number level of the atom. n Valence electrons are the only electrons generally involved in bond formation. n Equal the group #
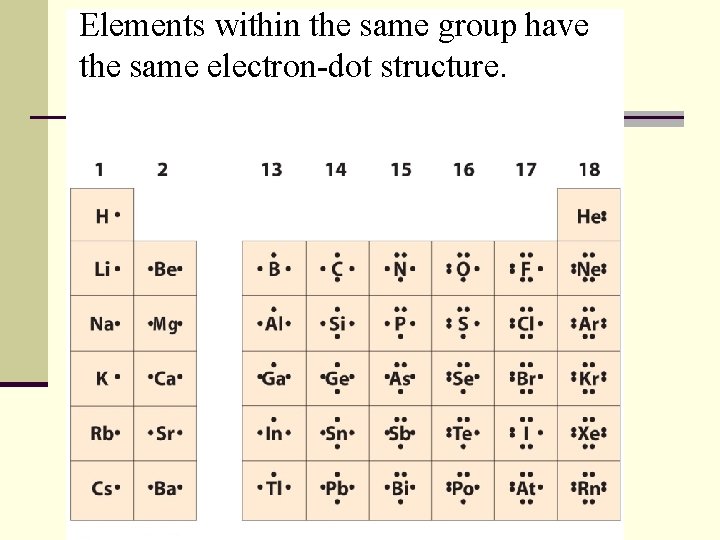
Elements within the same group have the same electron-dot structure.

A Positive Ion (Cation): n An atom that has lost one or more electrons. n + charge n Metals form cations
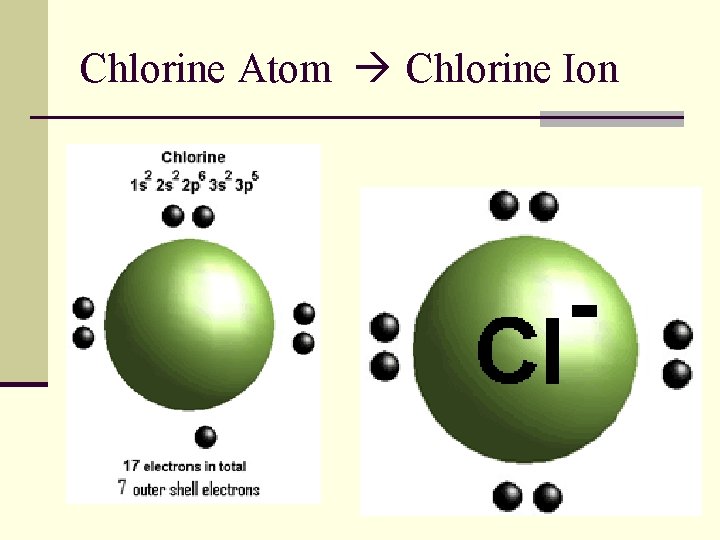
Chlorine Atom Chlorine Ion

A Negative Ion (Anion) n An atom that has gained one or more electrons. n Negatively (-) charged. n Nonmetals form anions

Check your Neighbor When Na, (11) loses its valence electron, what element does its configuration look like a. b. c. d. Neon Potassium Beryllium Sodium

Check your Neighbor When Na, (11) loses its valence electron, what element does its configuration look like ? a. b. c. d. Neon Potassium Beryllium Sodium

Ionic Bonds n Bonds that are formed by transfer of electrons from one element to the other. n Each element (now an ion) will have a complete octet after the transfer of electrons.
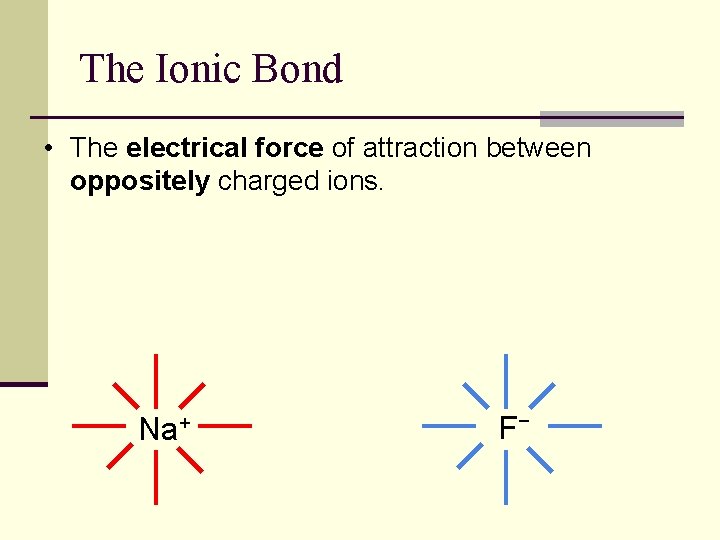
The Ionic Bond • The electrical force of attraction between oppositely charged ions. Na+ F−

Example: Formation of Lithium Fluoride . . Li + : F: . . Li+ : F- . . :

Is an Ionic Compound Electrically Neutral or Charged?

Naming ionic compounds n Put the metal name first n Put the nonmetal name second but change the ending to –ide n Na. Cl n K 2 O

Writing the formula for ionic compounds n Write down the symbols and charges n Criss cross the charges ignoring the signs n Don’t write as subscripts 1 n If the two charges are the same value there are no subscripts.

Links n http: //www. learner. org/interactives/periodic/gr oups_interactive. html n http: //www. wisconline. com/objects/View. Object. aspx? ID=GCH 2204 n http: //www. bbc. co. uk/schools/gcsebitesize/sci ence/add_aqa_pre_2011/atomic/ionicact. sht ml
- Slides: 16