IONIC BONDS Gaining electrons negative charge Losing electrons

IONIC BONDS

• Gaining electrons = negative charge • Losing electrons = positive charge • An atom which gains or loses electrons is called an ion
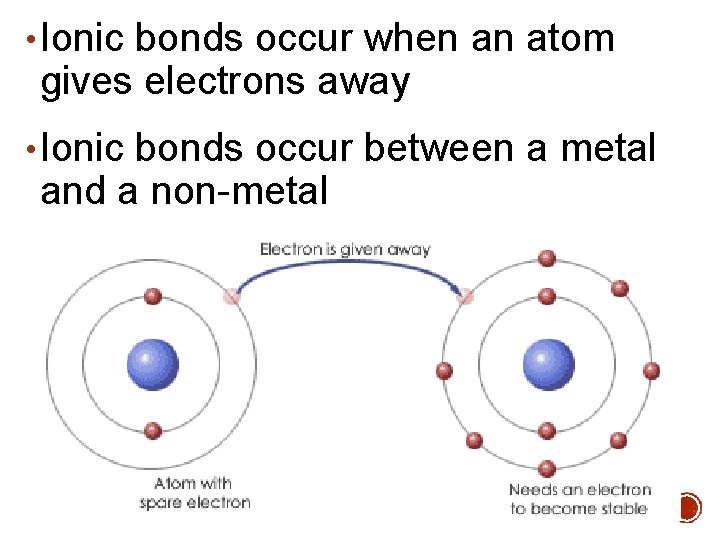
• Ionic bonds occur when an atom gives electrons away • Ionic bonds occur between a metal and a non-metal

• Anion = ion with a negative charge b • Cation = ion with a positive charge b • Ionic bond = the attraction between anions and cations b • Very easy to predict because they always work to neutralize the charges

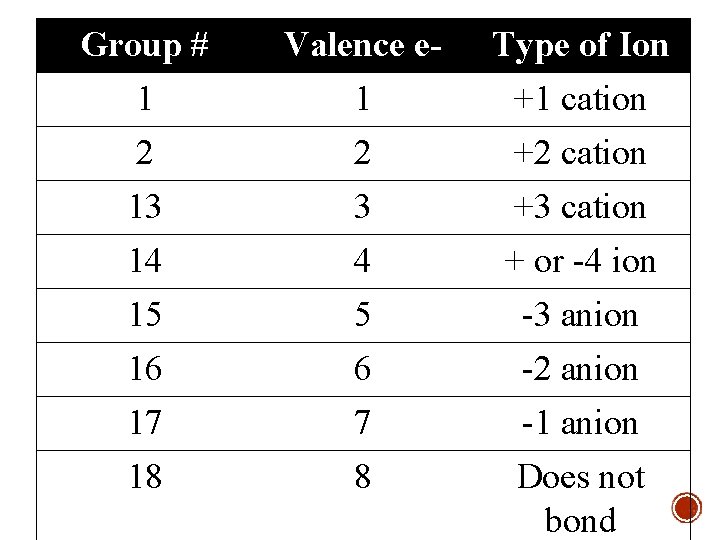
Group # Valence e- Type of Ion 1 2 13 14 15 16 17 18 1 2 3 4 5 6 7 8 +1 cation +2 cation +3 cation + or -4 ion -3 anion -2 anion -1 anion Does not bond
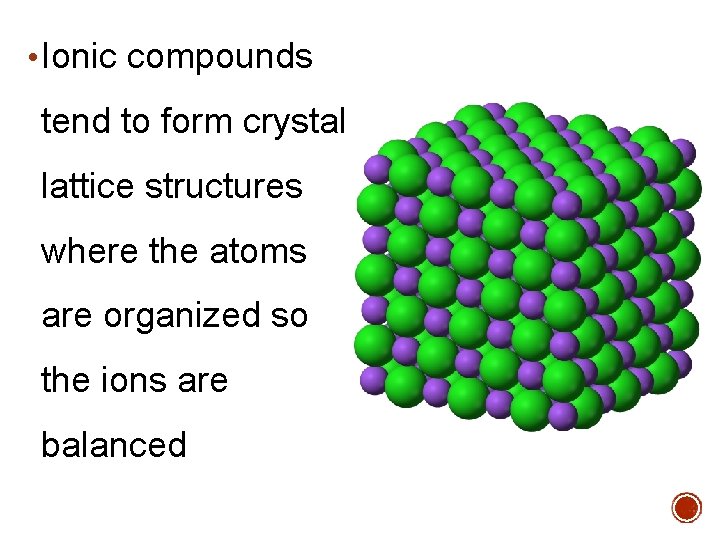
• Ionic compounds tend to form crystal lattice structures where the atoms are organized so the ions are balanced
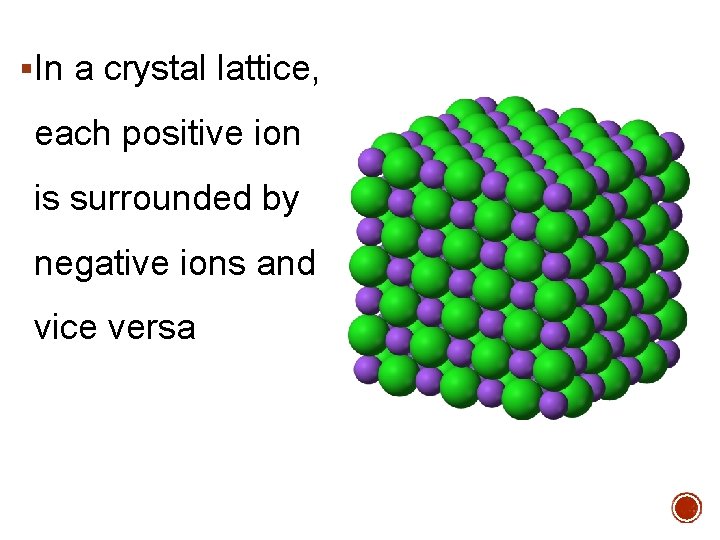
§In a crystal lattice, each positive ion is surrounded by negative ions and vice versa


• Ionic bonds are strong and require a large amount of energy to break • Therefore, ionic compounds have high melting and boiling points. • Solid ionic compounds are nonconductors because of the balance of the ions’ charges

• Liquid ionic compounds are conductors because their ions are free to move. • An electrolyte is an ionic compound in aqueous solution which conducts electricity.
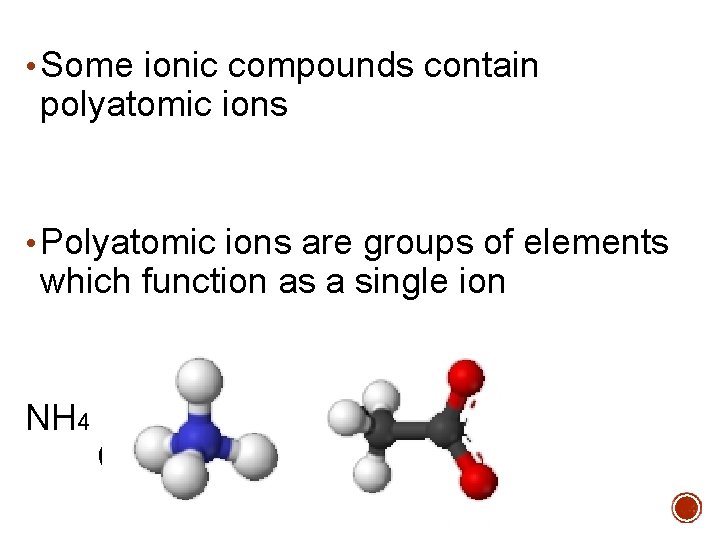
• Some ionic compounds contain polyatomic ions • Polyatomic ions are groups of elements which function as a single ion NH 4 C 2 H 3 O 2


METALLIC BONDS • Metallic bonds occur between metal atoms • In metallic bonds, atoms do not give away or share electrons
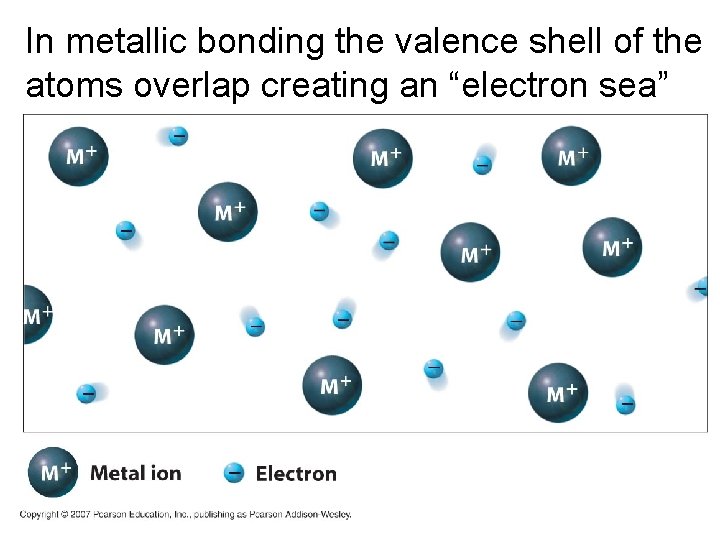
In metallic bonding the valence shell of the atoms overlap creating an “electron sea”
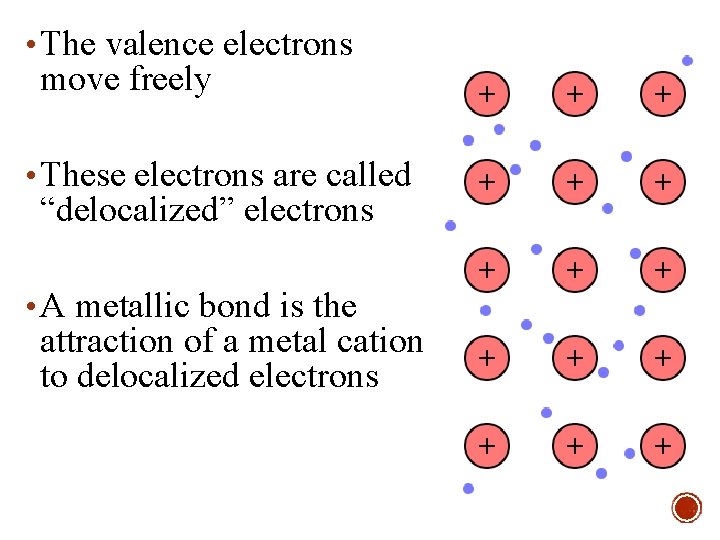
• The valence electrons move freely • These electrons are called “delocalized” electrons • A metallic bond is the attraction of a metal cation to delocalized electrons

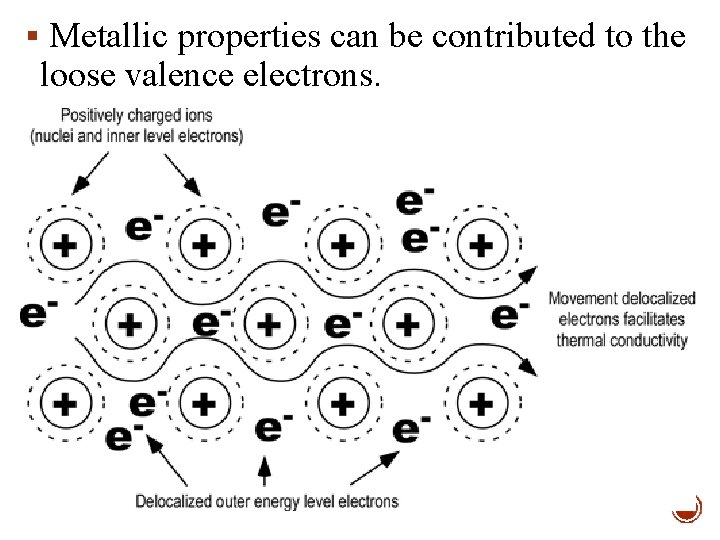
§ Metallic properties can be contributed to the loose valence electrons.
- Slides: 18