Ionic Bonds Formulas Na Cl Ionic Compounds composed
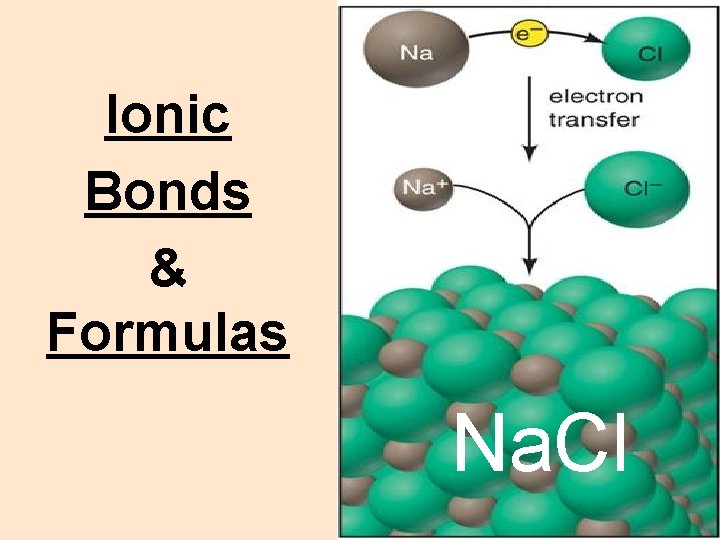
Ionic Bonds & Formulas Na. Cl
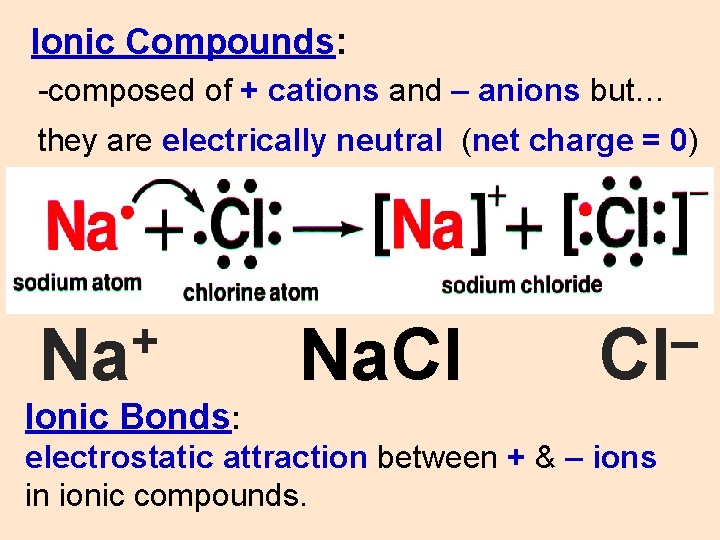
Ionic Compounds: -composed of + cations and – anions but… they are electrically neutral (net charge = 0) + Na Na. Cl – Cl Ionic Bonds: electrostatic attraction between + & – ions in ionic compounds.

Binary Compounds: contain only two different atoms Chemical Formula: shows type & number of atoms in substance. + –+ – –+– +–+ + – Formula Unit: the lowest whole-number ratio of atoms in an ionic compound. 1+ 1– 2+ 2– 4+ 2– Na. Cl Ca. S Pb. O 2 Na 2 Cl 2 Ca 2 S 2 Pb 2 O 4

Writing Formulas of Ionic Compounds: 1. Write symbol of cation (+), then anion (–). 2. Write charge of each ion as a superscript. 3. Write subscripts to balance the charges (crisscross charges) 4. Erase charges (neutral compound). potassium oxide K+ + 2– K+ O 2– +2 – 2 K O K 2 O magnesium nitride 2+ N 3– Mg 2+ 3– Mg N Mg 2+ +6 – 6 3 2 Mg N
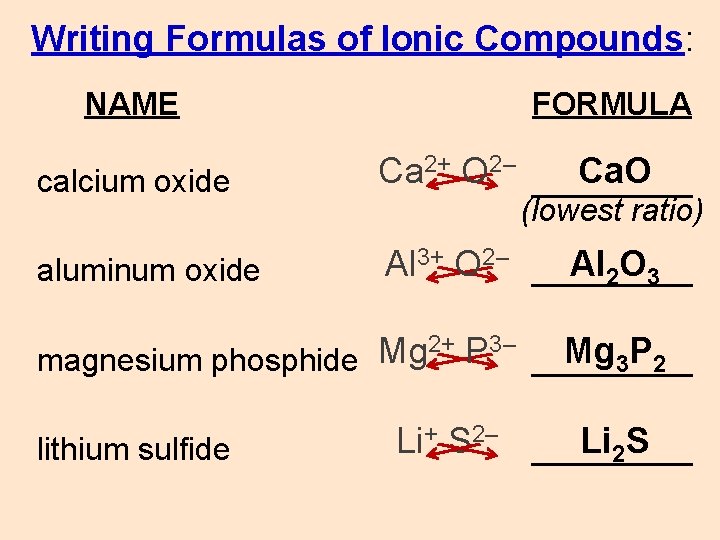
Writing Formulas of Ionic Compounds: NAME FORMULA calcium oxide Ca 2+ O 2– _____ Ca. O aluminum oxide Al 3+ O 2– _____ Al 2 O 3 (lowest ratio) 2+ P 3– Mg Mg 3 P 2 magnesium phosphide _____ lithium sulfide Li+ S 2– _____ Li 2 S
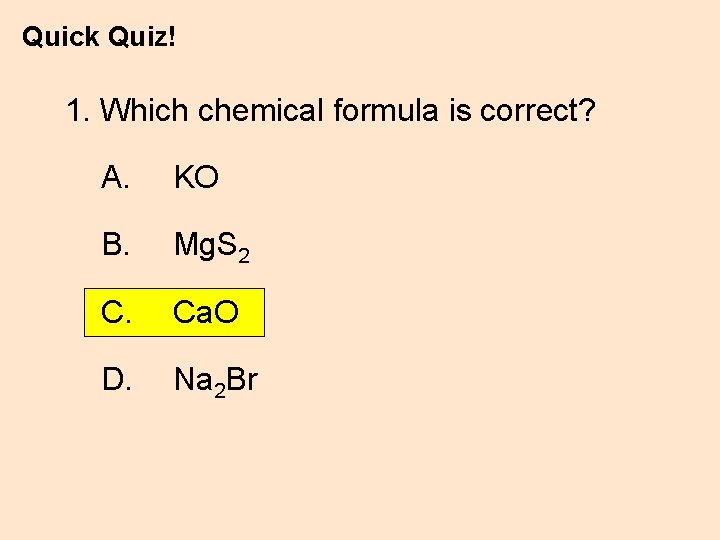
Quick Quiz! 1. Which chemical formula is correct? A. KO B. Mg. S 2 C. Ca. O D. Na 2 Br

Quick Quiz. 2. Which chemical formula is incorrect? A. KF 2 B. Ca. S C. Mg. O D. Na. Br
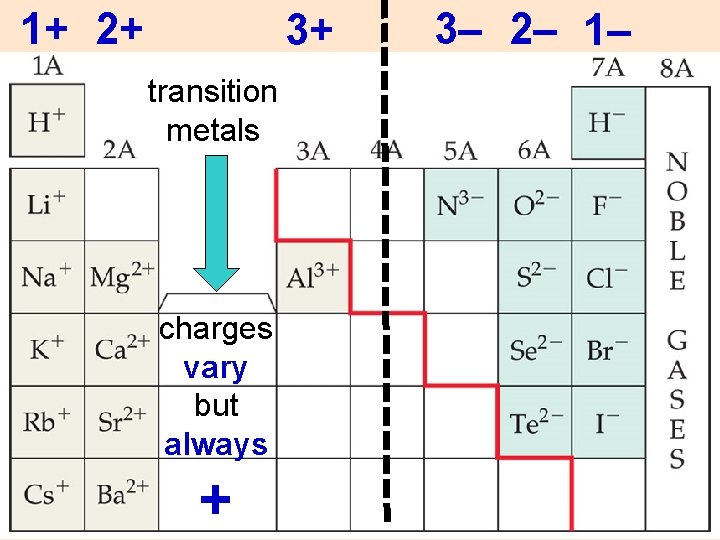
1+ 2+ 3+ transition metals charges vary but always + 3– 2– 1–
![Lewis dot diagrams + [Na] Al. Cl 3 Cl – Lewis dot diagrams + [Na] Al. Cl 3 Cl –](http://slidetodoc.com/presentation_image_h2/10463958254d42fbd53f838c8e72d71c/image-9.jpg)
Lewis dot diagrams + [Na] Al. Cl 3 Cl –
- Slides: 9