Ionic Bonds Chemistry Mrs Coyle Part A n

Ionic Bonds Chemistry Mrs. Coyle

Part A: n Ions n Lewis Dot Structure n Stable Octet (or Duet)
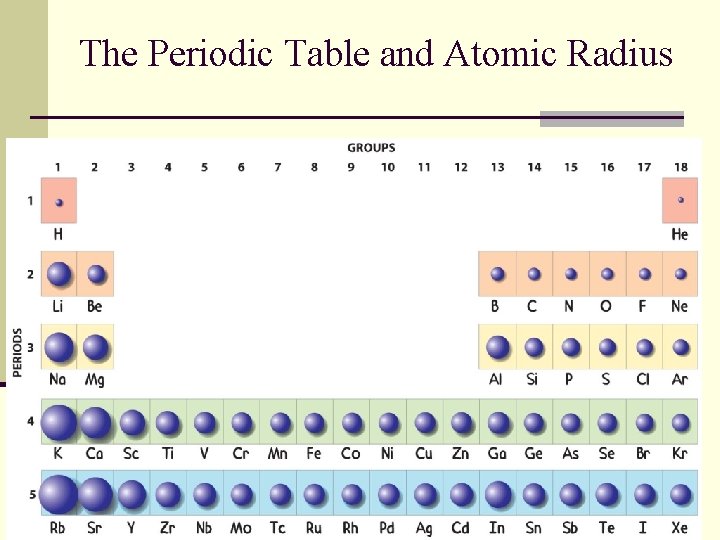
The Periodic Table and Atomic Radius

Valence Electrons n Valence electrons are the electrons in the highest occupied energy level of the atom. n Valence electrons are the only electrons generally involved in bond formation.

Electron Configuration of Sodium n 1 s 2 2 p 6 3 s 1 n Which is the valence electron for Na? n Answer: 1 s 2 2 p 6 3 s 1
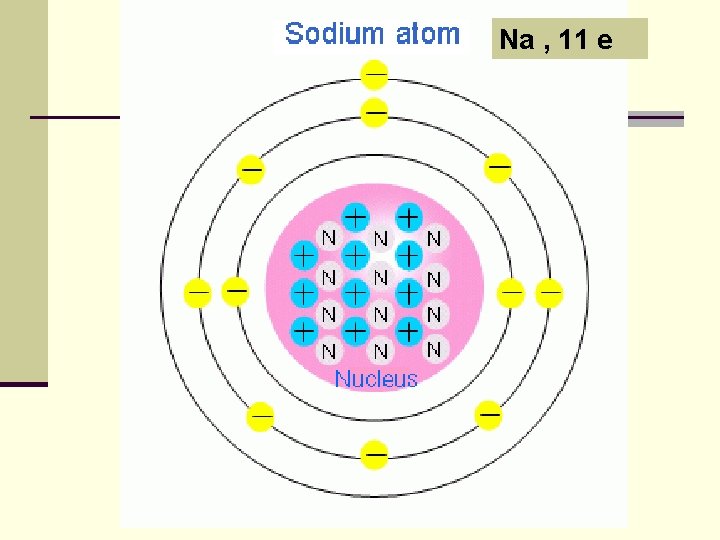
Na , 11 e
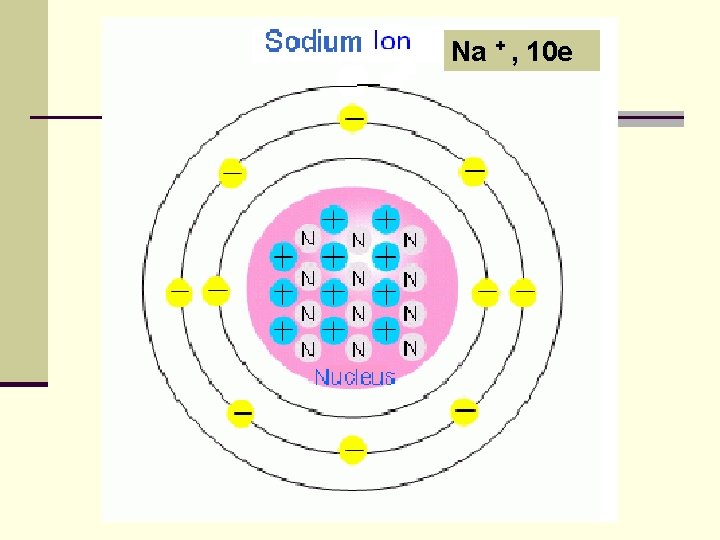
Na + , 10 e

A Positive Ion (Cation): n An atom that has lost one or more electrons. n + charge
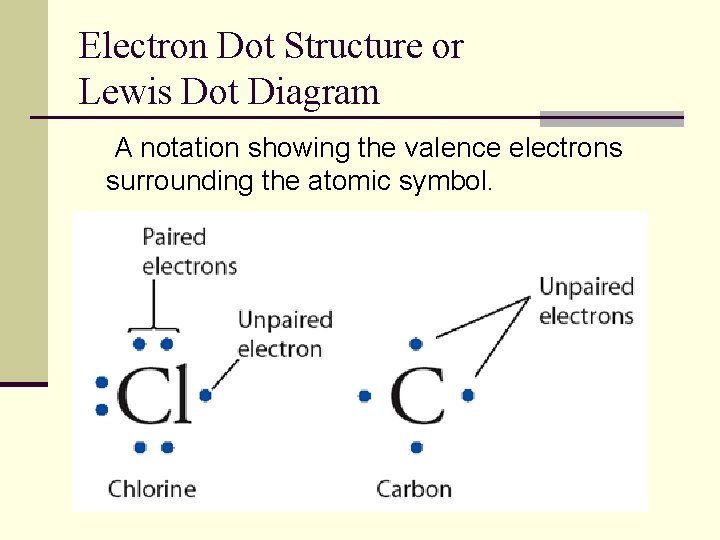
Electron Dot Structure or Lewis Dot Diagram A notation showing the valence electrons surrounding the atomic symbol.
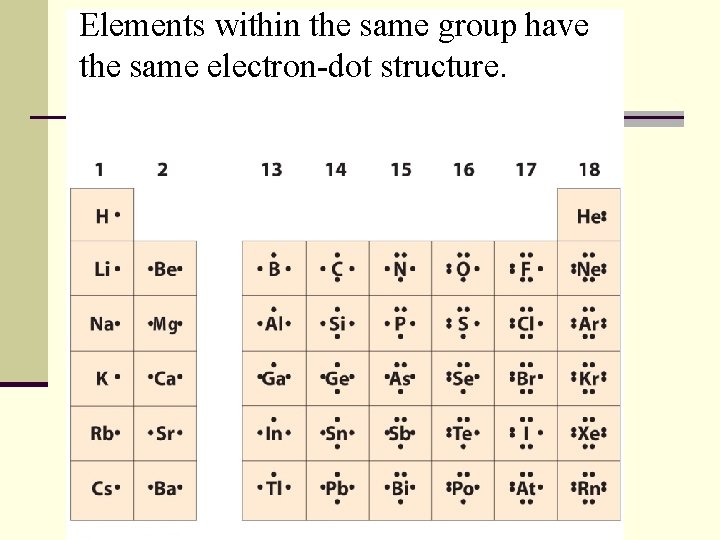
Elements within the same group have the same electron-dot structure.

A Negative Ion (Anion) n An atom that has gained one or more electrons. n Negatively (-) charged.
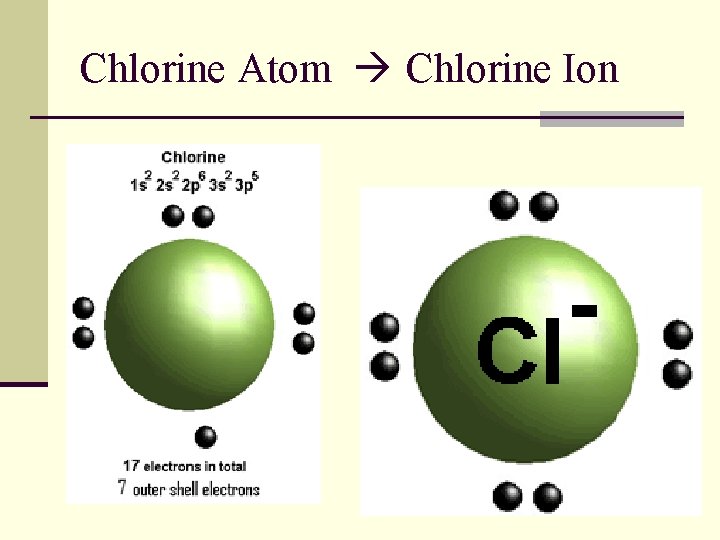
Chlorine Atom Chlorine Ion
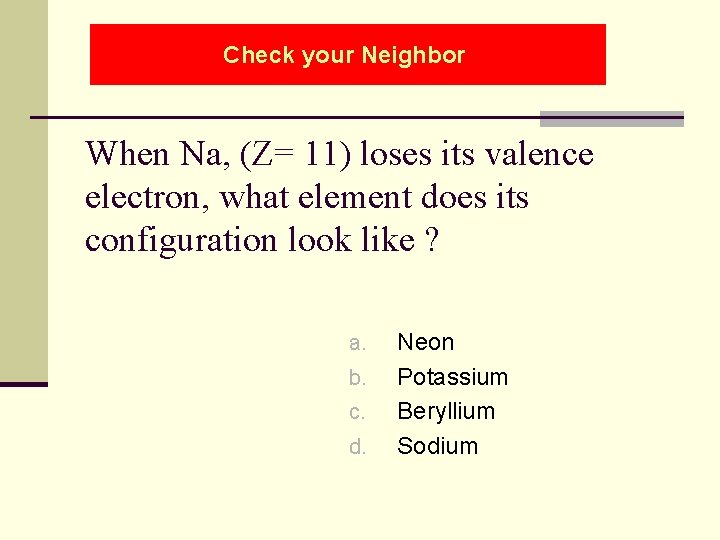
Check your Neighbor When Na, (Z= 11) loses its valence electron, what element does its configuration look like ? a. b. c. d. Neon Potassium Beryllium Sodium
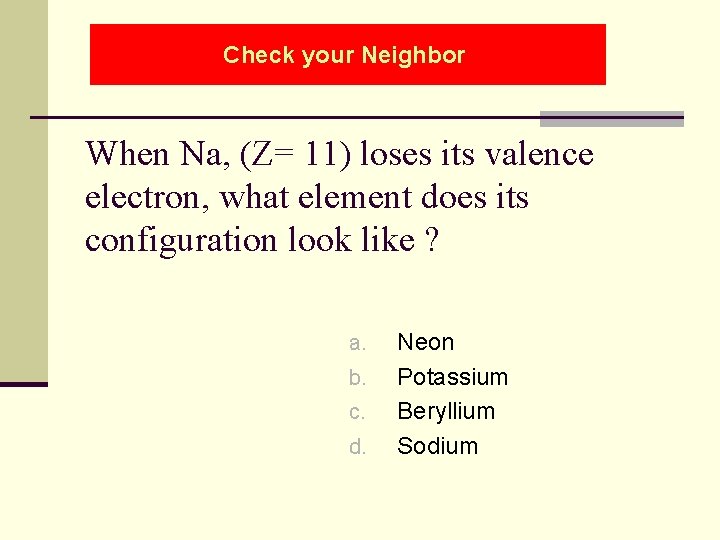
Check your Neighbor When Na, (Z= 11) loses its valence electron, what element does its configuration look like ? a. b. c. d. Neon Potassium Beryllium Sodium

A Stable Octet When the valence shell is full, the atom is stable, less likely to react. Example: Noble (Inert Gases) Kr

Note n Atoms tend to react in a way that would lead them to have a stable octet.

Part B: n Equations for Formation of Ions and Naming Ions n Ionic Bonds and Ionic Compounds

Equations for the Formation of Cations. H H+ + e- . Li Li+ + e- Mg: Mg 2+ + 2 e-

Equations for the Formation of Anions . : F: . . + e- : F - . . :

Naming cations (+). n Use the name of the metal. n Example: Na+ n Sodium ion.

Naming Anions(-) n Use the name of the nonmetal with the ending –ide. n Example: Fn Fluoride

Ionic Compounds n Are made up of: n a metal and a nonmetal ion. n polyatomic ions.

Naming Ionic Compounds, a First Look n Example: n Na. F Sodium Fluoride

Example: n Which of the following compounds are ionic? 1. 2. 3. 4. 5. Na. Cl CO 2 CO KF KNO 3

Some Common Ions

Bonding n Elements that do not have a complete octet, tend to react. n When the elements react bonds are formed.

Types of Bonds n Ionic n Metallic n Covalent

Ionic Bonds n Bonds that are formed by transfer of electrons from one element to the other. n Each element (now an ion) will have a complete octet after the transfer of electrons.
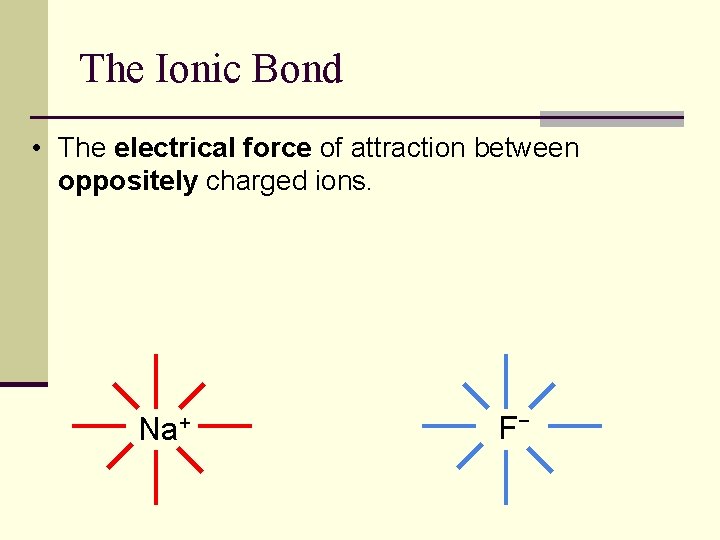
The Ionic Bond • The electrical force of attraction between oppositely charged ions. Na+ F−

Example: Formation of Lithium Fluoride . . Li + : F: . . Li+ : F- . . :

Is an Ionic Compound Electrically Neutral or Charged?
Movie Clip n http: //www. youtube. com/watch? v=Kj 3 o 0 Xvh. V q. Q&NR=1

Chemical Formula n A representation of the kinds and number of atoms in a substance.

Formula Unit n A chemical formula that shows the lowest whole number ratio of the atoms (ions) in an ionic compound. n Example: KCl, Mg Cl 2

The formula unit is used because ionic compounds have a lattice arrangement of ions. Ex: Na. Cl n Which ball represents the Na?
- Slides: 35