Ionic Bonding pg 82 Valence e transfer to

Ionic Bonding pg. 82 • Valence e- transfer to form crystal lattices • Metals (+) transfer e- to Nonmetals (-) • Cations (+) transfer e- to Anions (-) emeans electrons

Binary Ionic Compounds Made from two elements Cation keeps its name Ex: Magnesium (+2) Anion adds suffix –ide Ex: Fluoride (-1) …becomes Magnesium Fluoride Mg. F₂ pg. 82

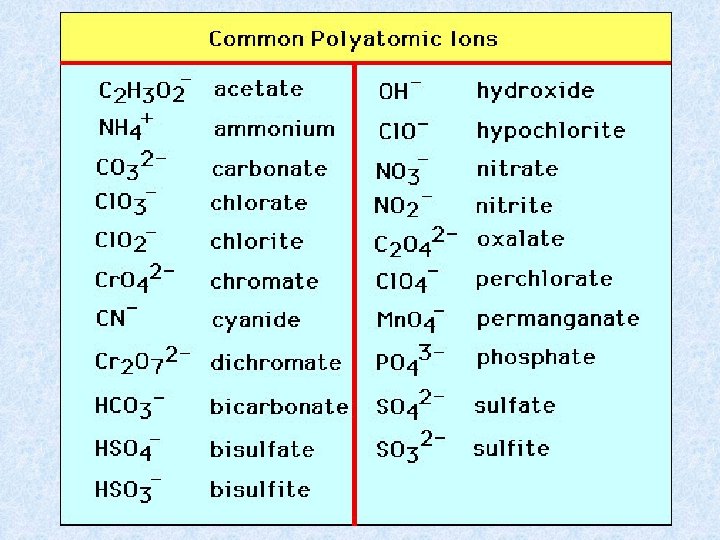
Polyatomic Ions pg. 82 • Covalently bonded group of atoms acting as one unit that can bond with other ions. • Positive or Negative Ex: Ammonium NH 4+ Hydroxide OHNitrate NO 3 Sulfate SO 42 -

What is the correct formula for a compound containing sodium and sulfur? • a. Na. S • b. Na 2 S c. Na. S 2 d. Na. S 3

What is the correct formula for a compound containing sodium and sulfur? • a. Na. S • b. Na 2 S • Answer: b c. Na. S 2 d. Na. S 3

Oxidation numbers are used to • a. keep track of the neutrons lost or gained by each atom. • b. keep track of the protons lost or gained by each atom. • c. keep track of the electrons lost or gained by each atom. • d. keep track of the quarks lost or gained by each atom.

Oxidation numbers are used to • a. • b. • c. • d. keep track of the neutrons lost or gained by each atom. keep track of the protons lost or gained by each atom. keep track of the electrons lost or gained by each atom. keep track of the quarks lost or gained by each atom. • Answer: c

The formation of an ionic bond involves the • a. transfer of electrons. • b. transfer of protons. • c. transfer of neutrons. • d. none of the above.

The formation of an ionic bond involves the • • a. transfer of electrons. b. transfer of protons. c. transfer of neutrons. d. none of the above. • Answer: a • Valence e- from metal to nonmetal

Fluorine, F, forms a binary ionic compound with lithium, Li. What is the name of this compound? • a. fluorine lithide • b. lithium fluorine • c. lithium fluoride • d. fluorine lithium

Fluorine, F, forms a binary ionic compound with lithium, Li. What is the name of this compound? • • a. b. c. d. fluorine lithide lithium fluorine lithium fluoride fluorine lithium • Answer: c • Metal/Cation first, add ide to nonmetal/anion
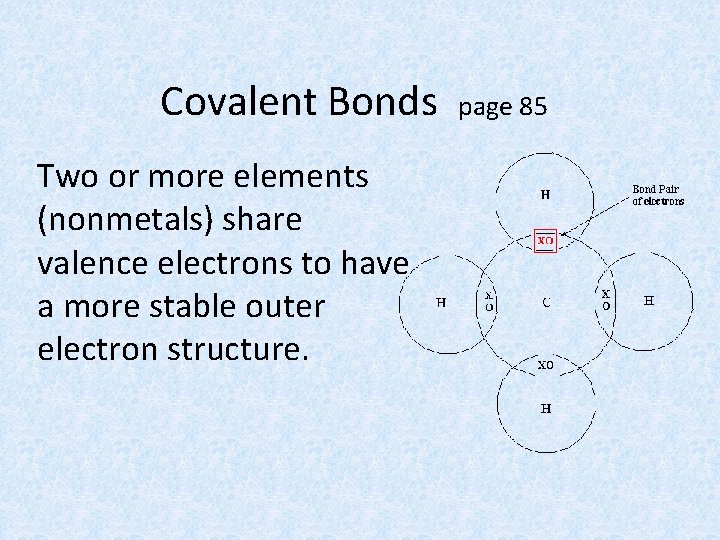
Covalent Bonds Two or more elements (nonmetals) share valence electrons to have a more stable outer electron structure. page 85

What is a Molecule? Pg. 85 • Only compounds formed from covalent bonds are called molecules. Metals bonded to nonmetals form ionic crystals not molecules! Circle the molecules and put an X on the compounds that are not molecules. Na. Cl Na. OH CO 2 H 2 O CH 4 Al 2 O 3 Ca. Cl 2 Br 2 C 6 H 12 O 6 N 2 KI C 4 H 10

3 Types of Bonding pg. 85 • Ionic Bonds – Metal ions bond to nonmetal ions • Covalent Bonds – Nonmetal atoms bond with each other • Metallic Bonds – Metals bond with each other


Properties of Covalent Compounds • Share electrons between nonmetal atoms (elements on right side of the periodic table) • Low melting and boiling points • Brittle • Do not conduct electricity well
- Slides: 17