Ionic bonding metal nonmetal Ionic bonds form a


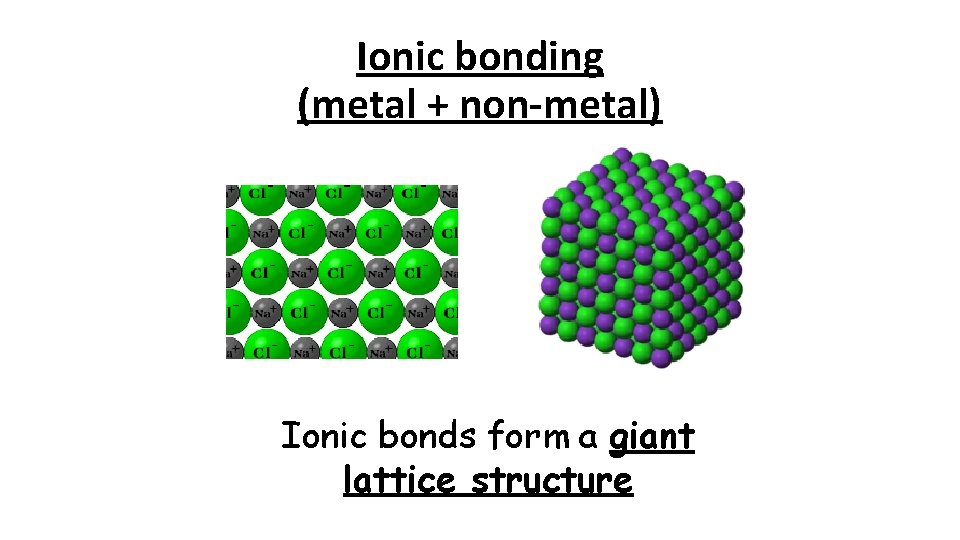
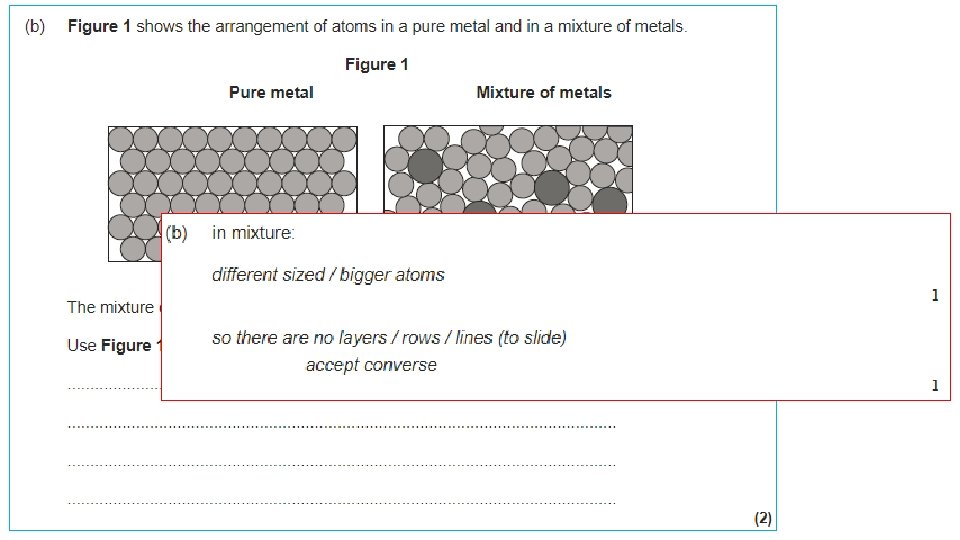
Ionic bonding (metal + non-metal) Ionic bonds form a giant lattice structure
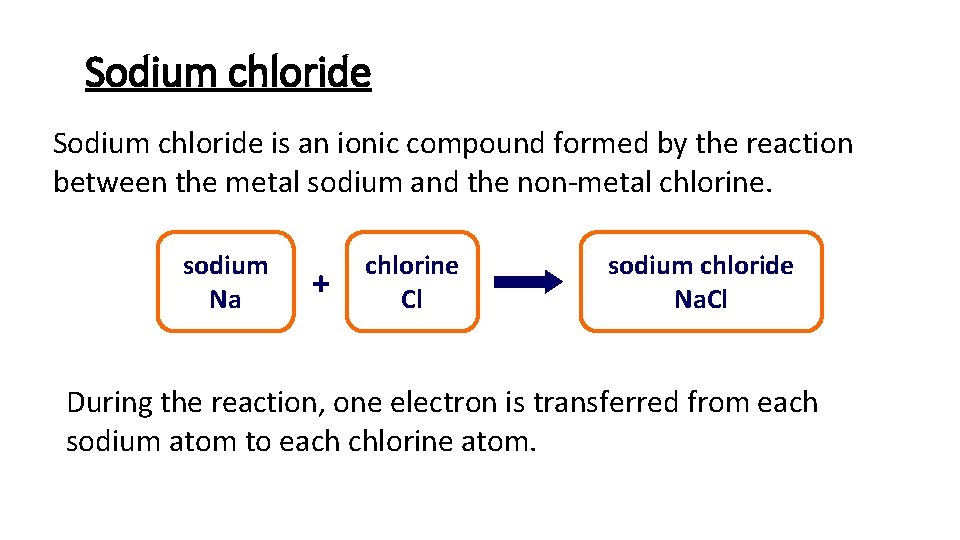
Sodium chloride is an ionic compound formed by the reaction between the metal sodium and the non-metal chlorine. sodium Na + chlorine Cl sodium chloride Na. Cl During the reaction, one electron is transferred from each sodium atom to each chlorine atom.
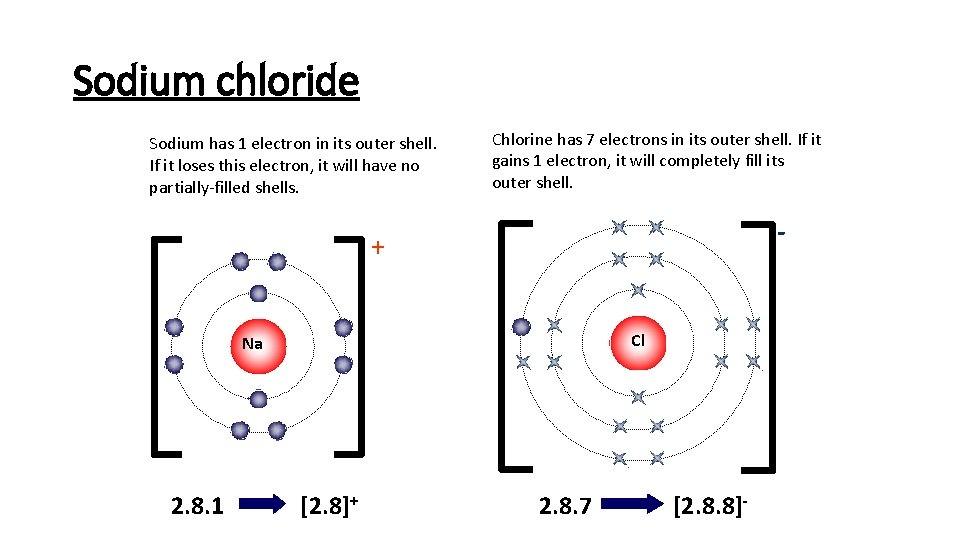
Sodium chloride Sodium has 1 electron in its outer shell. If it loses this electron, it will have no partially-filled shells. Chlorine has 7 electrons in its outer shell. If it gains 1 electron, it will completely fill its outer shell. - + Cl Na 2. 8. 1 [2. 8]+ 2. 8. 7 [2. 8. 8]-

Sodium chloride The positive sodium ions and the negative chloride ions are strongly attracted to each other and form an ionic bond. - + Na Cl
![Lithium Oxide + Li 2. 1 2[2]+ O + Li 2. 6 [2. 8]2 Lithium Oxide + Li 2. 1 2[2]+ O + Li 2. 6 [2. 8]2](http://slidetodoc.com/presentation_image_h2/6d3d3462c2d54f116938213433958444/image-7.jpg)
Lithium Oxide + Li 2. 1 2[2]+ O + Li 2. 6 [2. 8]2 -
![- Magnesium fluoride F 2+ [2. 8]- 2. 7 Mg F 2. 8. 2 - Magnesium fluoride F 2+ [2. 8]- 2. 7 Mg F 2. 8. 2](http://slidetodoc.com/presentation_image_h2/6d3d3462c2d54f116938213433958444/image-8.jpg)
- Magnesium fluoride F 2+ [2. 8]- 2. 7 Mg F 2. 8. 2 [2. 8]2+ -
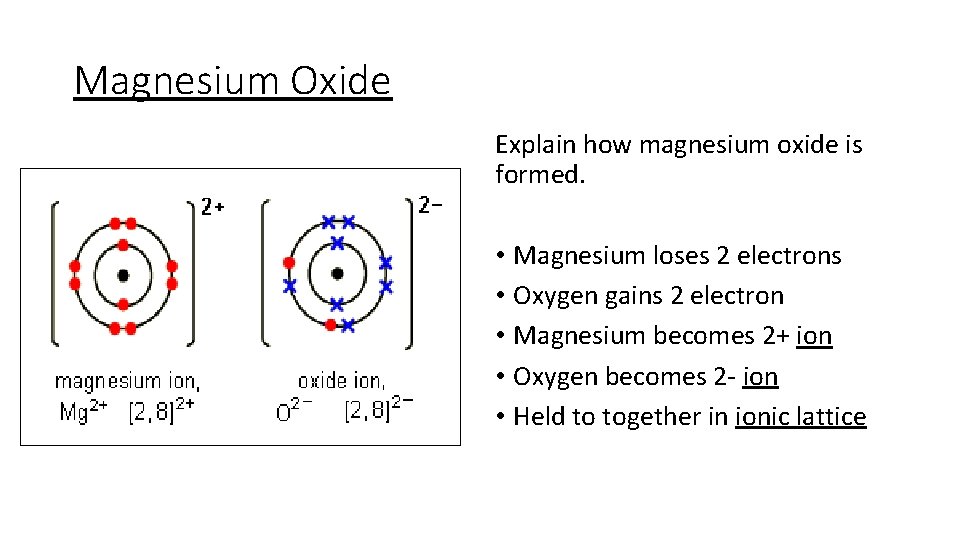
Magnesium Oxide Explain how magnesium oxide is formed. • Magnesium loses 2 electrons • Oxygen gains 2 electron • Magnesium becomes 2+ ion • Oxygen becomes 2 - ion • Held to together in ionic lattice
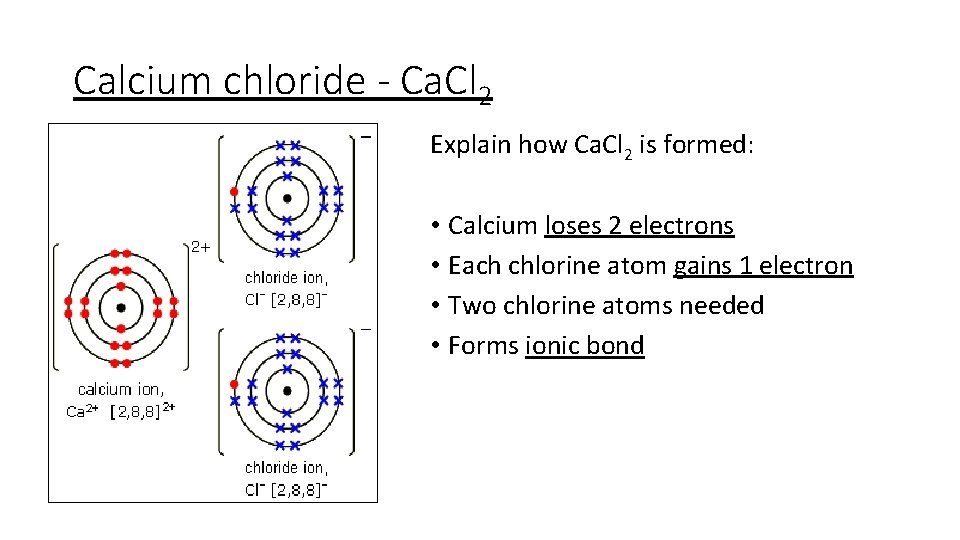
Calcium chloride - Ca. Cl 2 Explain how Ca. Cl 2 is formed: • Calcium loses 2 electrons • Each chlorine atom gains 1 electron • Two chlorine atoms needed • Forms ionic bond


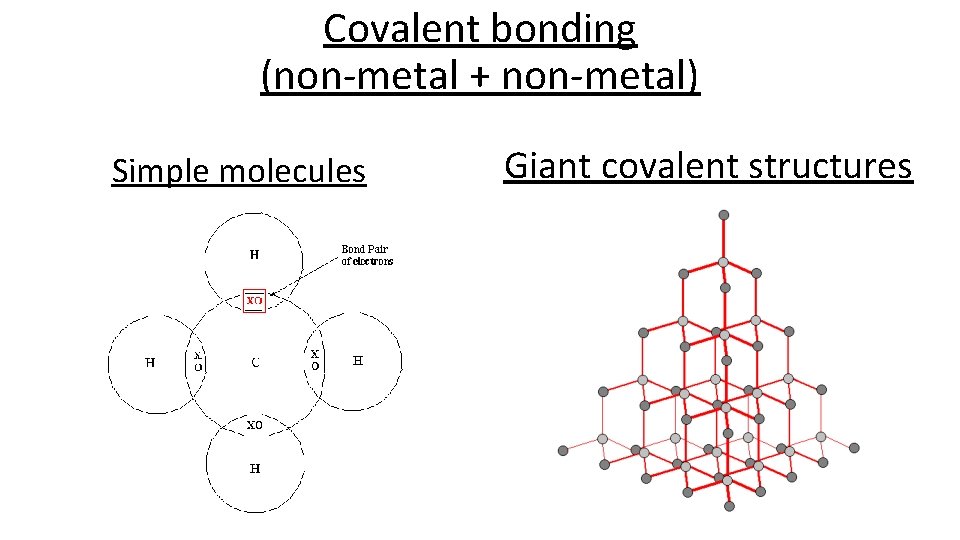
Covalent bonding (non-metal + non-metal) Simple molecules Giant covalent structures

Simple molecules HYDROGEN WAYS TO REPRESENT THE MOLECULE H Hydrogen atom needs one electron to complete its outer shell H Another hydrogen atom also needs one electron to complete its outer shell H H atoms share a pair of electrons to form a single covalent bond A hydrogen MOLECULE is formed
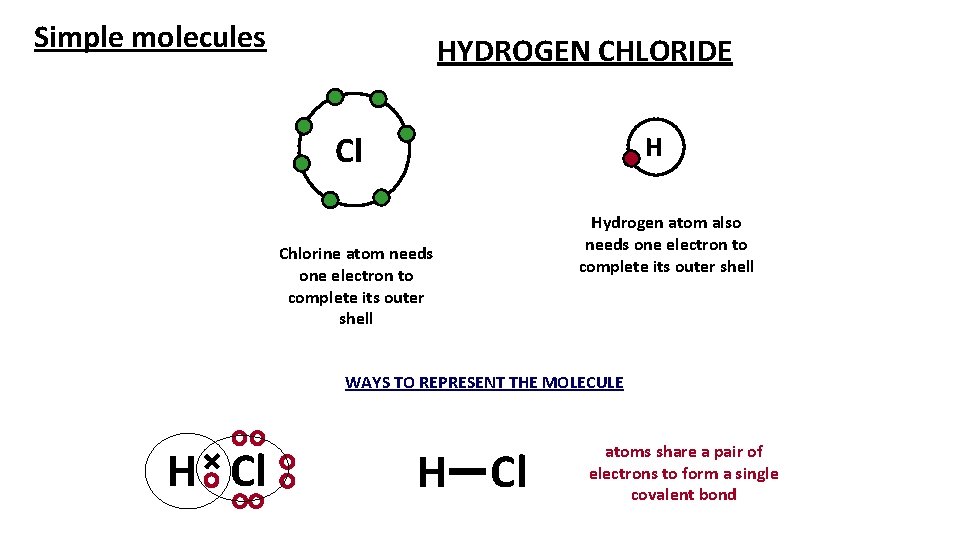
Simple molecules HYDROGEN CHLORIDE Cl H Chlorine atom needs one electron to complete its outer shell Hydrogen atom also needs one electron to complete its outer shell WAYS TO REPRESENT THE MOLECULE H Cl atoms share a pair of electrons to form a single covalent bond

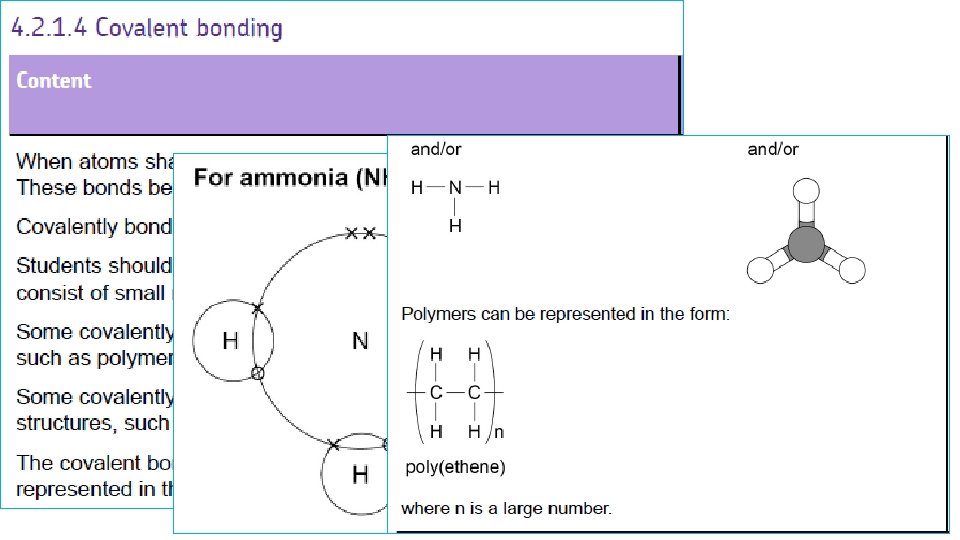
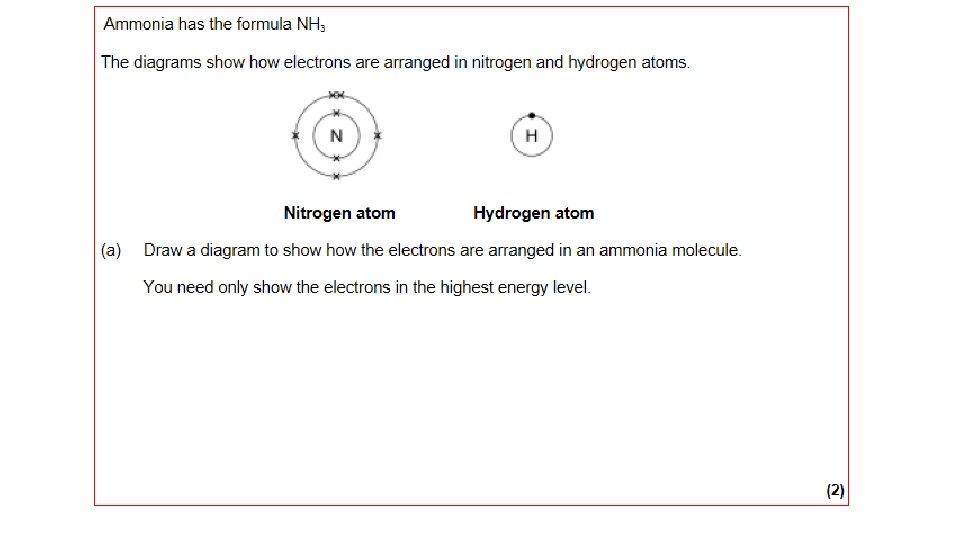
Simple molecules AMMONIA WAYS TO REPRESENT THE MOLECULE H N H H Each hydrogen atom needs one electron to complete its outer shell N H H H Nitrogen atom needs 3 electrons to complete its outer shell H N H H Nitrogen can only share 3 of its 5 electrons otherwise it will exceed the maximum of 8 A LONE PAIR REMAINS

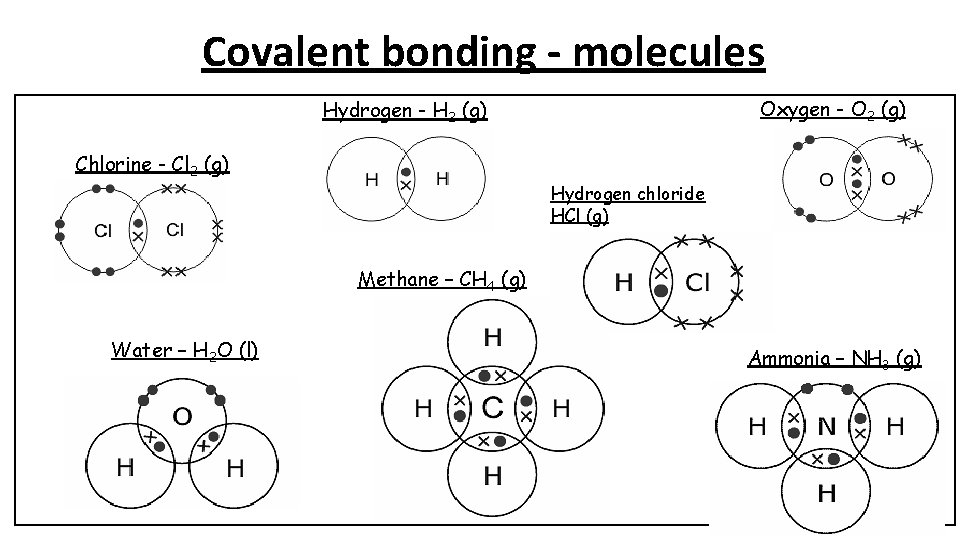
Covalent bonding - molecules Oxygen - O 2 (g) Hydrogen - H 2 (g) Chlorine - Cl 2 (g) Hydrogen chloride HCl (g) Methane – CH 4 (g) Water – H 2 O (l) Ammonia – NH 3 (g)
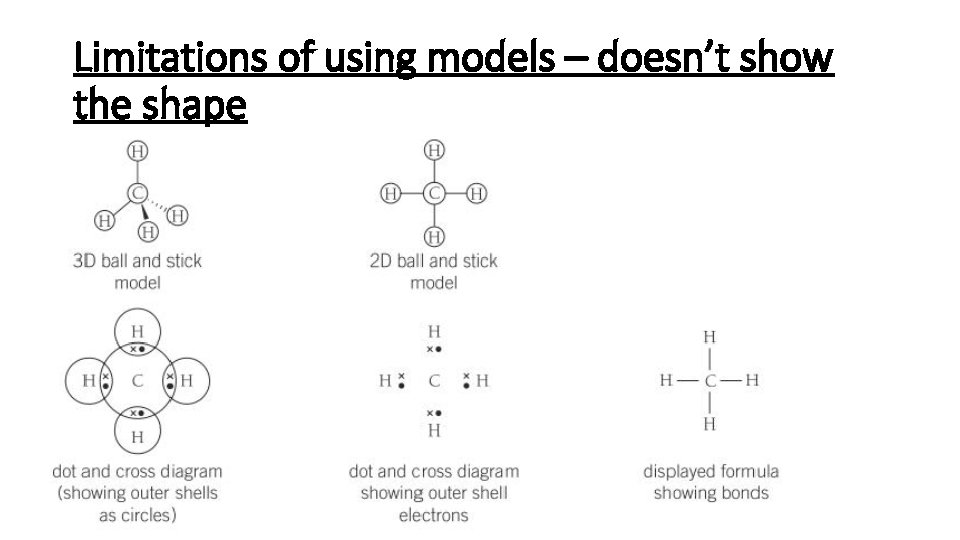
Limitations of using models – doesn’t show the shape


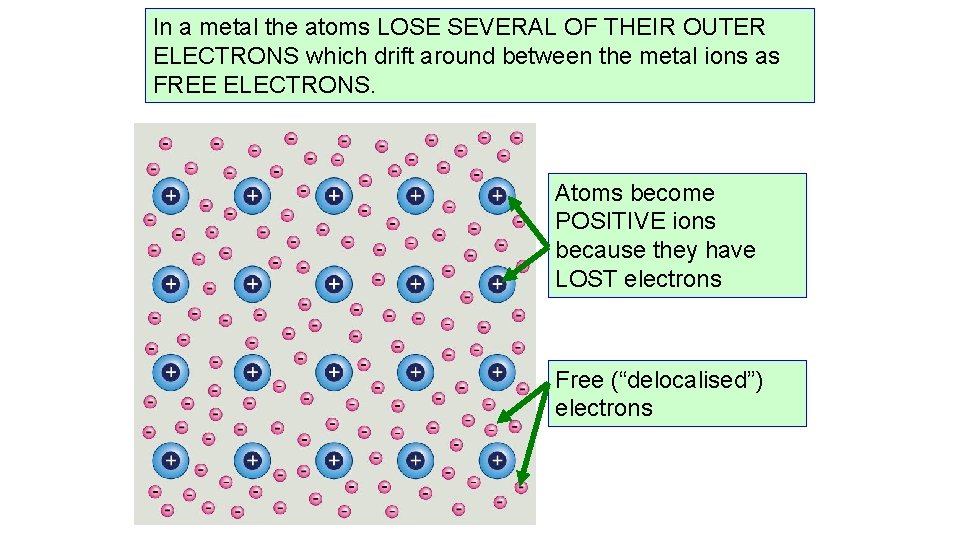
In a metal the atoms LOSE SEVERAL OF THEIR OUTER ELECTRONS which drift around between the metal ions as FREE ELECTRONS. Atoms become POSITIVE ions because they have LOST electrons Free (“delocalised”) electrons
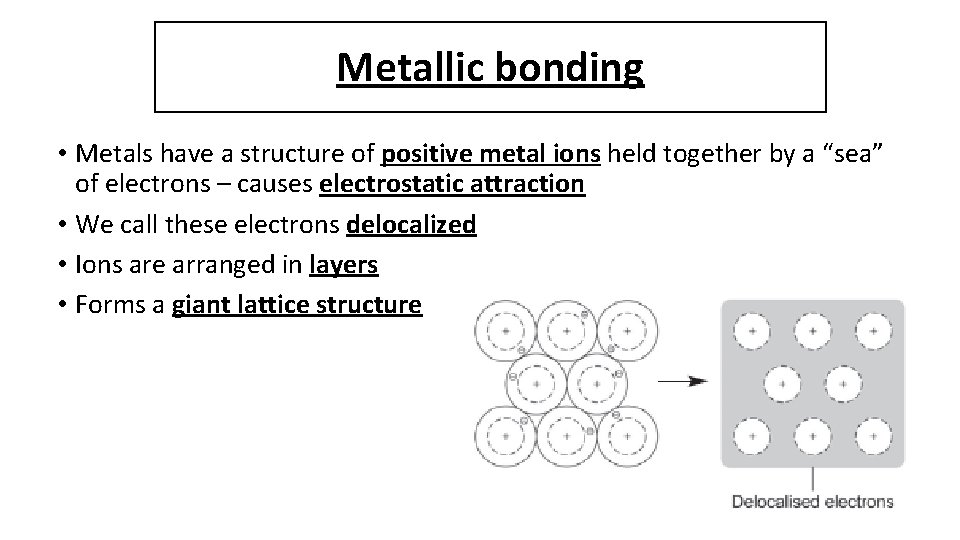
Metallic bonding • Metals have a structure of positive metal ions held together by a “sea” of electrons – causes electrostatic attraction • We call these electrons delocalized • Ions are arranged in layers • Forms a giant lattice structure




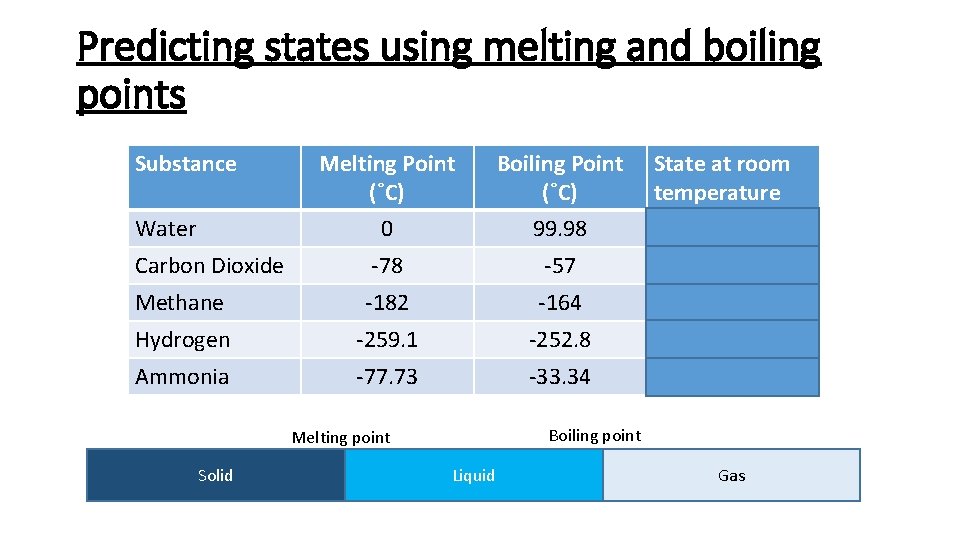
Predicting states using melting and boiling points Substance Melting Point (˚C) Boiling Point (˚C) 0 99. 98 Liquid Carbon Dioxide -78 -57 Gas Methane -182 -164 Gas Hydrogen -259. 1 -252. 8 Gas Ammonia -77. 73 -33. 34 Gas Water Boiling point Melting point Solid State at room temperature Liquid Gas


State Symbols (s) – solid (l) – liquid (g) – gas (aq) – dissolved in water Na. OH (aq) → Na+ (aq) + OH– (aq)


Properties of ionic compounds – giant lattices High Melting point – lots of ENERGY needed to break the strong bonds (strong electrostatic attraction) Solubility - Can dissolve in water which enables the ions to move Conduction - When MOLTEN or DISSOLVED IN WATER, ionic compounds can conduct electricity because the ions can carry current/charge (not electricity)






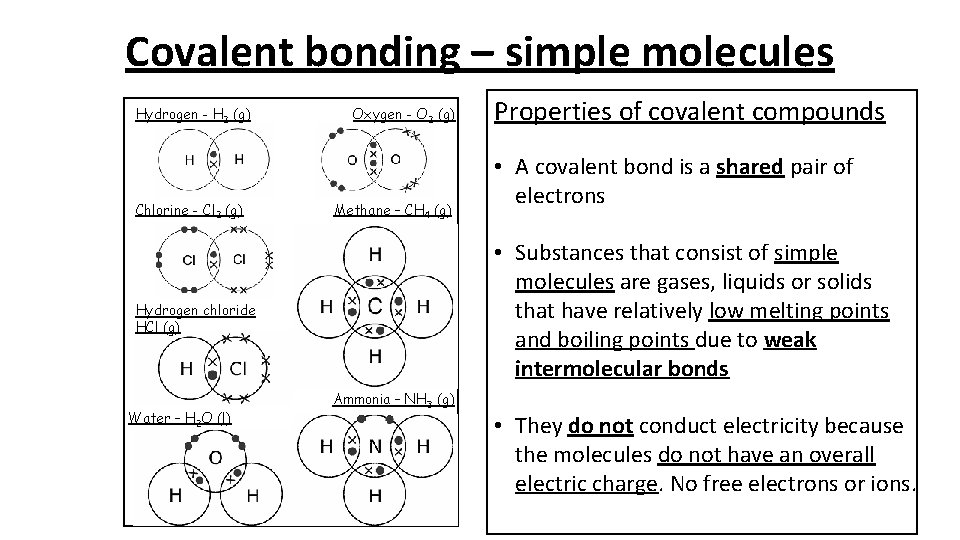
Covalent bonding – simple molecules Hydrogen - H 2 (g) Chlorine - Cl 2 (g) Oxygen - O 2 (g) Methane – CH 4 (g) • A covalent bond is a shared pair of electrons • Substances that consist of simple molecules are gases, liquids or solids that have relatively low melting points and boiling points due to weak intermolecular bonds Hydrogen chloride HCl (g) Water – H 2 O (l) Properties of covalent compounds Ammonia – NH 3 (g) • They do not conduct electricity because the molecules do not have an overall electric charge. No free electrons or ions.





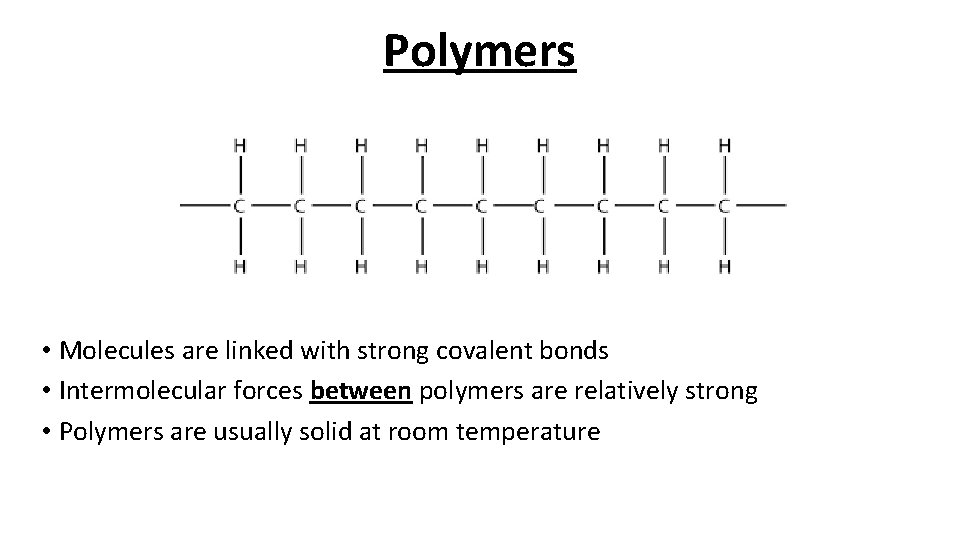
Polymers • Molecules are linked with strong covalent bonds • Intermolecular forces between polymers are relatively strong • Polymers are usually solid at room temperature



Carbon structures - diamond Giant covalent structure of diamond Diamond • Diamond is made only from carbon atoms. • Every carbon makes four covalent bonds to achieve a full outer shell. • Every carbon atom is bonded to four other carbon atoms. • This means the structure keeps on growing! • We make a Giant Covalent Structure. Key properties – • Diamond is very hard. • High melting points – because it has strong covalent bonds (which take a lot of energy to break)


Carbon structures - graphite Giant covalent structure of graphite Graphite • Graphite is made only from carbon atoms. • Every carbon makes 3 covalent bonds to achieve a full outer shell. • Forms hexagonal rings, arranged in layers • Weak intermolecular forces between the layers • Every carbon atom is bonded to three other carbon atoms Key properties – • High melting point • Conducts electricity – free • Graphite is very soft. electrons • Slippery – arranged in layers









Metallic bonding – giant structures Conducts electricity and heat Delocalised electrons can move through the structure Malleable Ions arranged in layers so ions are able to slide over each other High melting and boiling points Ions held together by strong electrostatic attraction so needs a lot of energy to break the bonds
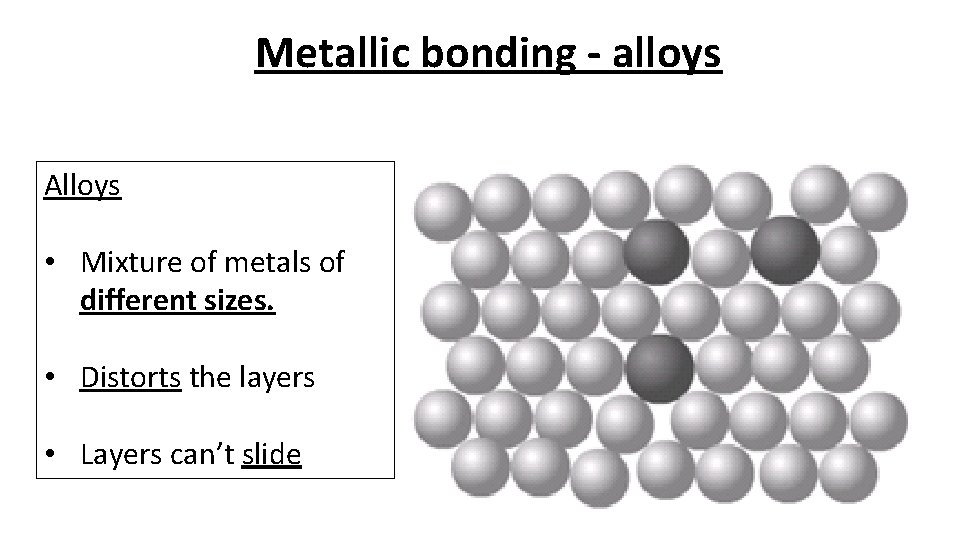
Metallic bonding - alloys Alloys • Mixture of metals of different sizes. • Distorts the layers • Layers can’t slide







Covalent bonding - Giant Besides graphite and diamond, carbon can also form another type of giant covalent structure. Fullerenes (named after the scientists that discovered them) are made by conjoined hexagonal carbon rings Possible uses of Fullerenes in the future could be: • Drug delivery • In lubricants • As catalysts in reactions • To make carbon nanotubes to reinforce structures


Nano particles • Structures that are 1 -100 nm in size, or of the order of a few hundred atoms Particle name Symbol Diameter (nm) Smaller Nano particle than 100 – 2500 nm PM 2. 5 Coarse PM 10 2500 nm – 10 000 nm particle Diameter (m) 1 x 10 -7 m – 2. 5 x 10 -6 m – 1 x 10 -5 m

Nano particles

Nanoparticles Advantages • Large surface area makes them effective catalysts. • Nanotubes can be used in small scale circuits as nanowires. Disadvantages • So small they can enter the skin and therefore the bloodstream. • Easily become airborne, breathing in can potentially damage the lungs. Nanoparticles are present in sun screens May be used to develop faster computers, lighter construction materials and new coatings




- Slides: 79