Ionic bonding Li O 2 Li O Li2O2
![Ionic bonding: Li + O 2 Li + O [Li]2+[O]2– or 4 Li + Ionic bonding: Li + O 2 Li + O [Li]2+[O]2– or 4 Li +](https://slidetodoc.com/presentation_image/3e3a82b6f525da9e155768ac01b09a73/image-1.jpg)
Ionic bonding: Li + O 2 Li + O [Li]2+[O]2– or 4 Li + O 2 2[Li]2+[ O]2– 3 p+ 2 e-1 e 4 n 0 1 e 6 e- 2 e- 3 p+ 2 e-1 e 4 n 0 Li Li 1 e- O 3 p+ 2 e 4 n 0 8 e- 2 e- 8 p+ 8 n 0 3 p+ 2 e 4 n 0 [Li]2+ [ O ]2–
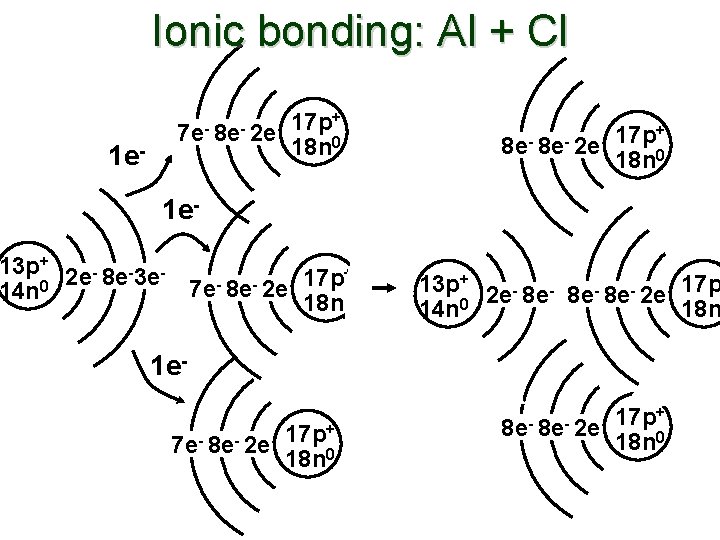
Ionic bonding: Al + Cl 1 e- 7 e- 8 e- 2 e- 17 p+ 18 n 0 1 e- 13 p+ 2 e- 8 e-3 e+ 17 p 7 e 8 e 2 e 14 n 0 18 n 0 13 p+ 2 e- 8 e- 8 e- 2 e- 17 p 14 n 0 18 n 1 e 7 e- 8 e- 2 e- 17 p+ 18 n 0
![Ionic bonding: Al + Cl Al + 3 Cl [Al]3+[Cl]3– Cl Al Cl Cl Ionic bonding: Al + Cl Al + 3 Cl [Al]3+[Cl]3– Cl Al Cl Cl](http://slidetodoc.com/presentation_image/3e3a82b6f525da9e155768ac01b09a73/image-3.jpg)
Ionic bonding: Al + Cl Al + 3 Cl [Al]3+[Cl]3– Cl Al Cl Cl [Al]3+ [ Cl ] 3–

Ionic vs. Covalent bonding • Thus far we have looked at when atoms bond due to the transfer of electrons • An ionic bond forms when an atom has a greater attraction for e–s than a second atom • However, if two atoms have approximately the same pull on electrons, they share the electrons (forming a “covalent” bond) • Read “Covalent bonding” and “Covalent bonding continued”.
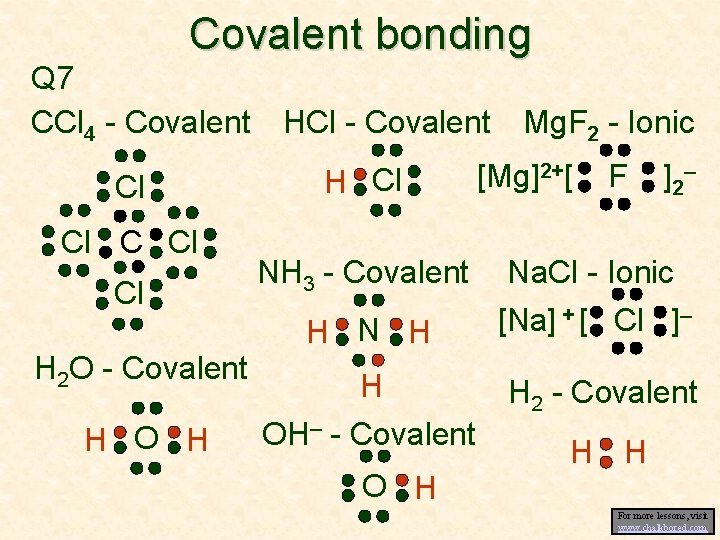
Covalent bonding Q 7 CCl 4 - Covalent Cl Cl Cl H 2 O - Covalent H O H HCl - Covalent H Cl Mg. F 2 - Ionic [Mg]2+[ F ] 2– NH 3 - Covalent Na. Cl - Ionic H N H [Na] + [ Cl ]– H OH– - Covalent H 2 - Covalent O H H H For more lessons, visit www. chalkbored. com
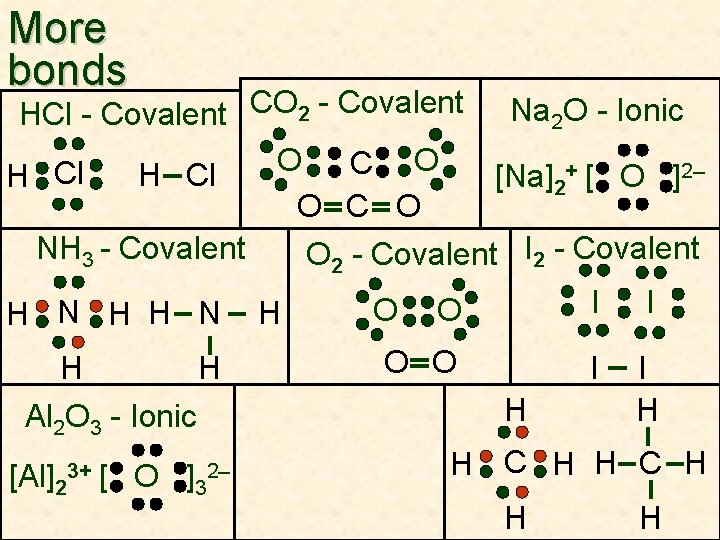
More bonds HCl - Covalent CO 2 - Covalent Na 2 O - Ionic O O C H Cl [Na]2+ [ O ]2– O C O NH 3 - Covalent O 2 - Covalent I 2 - Covalent H N H H Al 2 O 3 - Ionic [Al]23+ [ O ]32– H O I I O O I I H O H H C H H H
- Slides: 6