Ionic Bonding Ionic Bonding What Is It Ionic
Ionic Bonding
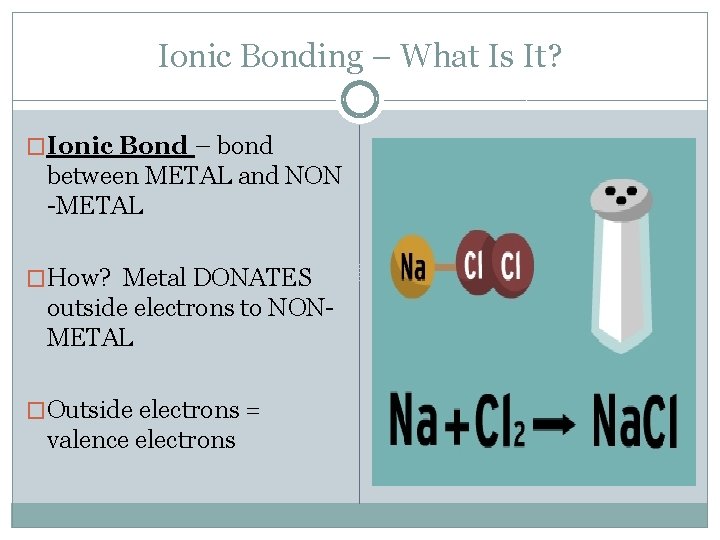
Ionic Bonding – What Is It? �Ionic Bond – bond between METAL and NON -METAL �How? Metal DONATES outside electrons to NONMETAL �Outside electrons = valence electrons
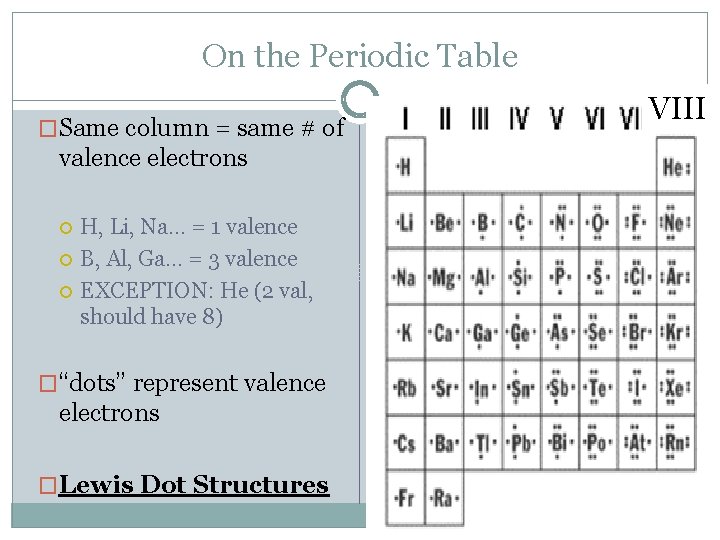
On the Periodic Table �Same column = same # of valence electrons H, Li, Na… = 1 valence B, Al, Ga… = 3 valence EXCEPTION: He (2 val, should have 8) �“dots” represent valence electrons �Lewis Dot Structures VIII

Lewis Dot Structures �Example: Nitrogen (N) �Valence Electrons: 5 . . N. . �TREAT IT LIKE A SCHOOL DANCE �Symbol in center �One valence electron on each side
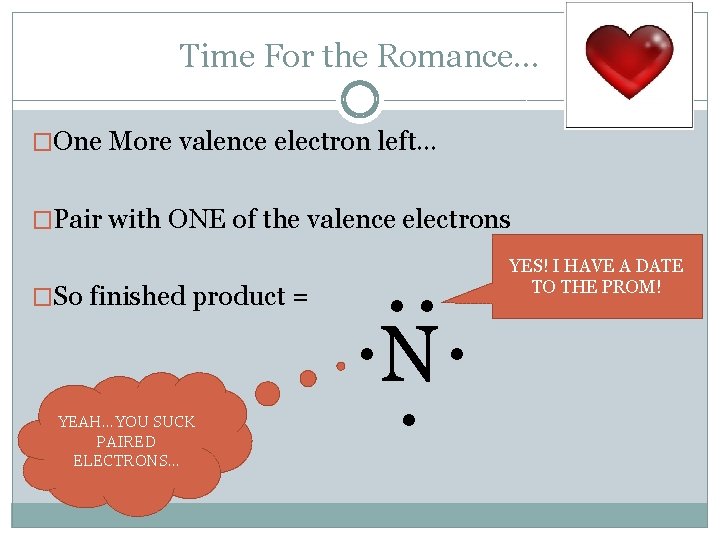
Time For the Romance… �One More valence electron left… �Pair with ONE of the valence electrons �So finished product = YEAH…YOU SUCK PAIRED ELECTRONS… . . . N. . YES! I HAVE A DATE TO THE PROM!

Quick Quiz #1 �Write the Lewis Dot Structures for the Following: �Hydrogen �Aluminum �Carbon �Sulfur �Bromine �Krypton

Lewis Dot and Ionic Bonding Metal Donates to Non-Metal �Metal: LOSES electron (positive charge) �Non-Metal: GAINS electron (negative charge) Metals on LEFT side of table Non-Metals on RIGHT side Goal: Non-metal = 8 valence
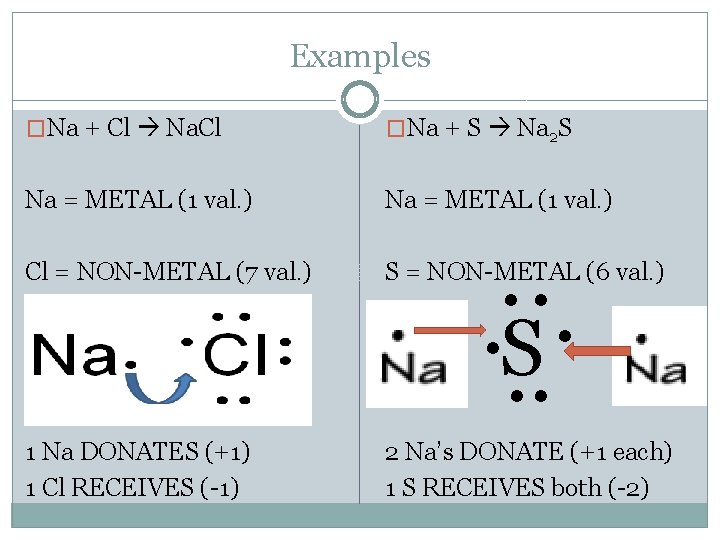
Examples �Na + Cl Na. Cl �Na + S Na 2 S Na = METAL (1 val. ) Cl = NON-METAL (7 val. ) S = NON-METAL (6 val. ) 1 Na DONATES (+1) 1 Cl RECEIVES (-1) 2 Na’s DONATE (+1 each) 1 S RECEIVES both (-2) . . S. .

Charged Atoms �Ions – atoms with a positive or negative charge �Metals – LOSE electrons (positive charge) = cation Cats are cute POSITIVE �Non-Metals – GAIN electrons (negative charge) = anion Onions smell bad Negative
- Slides: 9