IONIC BONDING IONIC BONDING Usually between metal nonmetal

IONIC BONDING

IONIC BONDING • Usually between metal & nonmetal elements (or can have polyatomic ions) • Electrons are transferred between elements • Nonmetal takes the e- from a metal • Forms Ions: Cations & Anions • Electrostatic forces hold ions together
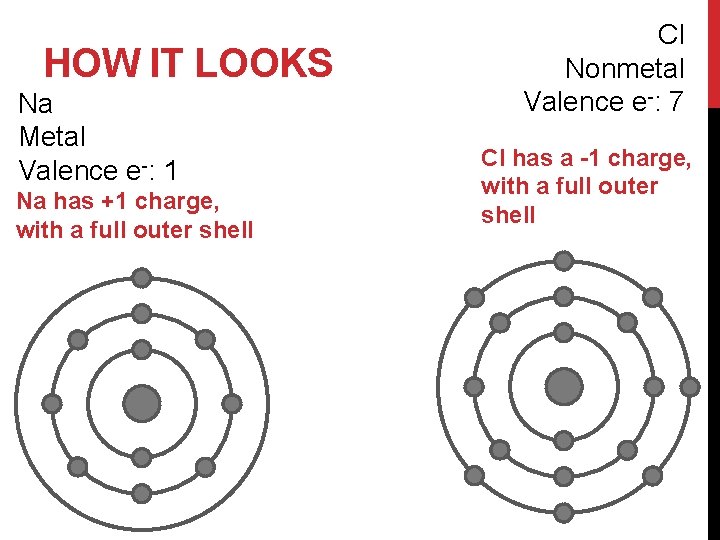
HOW IT LOOKS Na Metal Valence e-: 1 Na has +1 charge, with a full outer shell Cl Nonmetal Valence e-: 7 Cl has a -1 charge, with a full outer shell
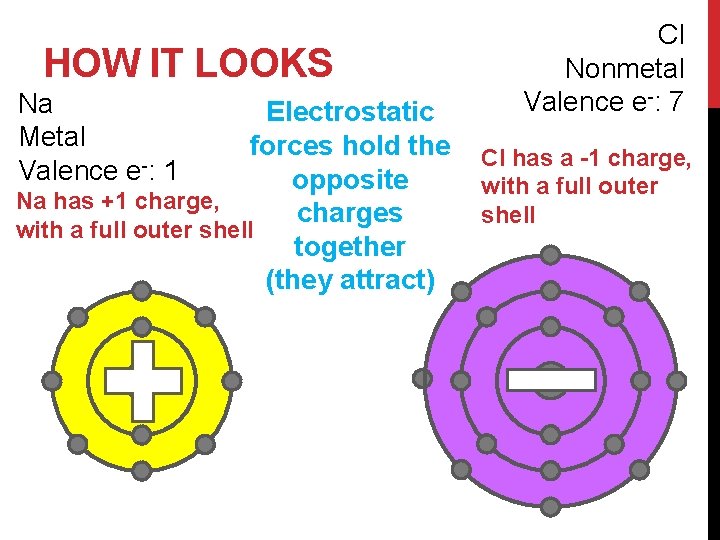
HOW IT LOOKS Na Metal Valence e-: 1 Electrostatic forces hold the opposite Na has +1 charge, charges with a full outer shell together (they attract) Cl Nonmetal Valence e-: 7 Cl has a -1 charge, with a full outer shell

FORMULA UNIT VS. COMPOUNDS • Formula unit: simplest collection of ions • Ionic Compounds: compound of cations & anions that combine so that the overall charge is neutral Formula Unit: Na+ & Cl- -++-+ -+ - Ionic Compound: Na. Cl

-++-+ • Crystal lattice structure - + STRUCTURE • repeating arrangement of ions • Typically solids

MELTING AND BOILING POINT • Ions are strongly bonded together by electrostatic forces • Lots of energy needed to break attraction between ions • Results in HIGH melting points & boiling points

HARD BUT BRITTLE • A slight shift in a row of ions causes a large buildup of repulsion • Makes the layers separate completely, causing ionic compounds to-be brittle - + + + -

CONDUCTIVITY • Solid ionic compounds – do not conduct electricity • b/c overall charge is neutral • However, in aqueous form they can conduct electricity +-+ + -+ • Aqueous: dissolved in water • Compound splits into ions & can complete currents • Similarly: molten ionic compounds can
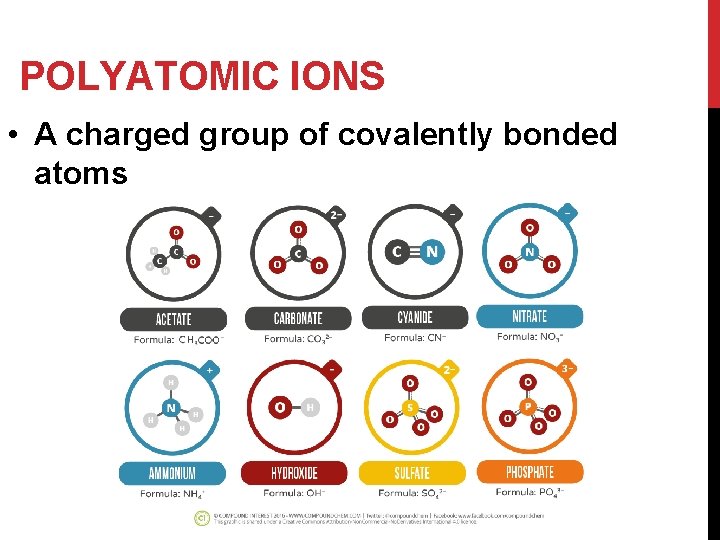
POLYATOMIC IONS • A charged group of covalently bonded atoms

WRITING IONIC FORMULAS • Elements that form cations and anions combine so that the overall charge is neutral • Cation always written first • Metal or ammonia • Anion always written second • Nonmetal or a negative polyatomic Periodic table charges are important!
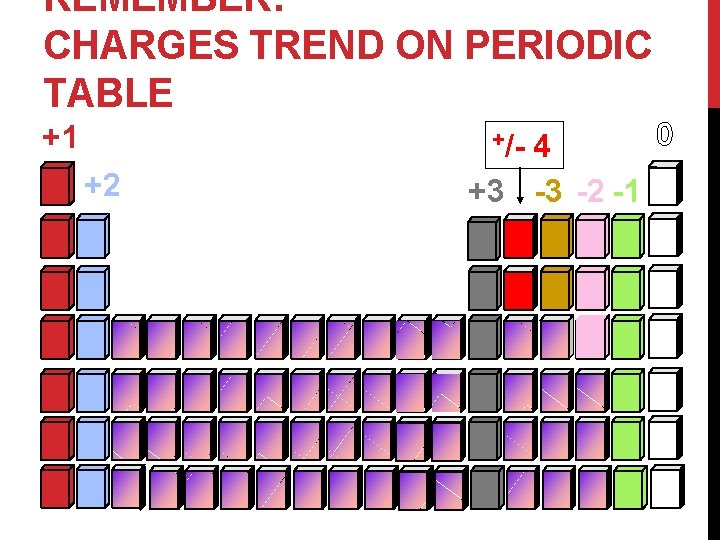
REMEMBER: CHARGES TREND ON PERIODIC TABLE +1 0 4 +3 -3 -2 -1 +/- +2

CREATING IONIC COMPOUNDS: POTASSIUM & FLUORINE K Potassium – 1 valence e-, wants to lose 1 e- F Fluorine – 7 valence e-, wants to gain 1 e- K +1 F -1 • K will transfer an e- to F • Now F is full, charge becomes -1, and K is filled becoming a +1 • The formula is written by counting the # of K’s and F’s present to fill all outer shells. • Neutral Ionic Compound b/c charges add to zero. + 1 + -1 = 0 Ans: KF
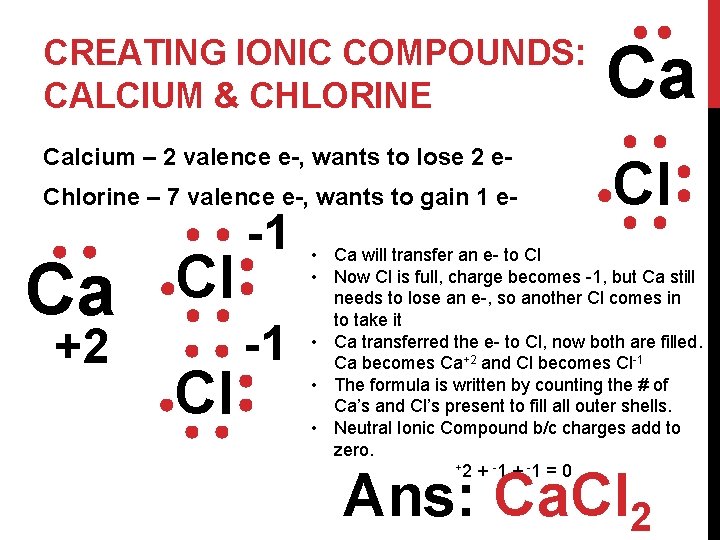
CREATING IONIC COMPOUNDS: CALCIUM & CHLORINE Ca Calcium – 2 valence e-, wants to lose 2 e- Cl Chlorine – 7 valence e-, wants to gain 1 e- Ca +2 Cl Cl -1 -1 • Ca will transfer an e- to Cl • Now Cl is full, charge becomes -1, but Ca still needs to lose an e-, so another Cl comes in to take it • Ca transferred the e- to Cl, now both are filled. Ca becomes Ca+2 and Cl becomes Cl-1 • The formula is written by counting the # of Ca’s and Cl’s present to fill all outer shells. • Neutral Ionic Compound b/c charges add to zero. + 2 + -1 = 0 Ans: Ca. Cl 2

CRISS CROSS WAY (DROP & SWAP): CALCIUM & CHLORINE • Write the symbols for Calcium & Chlorine • Determine charge for each atom • Criss-Cross charges (drop & swap) • Ignore + and – signs • Neutral Ionic Compound b/c charges add to zero. (1 x +2) + (2 x -1) 2 + -2 = 0 +2 Ca Cl Ca 1 Cl 2 Ca. Cl 2 -1

IONIC FORMULAS • Determine the compound formula when the following bond: • Magnesium & Sulfur • Aluminum & Oxygen • Sodium & Sulfur With polyatomic ions (drop & swap) • Strontium & Hydroxide • Potassium & Sulfate • Ammonia & Nitrate

MAGNESIUM & SULFUR Mg Magnesium – 2 valence e-, wants to lose 2 e. Sulfur – 6 valence e-, wants to gain 2 e- +2 -2 Mg S +2 Mg -2 S S • Mg will transfer an e- to S • Neither are full, so Mg will transfer another eto S • Now both are filled. Mg becomes Mg+2 and S becomes S-2 • The formula is written by counting the # of Mg’s and S’s present to fill all outer shells. • Because same number is needed, can simplify to 1 atom of each element. • Can also do by criss-crossing the charges, same answer • Neutral Ionic Compound b/c charges add to zero. (1 x +2) + (1 x -2) 2 + -2 = 0 Ans: Mg. S
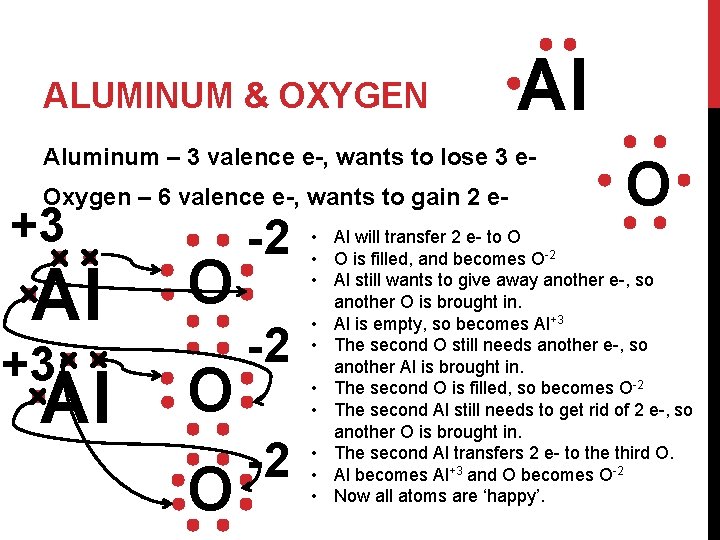
ALUMINUM & OXYGEN Al Aluminum – 3 valence e-, wants to lose 3 e. Oxygen – 6 valence e-, wants to gain 2 e- +3 Al O O O -2 -2 -2 O • Al will transfer 2 e- to O • O is filled, and becomes O-2 • Al still wants to give away another e-, so another O is brought in. • Al is empty, so becomes Al+3 • The second O still needs another e-, so another Al is brought in. • The second O is filled, so becomes O-2 • The second Al still needs to get rid of 2 e-, so another O is brought in. • The second Al transfers 2 e- to the third O. • Al becomes Al+3 and O becomes O-2 • Now all atoms are ‘happy’.

ALUMINUM & OXYGEN +3 Al -2 O Ans: Al 2 O 3 • The formula is written by counting the # of Al’s and O’s present to fill all outer shells. There were 2 Aluminum atoms and 3 oxygen atoms. So the answer is Al 2 O 3 • Can also do by criss-crossing the charges of each atom. You’d get the same answer Neutral Ionic Compound b/c charges add to zero. (2 x +3) + (3 x -2) 6 + -6 = 0

SODIUM & SULFUR Sodium – 1 valence e-, wants to lose 1 e. Sulfur – 6 valence e-, wants to gain 2 e- +1 Na S + Na -2 S -2 Na • • S Na will transfer 1 e- to S Na is empty, so becomes Na+ S isn’t filled, so another Na is brought in. The second Na transfers 1 e- to S. Na becomes Na+ and S becomes S-2 Now all atoms are ‘happy’. The formula is written by counting the # of Na’s and S’s present to fill all outer shells. There were 2 Sodium atoms and 1 sulfur atoms. So the answer is Na 2 S • Can also do by criss-crossing the charges of each atom. You’d get the same answer Neutral Ionic Compound b/c charges add to zero. (2 x +1) + (1 x -2) 2 + -2 = 0 Ans: Na 2 S
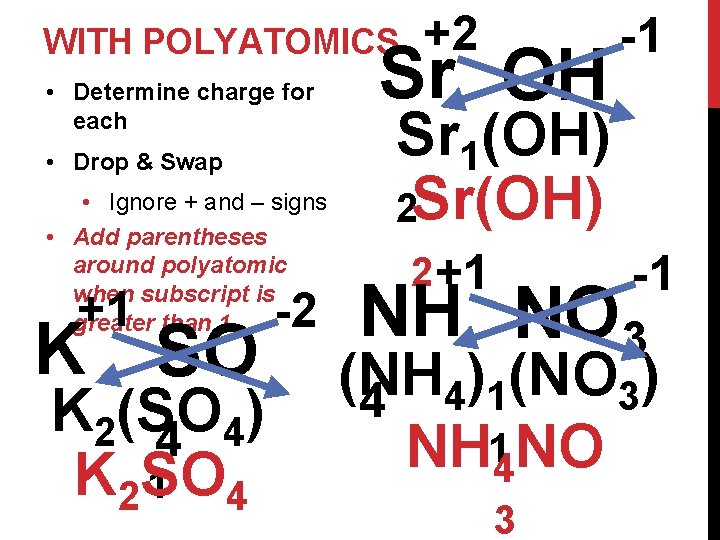
WITH POLYATOMICS • Determine charge for each • Drop & Swap • Ignore + and – signs • Add parentheses around polyatomic when subscript is greater than 1 +1 K SO K 2(SO ) 4 4 K 2 SO 1 4 -2 +2 Sr OH -1 Sr 1(OH) 2 Sr(OH) 2 +1 NH -1 NO 3 (NH ) (NO ) 3 4 4 1 NH 14 NO 3

- Slides: 22