Ionic Bonding Ionic Bonding Metals Electron donors Donate

Ionic Bonding

Ionic Bonding • Metals – Electron donors – Donate their valence electrons to become a positive ion (cation) • Nonmetals – Electron acceptors – Accept valence electrons to become a negative ion (anion)
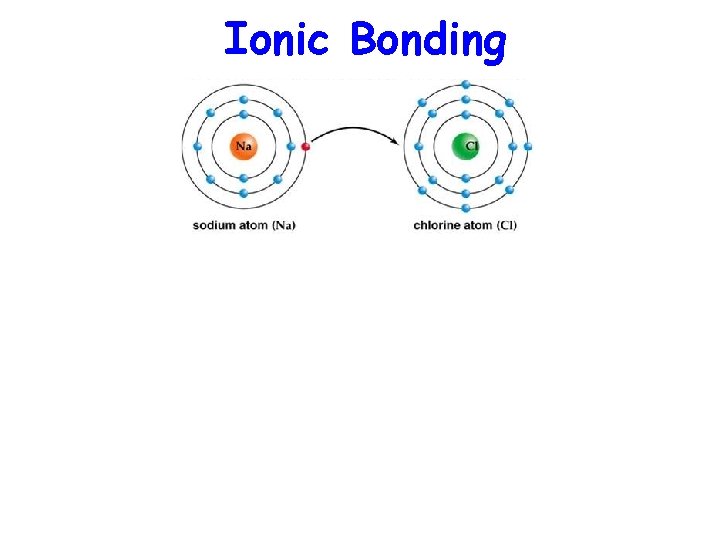
Ionic Bonding
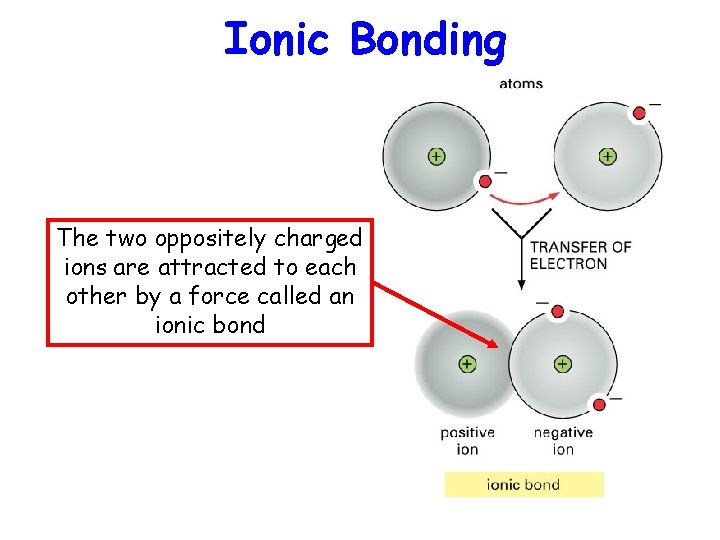
Ionic Bonding The two oppositely charged ions are attracted to each other by a force called an ionic bond

Monatomic Cations H+ Li+ Na+ K+ Mg 2+ Ca 2+ Ba 2+ Al 3+ Name Hydrogen Lithium Sodium Potassium Magnesium Calcium Barium Aluminum

Monatomic Anions F- Name Fluoride Cl- Chloride Br- Bromide I- Iodide O 2 - Oxide S 2 - Sulfide N 3 - Nitride P 3 - Phosphide
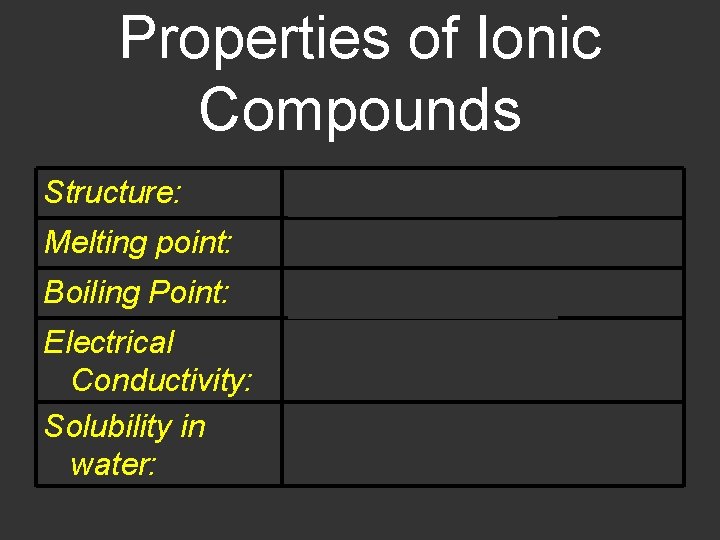
Properties of Ionic Compounds Structure: Crystalline solids Melting point: Generally high Boiling Point: Generally high Electrical Conductivity: Solubility in water: Excellent conductors, molten and aqueous Generally soluble

Na. Cl Crystal Lattice Ionic compounds form solids at SATP. Ionic compounds organize in a characteristic crystal lattice of alternating positive and negative ions. All lattices are arranged so that each ion has the greatest possible number of oppositely charged ions close by, while keeping similarly charged ions as far away as possible
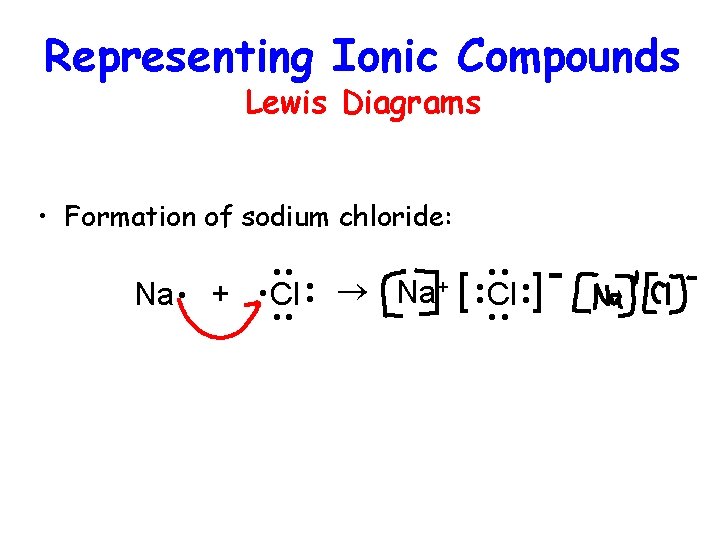
Representing Ionic Compounds Lewis Diagrams • Formation of sodium chloride: ® Na+ [ Cl ·· ] ·· ·· ·· Cl · Na · + ··
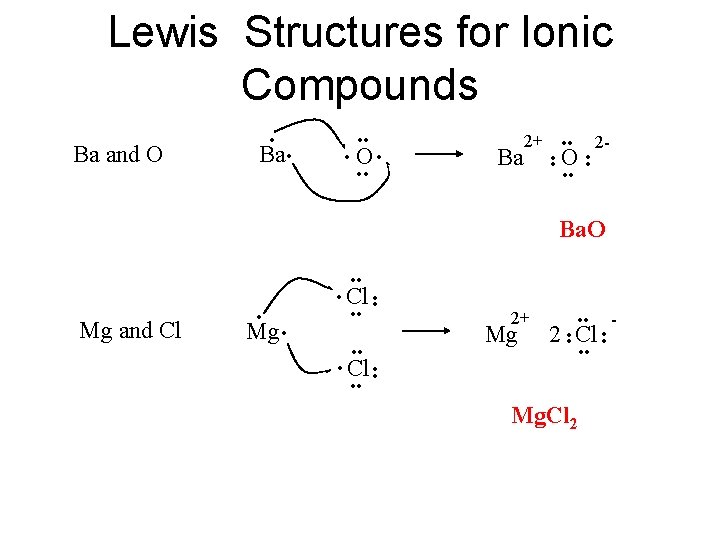
Lewis Structures for Ionic Compounds • O • • • 2+ • • Ba O • • 2 - • • Ba • • • Ba and O • Ba. O • • 2 Cl • Cl • • Mg. Cl 2 • • - • • Mg • 2+ • • Mg and Cl • • Cl • •

Representing Ionic Compounds Criss-Cross Method For monatomic ions: Take the absolute value of the ionic charge for the cation and make it the subscript for the anion and vice versa. Example: Al 3+ and Cl. The 3 becomes the subscript for the chloride ion and the 1 becomes understood for aluminum. Forming aluminum chloride: Al. Cl 3

Representing Ionic Compounds Criss-Cross Method For polyatomic ions: Additional step of including brackets around the polyatomic ion if it has a subscript other than one. Example: Mg 2+ and OHThe 2 becomes the subscript for the hydroxide ion, but brackets are needed to indicate 2 of each the O and the H. The 1 becomes the understood subscript for Mg. Forming magnesium hydroxide: Mg(OH)2

Polyatomic Ions NICK the CAMEL ate a CLAM for SUPPER in PHOENIX • Underlined letter represents the symbol of the element. • The consonants represent the number of oxygen • The vowels represent the negative charge. Eg. Underlined letter= N Number of consanants= 3 Number of vowels= 1 NO 3 - Nitrate represents oxygens represents charge

Practice Problems p. 68 #1, 4 p. 71 #1 -5 p. 73 #8 -14 p. 74 #1 -5
- Slides: 14