Ionic Bonding Covalent Bonding VSEPR theory Molecular Polarity

Ionic Bonding Covalent Bonding VSEPR theory Molecular Polarity Edward Wen, Ph. D

Why compounds are so Different from each other? • Table sugar C 12 H 22 O 11 : white crystal, soluble in water, aqueous solution not conducting electricity • Table salt Na. Cl: white crystal, soluble in water, aqueous solution conducting electricity • Butane C 4 H 10: colorless liquid, volatile, insoluble in water, flammable • Glycerol C 3 H 6 O 3: colorless liquid, not volatile, soluble in water, taste sweet 2

Chapter outline 1. Ionic bonding 2. • • • Covalent bonding Lewis structure Molecular geometry (VSEPR) Molecular polarity

Bonding Theories Bonding: the way atoms attach to make molecules • How and Why atoms attach together in the manner they do is central to chemistry Why studying chemical bonding? 1) predict the shapes of molecules and properties of substances 2) design and build molecules with particular sets of chemical and physical properties, for such as drug design in pharmaceutical industries. 4

Lewis Bonding Theory • Atoms bond because bonding results in a more stable Electron Configuration • by either transferring or sharing electrons so that all atoms obtain an Outer Shell with 8 electrons Octet Rule üSome exceptions: H, He, Li, Be. üHow to remember? Everyone wants to have an electron configuration like a Noble Gas 5

Lewis Symbols of Atoms also Electron Dot Symbols • Symbol of element : Nucleus and Inner electrons • Dots: around the Symbol as Valence electrons ü put one electron on each side first, then pair ü elements in the same group have the same number of valence electrons same Lewis dot symbols 6

Lewis Symbols of Ions • Cations (Cn+) have no valence electrons üValence electron(s) is lost • Anions (An-): 8 valence electrons üElectrons gained in the formation of the anion 7

Ionic Bonds Bonding : Metal + Nonmetal • Metal loses electrons to form cation • Nonmetal gains electrons to form anion Ionic bond results from + & - attraction (Coulombic force) ü larger charge = stronger attraction ü smaller ion = stronger attraction • Lewis Theory allow us to predict the correct formulas of ionic compounds 8

Ionic Compounds Ions: Metals (Cation Mx+) and Nonmetals (Anion Ny -) • No individual molecules!! • have a 3 -dimensional array of cations and anions made of formula units: Na. Cl, Mg. O • Na+ Cl- Na+ Cl • Cl- Na+ • Na+ Cl- 9
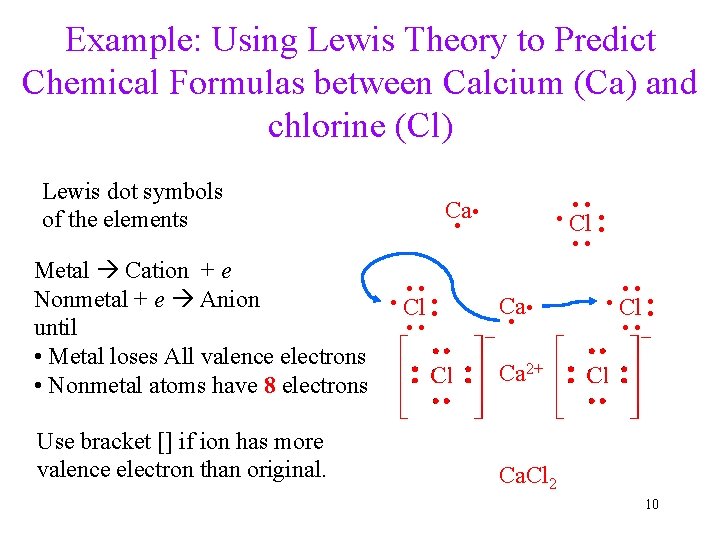
Example: Using Lewis Theory to Predict Chemical Formulas between Calcium (Ca) and chlorine (Cl) Use bracket [] if ion has more valence electron than original. ∙ Cl ∙∙ ∙∙ ∙∙ Metal Cation + e Nonmetal + e Anion until • Metal loses All valence electrons • Nonmetal atoms have 8 electrons ∙ ∙ ∙ Cl ∙∙ ∙∙ ∙∙ Ca ∙ Cl ∙∙ ∙∙ ∙∙ Lewis dot symbols of the elements Ca 2+ Ca. Cl 2 10

More Example: Lewis Structures for Ionic Compound Al 2 S 3 First, Number of valence electrons: Al = ___, S = ___ charge of ions: Al ion = ___, S ion = ___ number of valence electrons on the ION: Al ion = ___, S ion = ___. Use bracket [] if ion has more valence electron than original. 11

Covalent Bonds Nonmetal atoms can form chemical bonds between them, such as water, carbon dioxide, and many others Nonmetal atoms DO NOT tend to lose its valence electrons How two NONMETAL atoms achieve OCTET (like noble gas atoms)? Nonmetal atoms _____ their valence electrons to form chemical bond 12

Lewis Formulas of Covalent Compounds Lewis structure shows: • pattern of Valence electron distribution • understand the bonding in many compounds • predict Shapes of molecules • predict Properties of molecules and how they will interact together 13

Single Covalent Bonds • Two atoms share ONE pair of electrons ü 2 electrons • One atom may have more than one single bond • Lewis structure: F 2 and H 2 O 14

Double Covalent Bond • two atoms sharing TWO pairs of electrons ü 4 electrons • shorter and stronger than single bond: O 2 • Lewis structure: O 2 15

Triple Covalent Bond • two atoms sharing 3 pairs of electrons: 6 electrons • shorter and stronger than single or double bond • Lewis structure: N 2 16

Practice: Which one(s) violate the Octet Rule? 17
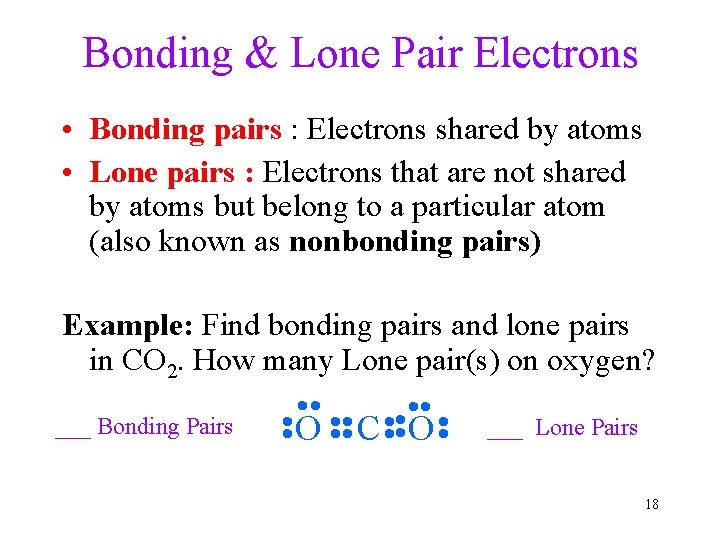
Bonding & Lone Pair Electrons • Bonding pairs : Electrons shared by atoms • Lone pairs : Electrons that are not shared by atoms but belong to a particular atom (also known as nonbonding pairs) Example: Find bonding pairs and lone pairs in CO 2. How many Lone pair(s) on oxygen? ___ Bonding Pairs • • • • • O • • C • • • O • • ___ Lone Pairs 18

Covalent Bonding within Polyatomic Ions • Polyatomic ion: Atoms within the polyatomic ion are held together by covalent bonds • form ionic bonds with ions of opposite charge üExample: Na+···NO 3 - ; NH 4+···SO 42 - Example: • Covalent N-H bonds within NH 4+ 19

Lewis Structures: common bonding patterns üC = 4 bonds & 0 lone pairs Ø 4 bonds = 4 single, or 2 double, or single + triple, or 2 single + double üN = 3 bonds & 1 lone pair üO = 2 bonds & 2 lone pairs üH and Halogen (F, Cl, Br) = 1 bond, üBe = 2 bonds & 0 lone pairs, üB = 3 bonds & 0 lone pairs (Not Octet in B!) B C N O F 20

HONC Rule for most neutral molecules to help identify central atom • The numbers of covalent bonds formed on #bonds: _____ H O N C Extended HONC rule: F S? Cl? Se? Br? P? As? Si 21

Lewis Structures for Covalent Molecules 1) Calculate the total number of Valence electrons available for bonding ü use group number of periodic table 2) Arrange the atoms and link with _______ bonds first Usually: Atoms capable of forming (more/fewer) bonds be the center (HONC rule) 22

Lewis Structures for Covalent Molecules 3) Attach atoms with pairs of electrons ü Start with _______ atoms to Octet ü H only wants 2 electrons ü then attach to Central atoms 4) Check if ALL atoms are ____tet. If central atoms are NOT ____tet, share electrons (to make multiple bonds) to complete the Octets of all the atoms 23

Practice: Drawing Lewis Structures Step by step! 1) 2) 3) 4) Count all valence electrons CO 2 Lay out electrons and link with single bonds (HONC) Attach atoms with pairs of electrons to complete OCTET, starting with terminal atoms Complete OCTET for central atoms, building multiple bonds if necessary (except for H) 24

Lewis Structures for Polyatomic Ions Same procedure, #charge affect #Valence electrons • Polyatomic Cations: take away electron from the total for each positive charge NH 4+ #Valence electrons = 5 + 4 x 1 ____ = 8 • Polyatomic Anions: add electron to the total for each negative charge SO 32 - #Valence electrons = 6 + 3 x 6 _____ = 26 HONC rule often does not apply!! 25

Example: Lewis structure for NO 31) 2) 3) 4) 5) Count all valence electrons. Lay out electrons and link with single bonds (which is central atom? ) Attach atoms with pairs of electrons to complete OCTET, starting with terminal atoms Complete OCTET for central atoms, building multiple bonds if necessary (except for H) Use bracket and label the charge 26

Exceptions to the Octet Rule • H & Li, lose one electron to form cation ü Li now has electron configuration like He ü H can also share or gain one electron to have configuration like He • Be : shares 2 electrons to form 2 single bonds • B : shares 3 electrons to form 3 single bonds • expanded octets for elements in Period 3 or below ü using empty valence d orbitals • some molecules have odd numbers of electrons ü NO 27

Molecular Geometry • Molecules are 3 -dimensional objects • Shape of a molecule like Geometric figures Molecular Geometry indicates • Positions of the Surrounding atoms with the Central atom in the center of the figure. Linear, Trigonal, Tetrahedral, Pentagonal, etc. • Bond Angles : angles between adjacent bonds. in water molecule ∠H-O-H = 105° 28

Valence Shell Electron Pair Repulsion (VSEPR) theory • Electron pairs attached to the same atom, either bonding or nonbonding, have repulsion against each other and stay apart as much as possible • The number of electron pairs (“electron groups”) on central atoms leads to specific molecular geometry (bond angles). 29

Molecular Geometry depends on #Electron Groups on Central Atom Each Bond (Shared e-) counts as 1 Electron group ü Single bond ü Double or Triple also as ____ electron group Lone Pair “: ” • counts as ONE electron group ü lone pairs “occupy space” around the central atom • take up slightly more space than bonding pairs ü Affects bond angles 30

Example: Use Lewis Structure to find #Electron Groups on Central Atom • ____ electron groups on each carbon atom 31
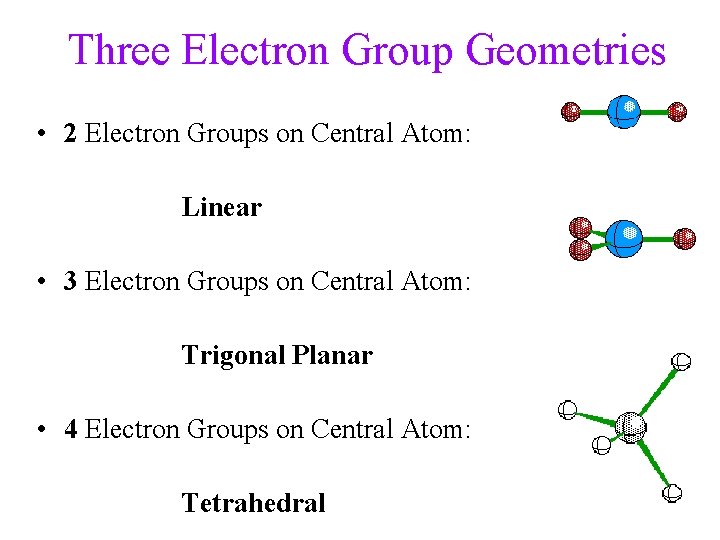
Three Electron Group Geometries • 2 Electron Groups on Central Atom: Linear • 3 Electron Groups on Central Atom: Trigonal Planar • 4 Electron Groups on Central Atom: Tetrahedral 32
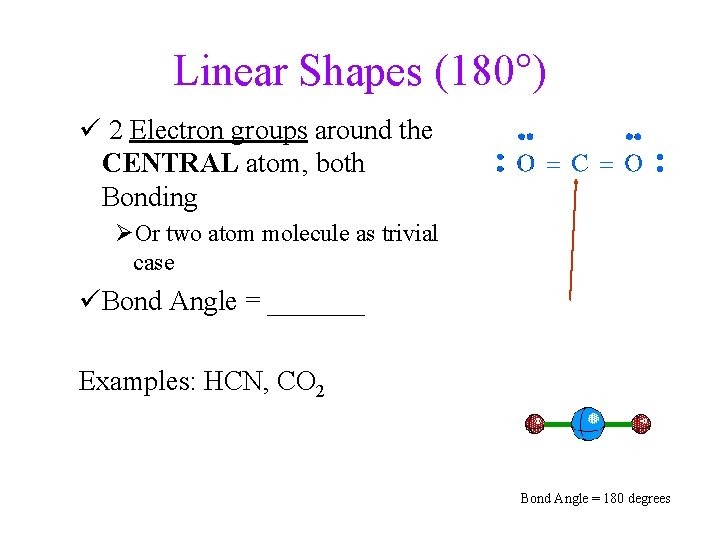
Linear Shapes (180°) ü 2 Electron groups around the CENTRAL atom, both Bonding ØOr two atom molecule as trivial case üBond Angle = _______ Examples: HCN, CO 2 Bond Angle = 180 degrees
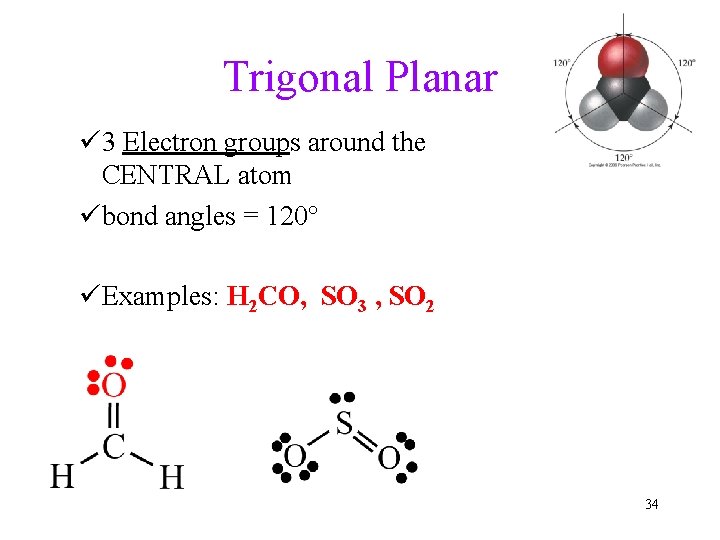
Trigonal Planar ü 3 Electron groups around the CENTRAL atom übond angles = 120° üExamples: H 2 CO, SO 3 , SO 2 34
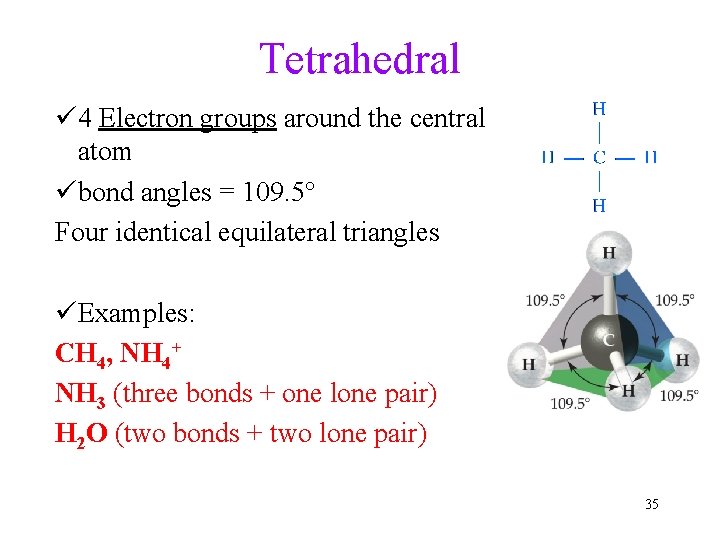
Tetrahedral ü 4 Electron groups around the central atom übond angles = 109. 5° Four identical equilateral triangles üExamples: CH 4, NH 4+ NH 3 (three bonds + one lone pair) H 2 O (two bonds + two lone pair) 35
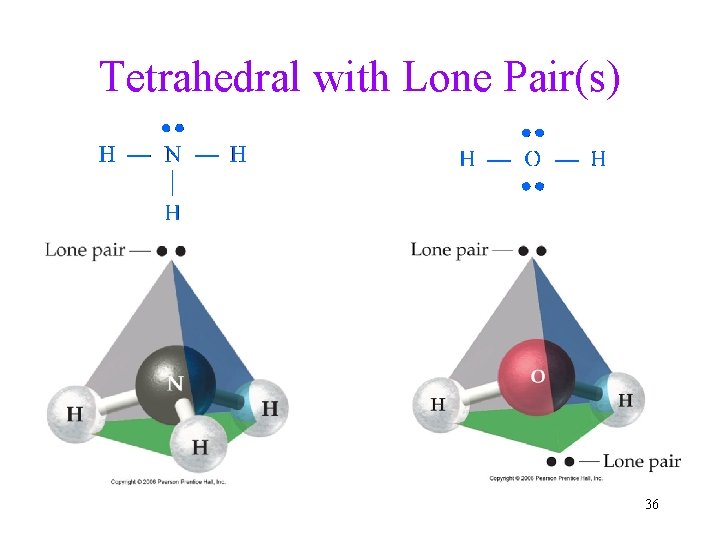
Tetrahedral with Lone Pair(s) 36

Molecular Geometry: Linear • • • #Electron Groups on Central Atom = ___ Bonding Groups = 2 Lone Pairs = 0 Electron Geometry = Linear Angle between Electron Groups = 180° 37
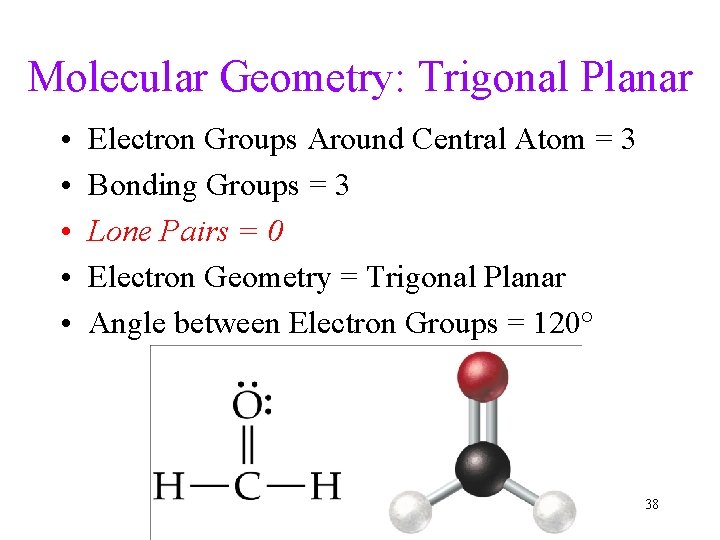
Molecular Geometry: Trigonal Planar • • • Electron Groups Around Central Atom = 3 Bonding Groups = 3 Lone Pairs = 0 Electron Geometry = Trigonal Planar Angle between Electron Groups = 120° 38

Molecular Geometry: Trigonal Bent • • • Electron Groups Around Central Atom = 3 Bonding Groups = 2 Lone Pairs = 1 Electron Geometry = Trigonal Planar Angle between Electron Groups = 120° 39
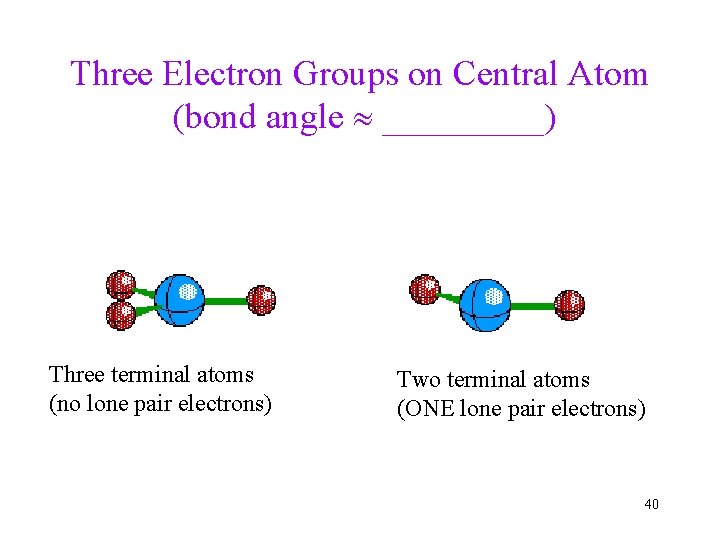
Three Electron Groups on Central Atom (bond angle _____) Three terminal atoms (no lone pair electrons) Two terminal atoms (ONE lone pair electrons) 40
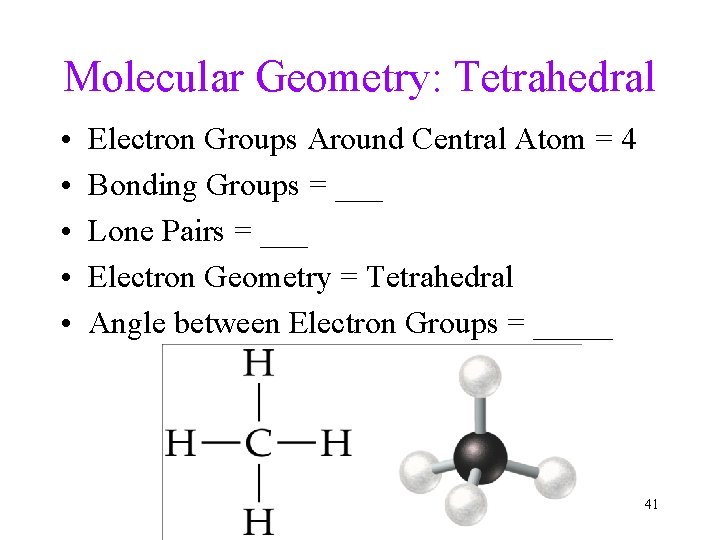
Molecular Geometry: Tetrahedral • • • Electron Groups Around Central Atom = 4 Bonding Groups = ___ Lone Pairs = ___ Electron Geometry = Tetrahedral Angle between Electron Groups = _____ 41

Molecular Geometry: Trigonal Pyramid • • • Electron Groups Around Central Atom = 4 Bonding Groups = ___ Lone Pairs = ___ Electron Geometry = Tetrahedral Angle between Electron Groups = 109. 5° 42

Molecular Geometry: Tetrahedral Bent • • • Electron Groups Around Central Atom = 4 Bonding Groups = 2 Lone Pairs = 2 Electron Geometry = Tetrahedral Angle between Electron Groups = 109. 5° 43
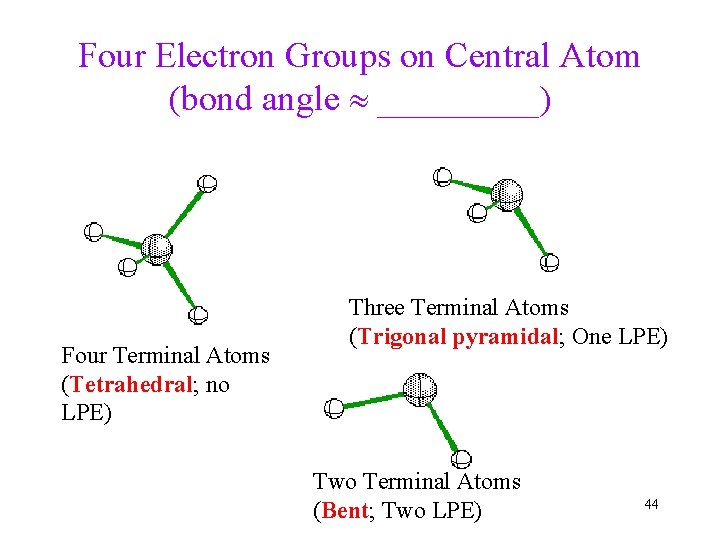
Four Electron Groups on Central Atom (bond angle _____) Four Terminal Atoms (Tetrahedral; no LPE) Three Terminal Atoms (Trigonal pyramidal; One LPE) Two Terminal Atoms (Bent; Two LPE) 44

Example: Shape and Bond Angle • sulfur trioxide • sulfite ion • carbon disulfide • ammonia • • Trigonal planar, 120° Trigonal pyramidal, 109. 5° Linear, 180° Trigonal pyramidal, 109. 5° 45
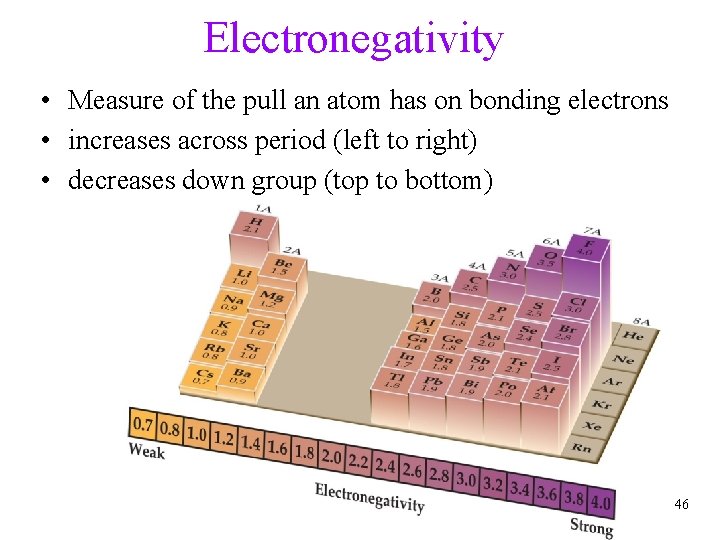
Electronegativity • Measure of the pull an atom has on bonding electrons • increases across period (left to right) • decreases down group (top to bottom) 46

Bond Polarity d+ H—F d- • bonding between unlike atoms results in unequal sharing of the electrons ü Atom with high electronegativity pulls the electrons in the bond (closer, further) to its side ü Atom that pulls electrons stronger has (larger, lower) electron density than the other Bond Polarity ü the end with the higher electronegativity gets a partial negative charge (d-) and the end that is electropositive gets a partial positive charge (d+) 47
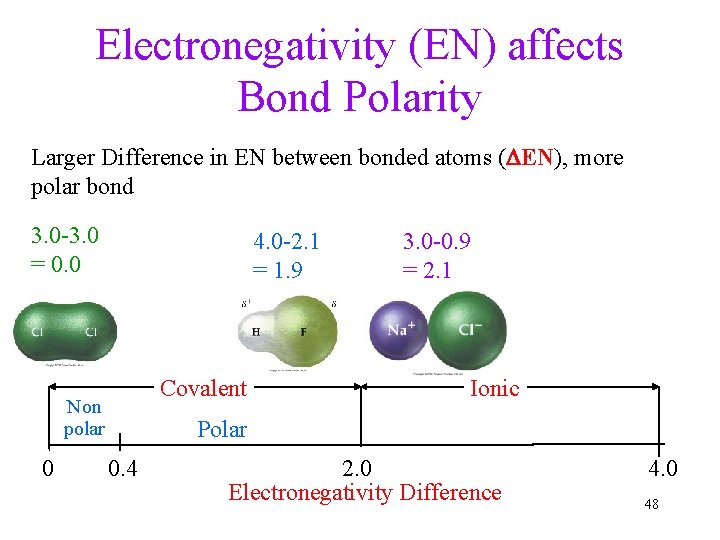
Electronegativity (EN) affects Bond Polarity Larger Difference in EN between bonded atoms (DEN), more polar bond 3. 0 -3. 0 = 0. 0 4. 0 -2. 1 = 1. 9 Covalent Non polar 0 3. 0 -0. 9 = 2. 1 Ionic Polar 0. 4 2. 0 Electronegativity Difference 4. 0 48

Bond Polarity: Dipole Moments • Dipole: a material with positively and negatively charged ends • Polar bonds or molecules have one end slightly positive, d+; and the other slightly negative, dü not “full” charges, come from nonsymmetrical electron distribution • Dipole Moment (m) : a measure of the size of the polarity ü measured in Debyes, D 49
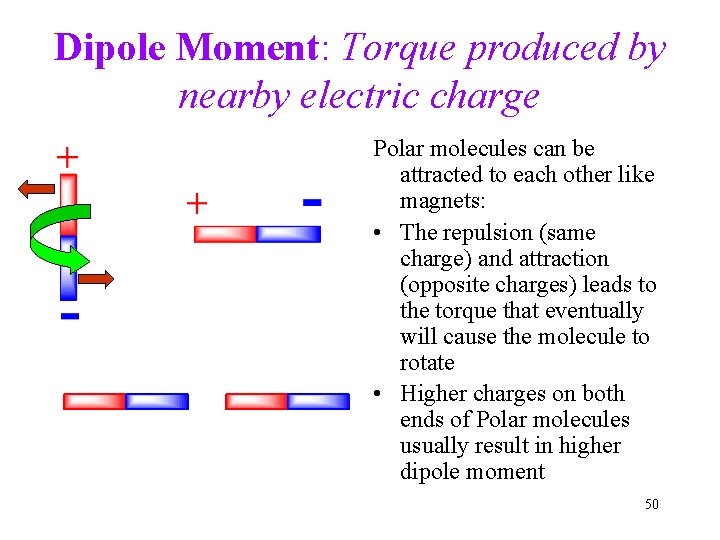
Dipole Moment: Torque produced by nearby electric charge Polar molecules can be attracted to each other like magnets: • The repulsion (same charge) and attraction (opposite charges) leads to the torque that eventually will cause the molecule to rotate • Higher charges on both ends of Polar molecules usually result in higher dipole moment 50

Polarity of Molecules Polarity of molecule requires 1) Polar bonds Ø electronegativity difference - theory Ø bond dipole moments - measured 2) Unsymmetrical shape so that Bond Polarity won’t offset each other Ø Bent and Trigonal Pyramidal molecules are POLAR Ø vector addition • Polarity affects the intermolecular forces of attraction 51
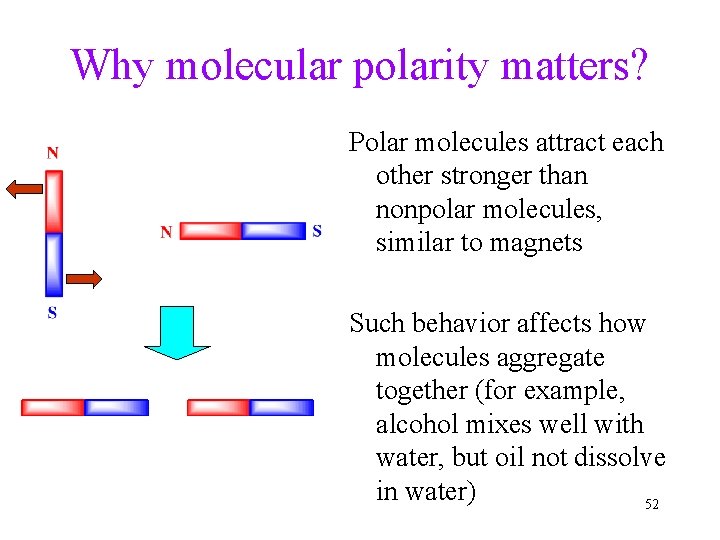
Why molecular polarity matters? Polar molecules attract each other stronger than nonpolar molecules, similar to magnets Such behavior affects how molecules aggregate together (for example, alcohol mixes well with water, but oil not dissolve in water) 52

Static charges attract Polar Molecule • Polar molecules can be attracted to Static charge Example: Styrofoam attracts to our clothing because the attractive force, due to its closer distance to the charge, overcomes the repulsion. • Online Demonstration (at 2: 27) 53
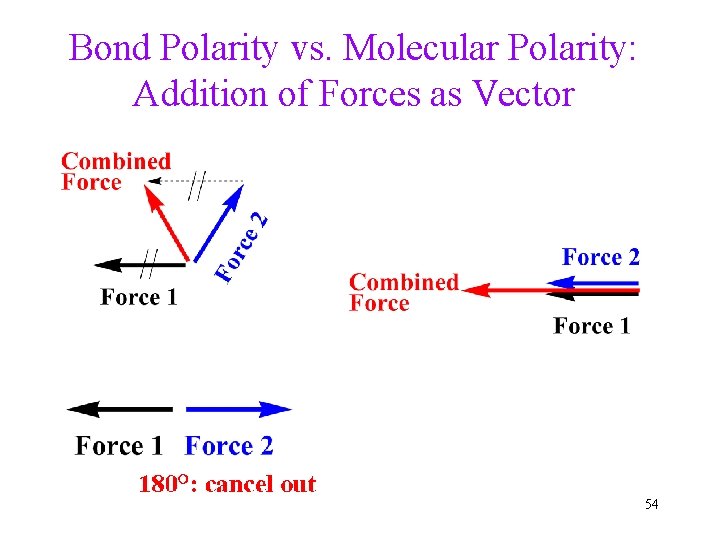
Bond Polarity vs. Molecular Polarity: Addition of Forces as Vector 54
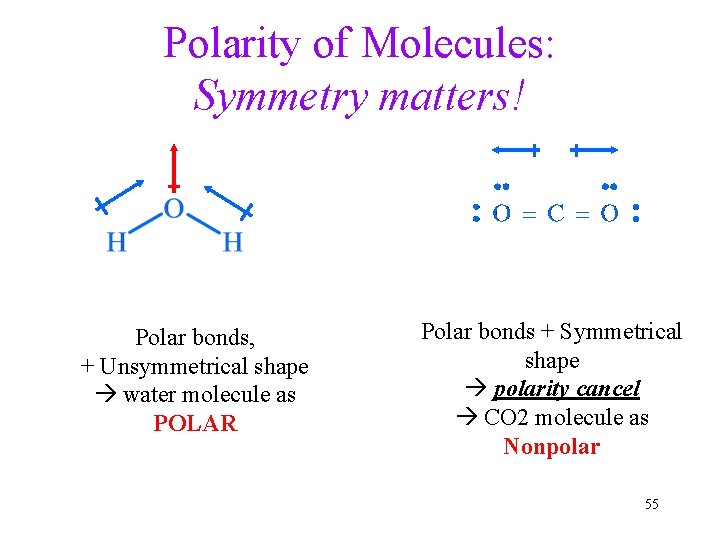
Polarity of Molecules: Symmetry matters! Polar bonds, + Unsymmetrical shape water molecule as POLAR Polar bonds + Symmetrical shape polarity cancel CO 2 molecule as Nonpolar 55
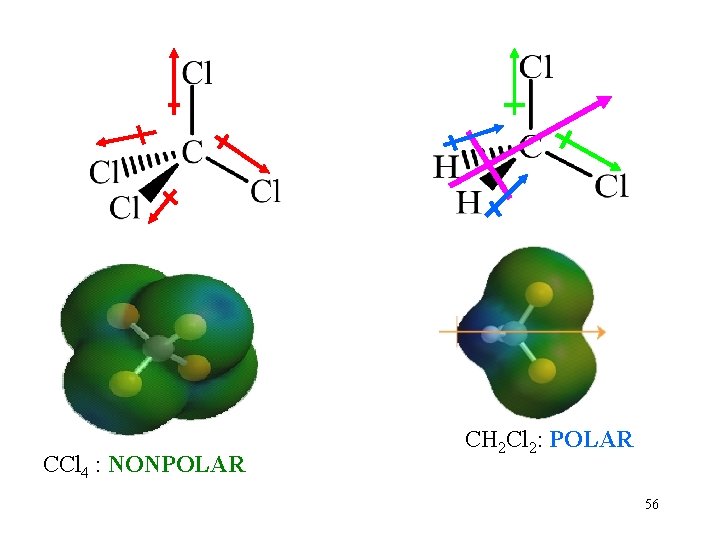
CCl 4 : NONPOLAR CH 2 Cl 2: POLAR 56
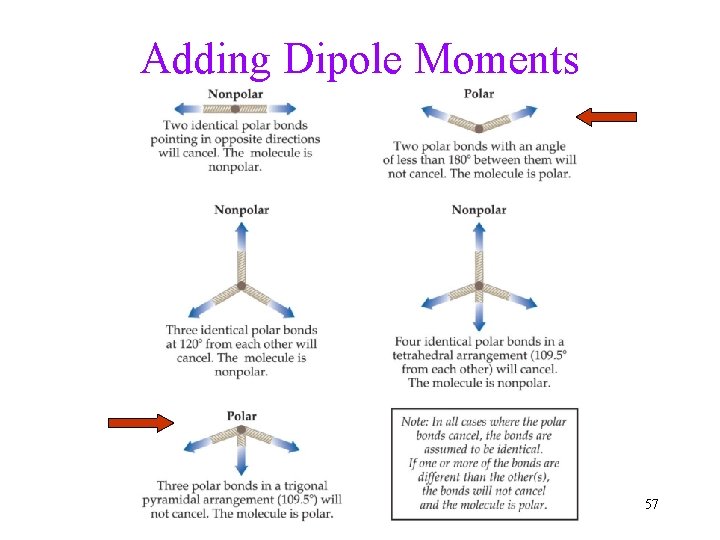
Adding Dipole Moments 57

How to Determine the Molecular Polarity • Step 1: First, draw Lewis structure • Step 2: use VSEPR theory to determine the molecular geometry/shape • Step 3: consider the bond dipole moment addition. 58
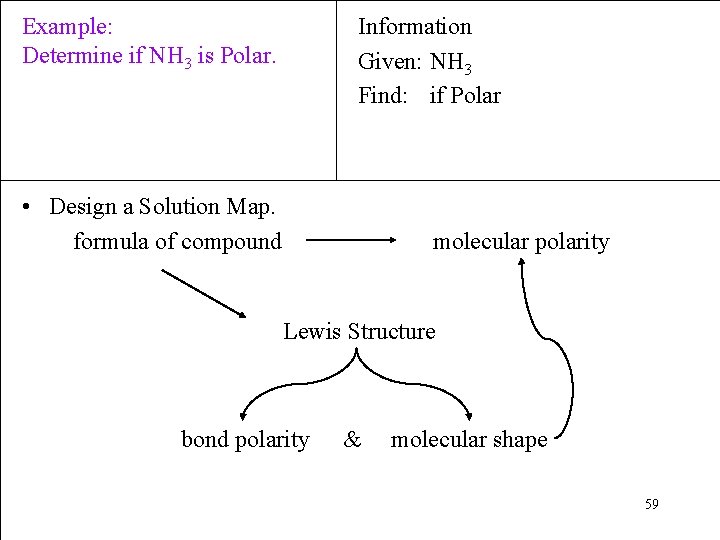
Example: Determine if NH 3 is Polar. Information Given: NH 3 Find: if Polar • Design a Solution Map. formula of compound molecular polarity Lewis Structure bond polarity & molecular shape 59

Practice: Polar or Nonpolar? • • • sulfur dioxide CH 2 O carbon disulfide Silicon tetrafluoride hydrogen sulfide phosphorous trichloride sulfur dichloride CHCl 3 Sulfur trioxide Hydrogen cyanide Hint: molecular geometry • Bent • Trigonal planar • Linear • Tetrahedral • Bent • Trigonal pyramidal • Bent • Trigonal planar 60 • Linear • tetrahedral
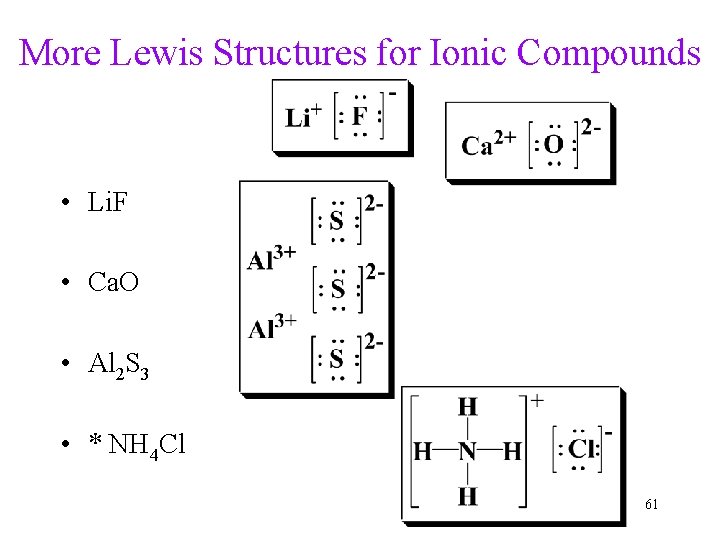
More Lewis Structures for Ionic Compounds • Li. F • Ca. O • Al 2 S 3 • * NH 4 Cl 61
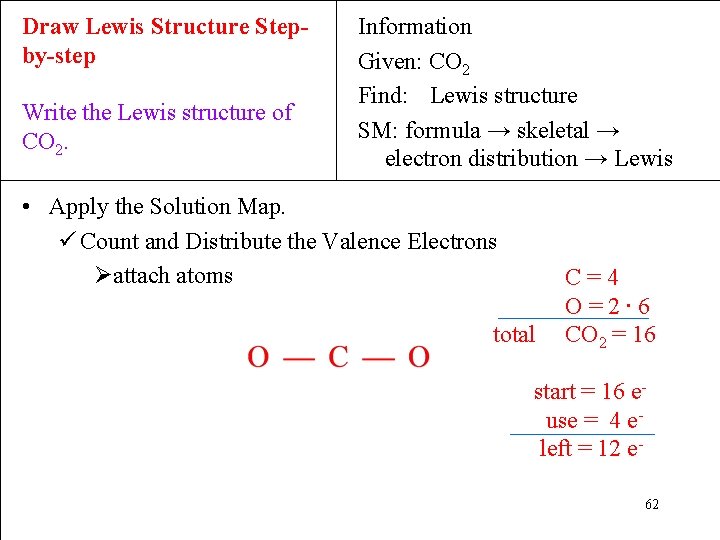
Draw Lewis Structure Stepby-step Write the Lewis structure of CO 2. Information Given: CO 2 Find: Lewis structure SM: formula → skeletal → electron distribution → Lewis • Apply the Solution Map. ü Count and Distribute the Valence Electrons Øattach atoms total C=4 O=2∙ 6 CO 2 = 16 start = 16 euse = 4 eleft = 12 e 62
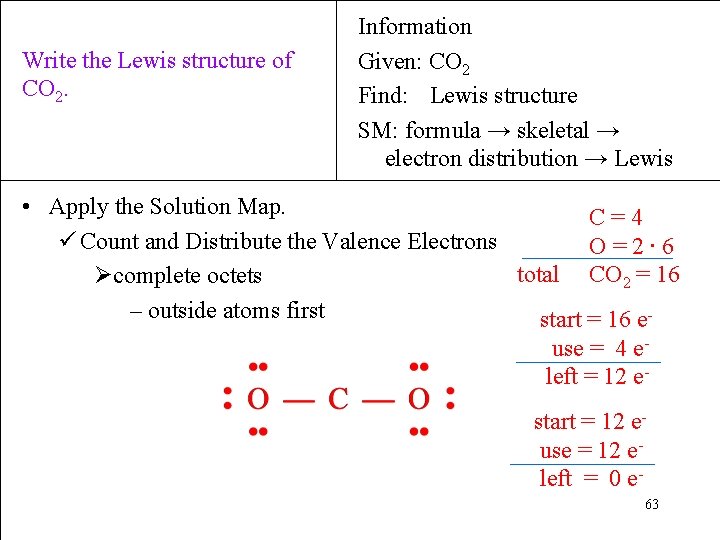
Write the Lewis structure of CO 2. Information Given: CO 2 Find: Lewis structure SM: formula → skeletal → electron distribution → Lewis • Apply the Solution Map. C=4 ü Count and Distribute the Valence Electrons O=2∙ 6 total CO 2 = 16 Øcomplete octets – outside atoms first start = 16 euse = 4 eleft = 12 estart = 12 euse = 12 eleft = 0 e 63
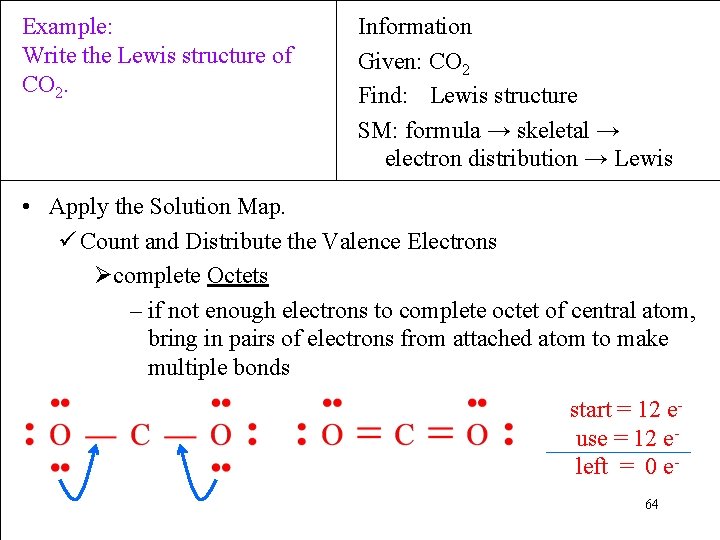
Example: Write the Lewis structure of CO 2. Information Given: CO 2 Find: Lewis structure SM: formula → skeletal → electron distribution → Lewis • Apply the Solution Map. ü Count and Distribute the Valence Electrons Øcomplete Octets – if not enough electrons to complete octet of central atom, bring in pairs of electrons from attached atom to make multiple bonds start = 12 euse = 12 eleft = 0 e 64

Example: Write the Lewis structure of CO 2. Information Given: CO 2 Find: Lewis structure SM: formula → skeletal → electron distribution → Lewis • Check total Start C = 4 e. O = 2 ∙ 6 e. CO 2 = 16 e- End bonding = 4 ∙ 2 elone pairs = 4 ∙ 2 etotal CO 2 = 16 e- The skeletal structure is symmetrical. All the electrons are accounted for. 65

Example: Determine if NH 3 is Polar. Information Given: NH 3 Find: if Polar SM: formula → Lewis → Polarity & Shape → Molecule Polarity • Apply the Solution Map. ü Draw the Lewis Structure ØCount all the VE ØLay out the atoms ØSingle bond first Øcomplete octets: from terminal atoms first; double or triple bond for the central atoms ∙∙ 66
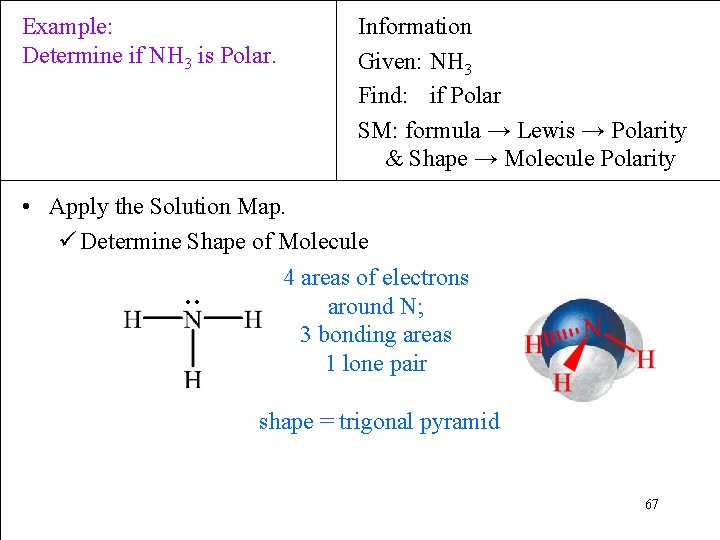
Example: Determine if NH 3 is Polar. Information Given: NH 3 Find: if Polar SM: formula → Lewis → Polarity & Shape → Molecule Polarity • Apply the Solution Map. ü Determine Shape of Molecule 4 areas of electrons around N; ∙∙ 3 bonding areas 1 lone pair shape = trigonal pyramid 67
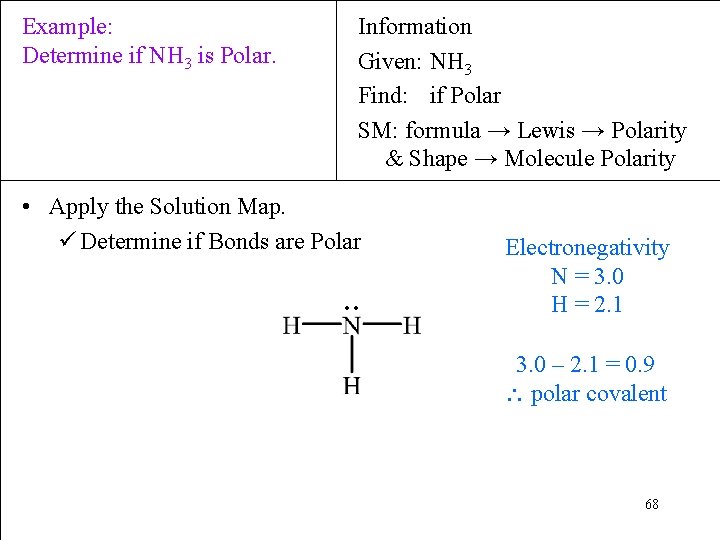
Example: Determine if NH 3 is Polar. Information Given: NH 3 Find: if Polar SM: formula → Lewis → Polarity & Shape → Molecule Polarity • Apply the Solution Map. ü Determine if Bonds are Polar ∙∙ Electronegativity N = 3. 0 H = 2. 1 3. 0 – 2. 1 = 0. 9 polar covalent 68
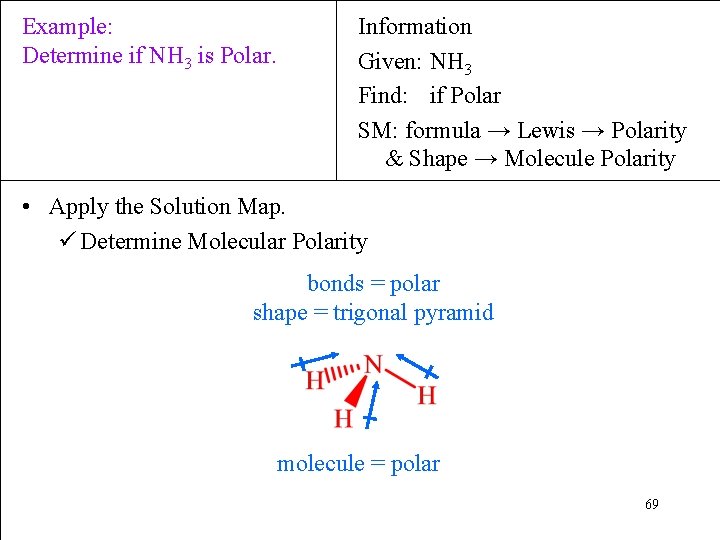
Example: Determine if NH 3 is Polar. Information Given: NH 3 Find: if Polar SM: formula → Lewis → Polarity & Shape → Molecule Polarity • Apply the Solution Map. ü Determine Molecular Polarity bonds = polar shape = trigonal pyramid molecule = polar 69
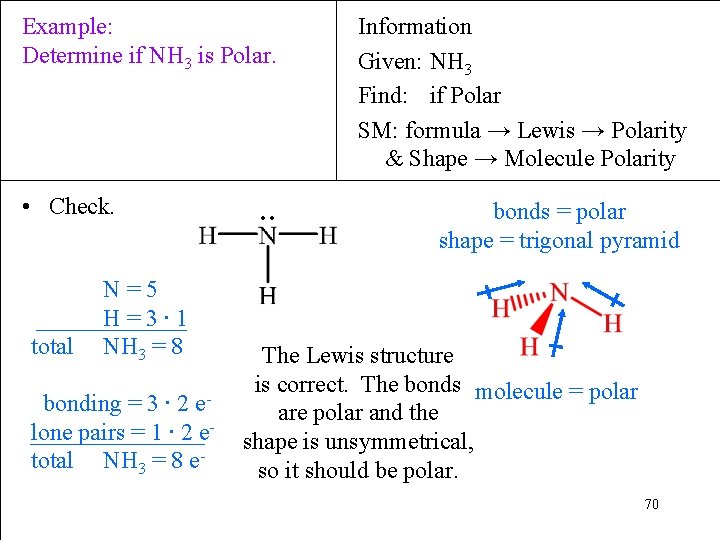
Example: Determine if NH 3 is Polar. • Check. total N=5 H=3∙ 1 NH 3 = 8 bonding = 3 ∙ 2 elone pairs = 1 ∙ 2 etotal NH 3 = 8 e- ∙∙ Information Given: NH 3 Find: if Polar SM: formula → Lewis → Polarity & Shape → Molecule Polarity bonds = polar shape = trigonal pyramid The Lewis structure is correct. The bonds molecule = polar are polar and the shape is unsymmetrical, so it should be polar. 70
- Slides: 70