Ionic Bonding Chemistry I Ionic Bonding Ionic Bonding

Ionic Bonding Chemistry I

Ionic Bonding

Ionic Bonding and the Octet Rule • Octet Rule: Atoms will lose, gain, or share electrons to achieve an octet (eight valence electrons; a noble gas configuration)

• In ionic bonding, – Metal atoms lose electrons to become like the noble gas immediately before them in the periodic table • Metals become positively charged cations Example: Mg: [Ne]3 s 2 Mg+: [Ne]3 s 1 Mg 2+: [Ne] not stable

– Nonmetal atoms gain electrons to become like the noble gas following them on the periodic table • Nonmetals become negatively charged anions Example Cl: [Ne]3 s 23 p 5 Cl-: [Ne]3 s 23 p 6 = [Ar] stable

• We can predict how many electrons an atom will lose or gain to achieve a stable, noble gas configuration, therefore, we can also predict the ionic charge
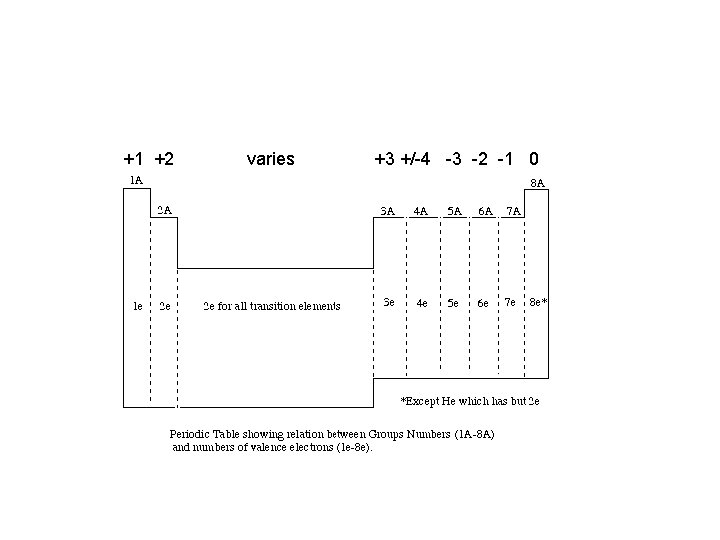
+1 +2 varies +3 +/-4 -3 -2 -1 0

Representing the formation of an ionic bond with Lewis structures • Draw the Lewis structure for each atom in the compound • Show the transfer of valence electrons from the metal to the nonmetal with an arrow • Continue adding atoms and showing transfers of electrons until the total number of electrons lost equals the total number of electrons gained. • Rewrite the dot structures showing the ions formed – All metal ions should have no dots and have a positive charge – All nonmetal ions should have 8 dots and have a negative charge

Examples

Writing formulas for ionic compounds • Chemical formulas – Represent the composition of substances – Show the kinds and numbers of atoms in the smallest representative unit of a substance.

• Formula Unit – The smallest representative unit of an ionic compound – The lowest whole number ratio of ions in the compound – Does not show the ionic charges
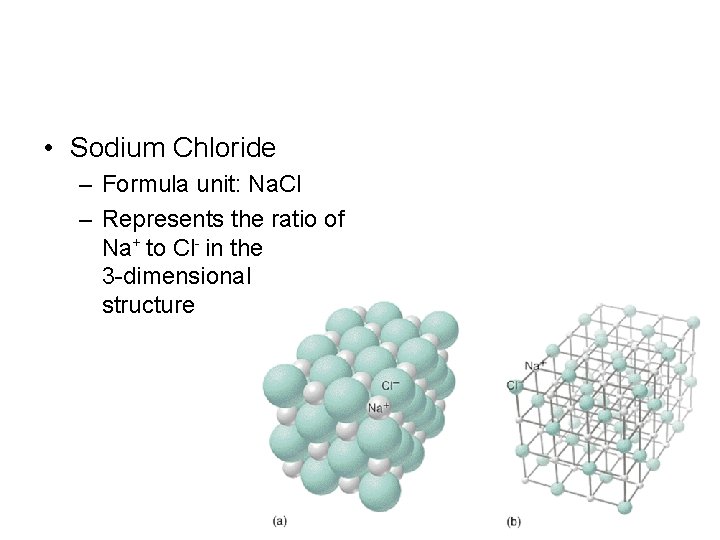
• Sodium Chloride – Formula unit: Na. Cl – Represents the ratio of Na+ to Cl- in the 3 -dimensional structure

• Mono-: one • Monatomic ions – Composed of only one atom which has an overall + or – charge – Examples: Na+1, O-2 • Poly-: many • Polyatomic ions – Composed of a group of atoms (many) with an overall charge – May contain subscripts (which you cannot change!) – Will usually contain at least two different elements – Examples: OH-1, CO 3 -2
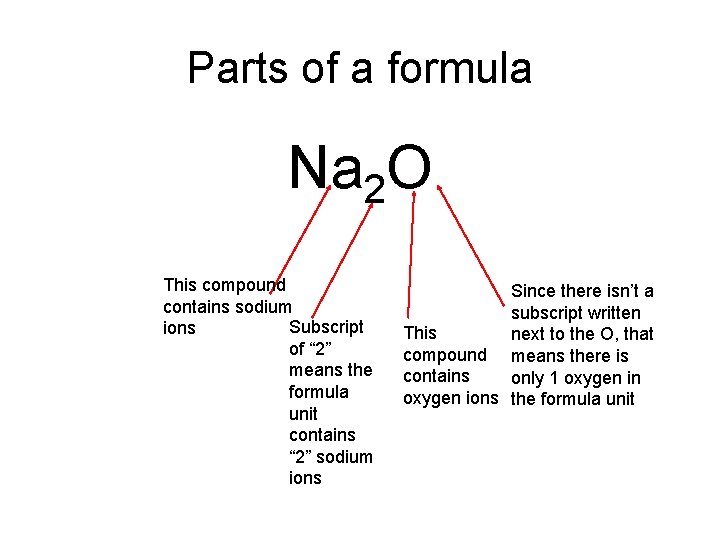
Parts of a formula Na 2 O This compound contains sodium Subscript ions of “ 2” means the formula unit contains “ 2” sodium ions Since there isn’t a subscript written This next to the O, that compound means there is contains only 1 oxygen in oxygen ions the formula unit
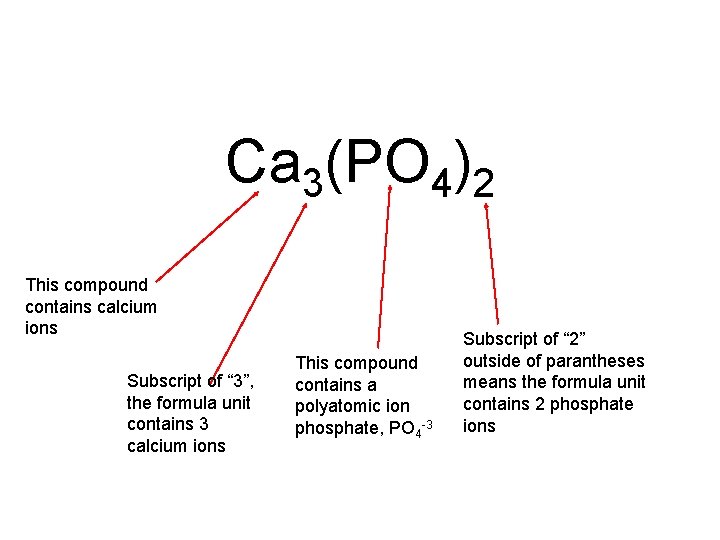
Ca 3(PO 4)2 This compound contains calcium ions Subscript of “ 3”, the formula unit contains 3 calcium ions This compound contains a polyatomic ion phosphate, PO 4 -3 Subscript of “ 2” outside of parantheses means the formula unit contains 2 phosphate ions

Na 2 SO 4 This compound contains the polyatomic ion sulfate, SO 4 -2 Since there are no parantheses, the formula unit contains “ 1” sulfate ion

Rules for writing ionic formulas • 1) The positively charged ion is written first

• 2) compounds are neutral, so the algebraic sum of the charges must add to zero (the total positive charge must cancel out the total negative charge)

• 3) never write “ 1” as a subscript

• 4)Put parantheses around polyatomic ions if they need a subscript (other than “ 1”) •

• 5)Reduce subscripts to simplest form

Other things to keep in mind… • Any ionic compound will be made of only one type of positive ion with one type of negative ion • You must know the charge of each ion – Predict monatomic ion charges from periodic table – Look up charges of polyatomic ions in chart

Shortcut: the crisscross method • Cross down the numerical value of each charge to become the subscript of the other ion • Make sure you consider if you have the simplest ratio; if not, reduce it
- Slides: 23